Research Article - Journal of Food Technology and Preservation (2020) Volume 4, Issue 1
Storage parameters to preserve fresh Smallanthus sonchifolius inulin in Andahuaylas.
David Juan RH*, Thomas AV, Denis Hernan GM, Gina Genoveva TR, Candida LLUniversidad Nacional José María Arguedas, Facultad de Ingenierías, Escuela Profesional de Ingeniería Agroindustrial, Av. Perú S/N, Talavera, Andahuaylas, Apurímac, Peru
- *Corresponding Author:
- David Juan RH
Universidad Nacional José María Arguedas,
Facultad de Ingenierías, Escuela Profesional de Ingeniería
Agroindustrial, Av. Perú
Tel: +51 (83) 421 992
E-mail: davisrh22@gmail.com
Accepted Date: 10 February, 2020
Abstract
Yacon storage parameters were determined to preserve inulin in amounts similar to that harvested.The variety under study was "Yurac Llajum", collected with 7.5 months of physiological maturity.Storage was carried out for 60 days, in conditioned rooms of temperature, relative humidity and light intensity, each factor in two levels. To obtain responses of the conservation of the inulin content of the yacon, to analyze the analysis of the inulin content at the beginning and end of storage, the results were analyzed by means of a 2 ^ 3 factorial arrangement, which obtained a response surface that the fresh yacon informed us It has obligations at a temperature of 15°C with relative humidity of 50% and a light intensity of 400 lm to obtain a variation of 50.16% in inulin content. Concluding that the fresh yacon storage parameters in a controlled manner allowed a variation of up to 50.16%.
Keywords
Storage, Conservation, Inulin, Yacon
Introduction
Yacon (Smallanthus sonchifolius) is a plant whose root is mainly attributed to its high content of oligofructans, which cannot be hydrolyzed by the human organism and cross the digestive tract without being metabolized, Low calories than sucrose, excellent for low-calorie diets and diabetic diets, reduce blood glucose levels. After harvesting, a progressive decrease in oligofructans occurs while that of simple sugars (glucose, fructose and sucrose) will increase. These two processes, the synthesis and degradation of oligofructans, are under enzymatic control, biochemical changes and storage conditions. During storage, yacon roots are quite susceptible to dehydration, as a consequence the roots lose weight and acquire a rough appearance that makes them less attractive to the consumer, changing their general characteristics. Studies have shown that storage conditions (temperature, relative humidity and light intensity) are environmental factors that alter the composition of yacon sugars (oligofructans and inulin), turning them into simple sugars such as glucose, the same as incompatible with the diabetic patient [1-4].
The storage of fresh yacon is an important aspect for the conservation of fructooligosaccharides; however, there is limited information on the forms or mechanisms of conservation in fresh, since in a few days most of the root degrades and consequently generates high glucose levels. Thus, the yacon reflects little expectation by marketers and final consumers because it does not have standardized parameters for post-harvest handling and storage; that when consumed they have the probability of being rejected or observed, in addition to threatening health, from the point of view of safety, it is more important to indicate that there are no specific storage studies that explain the changes that occur in the process of postharvest of yacon root. Reasons that allowed us to ask the following question:
¿What storage parameters does the fresh yacon require in order to preserve the inulin in amounts similar to that harvested? And whose objective was to determine the storage parameters required by the fresh yacon in order to preserve the inulin in amounts similar to that harvested; This allowed the selection of temperature, relative humidity and intensity of storage light that retains inulin in amounts similar to that harvested.
Seminario and Valderrama, indicate that the properties of yacon have been attributed to its oligofructan content, so it is important to know how the chemical composition of the roots changes in their main stages of development; in studies on the subject indicates that after harvest a progressive decrease in oligofructans will occur while that of simple sugars (glucose, fructose and sucrose) will increase. These two processes, the synthesis and degradation of oligofructans, are under enzymatic control, biochemical changes and storage conditions. During storage, yacon roots are quite susceptible to dehydration when exposed directly to the sun. As a result of dehydration, the roots lose weight and acquire a rough appearance that makes them less attractive to the consumer, changing their general characteristics, if the roots are not going to be commercialized directly after the harvest and are going to be stored for a short period In time, it is preferable to pack with dirty roots (with soil). In this way the soil adhered to the roots can help dehydration be slower and less damage occurs [1,3].
COUNCIL indicates that yacon tubers are fusiform and can vary considerably in size, shape and flavor, the color of their shell varies from dark brown to opaque purple, even to orange. Internally the tuber is presented as a transparent fleshy body. Tubers generally weigh 200 to 500 grams, but can weigh 2 kilograms. Also It indicates that for now in the market there is no official standard for classifying roots according to a caliber or size. In order to estimate the type of roots produced and the relative proportion in the harvest, in Cajamarca the roots have been classified into 03 categories: Roots of first. They are the largest, exceed 20 cm long, is between 07 and 10 cm of greater diameter and a weight not less than 300 g. Roots of second. They are those that are between 12 to 20 cm long and 05 to 06 cm in diameter with a weight that varies from 120 to 300 g. Roots of third. Considered non-commercial, its length is less than 12 cm, its greater diameter is less than 05 cm and its weight is less than 120 g [1,5].
Regarding nutritional characteristics and chemical composition, yacon has been the subject of attention in decades since it provides bioactive compounds of importance for human health. Its composition is made up of main substances of water and carbohydrates, which are mainly stored in the form of oligofructans and other free sugars. The percentage of root water is around 83 to 90% of the fresh weight. Due to the high water content, the energy value of the root is low. This factor also reduces its useful life in environmental conditions over 7 days since the internal tissues of roots are very delicate, characteristic that predispose to suffer cracks or be easily broken during harvest, packaging and transport [6-8].
Yacon has been reported as a good source of phenol oxidase, which catalyzes the oxidation of phenolic compounds to quinones, after polymerization, they have the typical pigments, known enzymatic oxidation of brown or black color in fruits and vegetables. According to Butler and Rivera, it is important to consider the sub-layer of the shell when studying the yacon extraction process because it is this part of the root that tannins and polyphenols are centered. During peeling and the yacon processing membrane when cells break, polyphenols and tannins are available to mix with other components, especially cytoplasmic enzymes, which cause a process known as enzymatic oxidation, and the skin quickly becomes dark when exposed on air. This oxidation is carried out in the presence of free oxygen, quickly darkening the surface of the yacon tuberous roots, damaging their appearance and their products. From a practical point of view, the control of enzymatic browning is generally limited to the inhibition of the enzyme because polyphenol oxidase and peroxidases enzymes are responsible for the browning reaction [9-11].
With respect to the carbohydrates present in the yacon, among sugars found are monosaccharides fructose and glucose, and sucrose and oligofructans fructooligofructans, and traces of starch and inulin. The roots contain between 10 and 14% dry matter, which is composed of approximately 90% carbohydrates. The sugar composition varies significantly due to factors such as agriculture, season, harvest, weather and postharvest temperature. Unlike most tubers and roots that store carbohydrates in the form of starch, yacon roots contain essentially fructooligofructans (OLIGOFRUCTANOS), sugars that cannot be digested directly by the human body due to the lack of necessary enzymes for the metabolism of these elements and are considered bioactive compounds in food. There is a confusion of terms when referring to the predominant type of carbohydrates in yacon roots. In several studies in the literature, it is claimed that yacon roots contain inulin main component. Although many scientific references citing this information, this is not accurate, since, strictly speaking, the yacon contains only fructooligofructans. The difference between OLIGOFRUCTANOS and Inulin is the number of fructose molecules. In inulin, this number varies between 2 and 60, while in OLIGOFRUCTANOS the number ranges between 2 and 10. This means that OLIGOFRUCTANOS can be considered as an inulin subgroup, so some authors prefer to use the term inulin-type fructooligofructans when more accurately refer to the nature of these sugars. Although the proportion of each can vary, sugar can be considered on the basis of the following dry composition of 40 to 70% OLIGOFRUCTANOS, 5 to 15% sucrose, 5 to 15% fructose and less than 5% glucose [1,6,12,13].
Fructans are fructose polymers, structurally and metabolically related to sucrose. They consist of homologous series of oligo and non-reducing polysaccharides, each containing a residue more than fructose than the previous member of the series, so that the simplest fructan is a trisaccharide. Fructans are oligo or polysaccharides, consisting of a molecule of sucrose, to which fructose residues are linked by beta (2-1) and beta (2-6) glucosidic bonds, which can be linear or branched [14].
Goto et al. demonstrated that the oligosaccharides present in the yacon have fructose residues linked by beta glucosidic bonds (2-1) with a terminal sucrose unit and carrying one of the inulin type [15,16].
Yacon tuberous roots have a high content of soluble sugars (820 ± 30 mg/g of dry matter), fructose (589 ± 38 mg/g of dry matter) and low polymerization fructans (GP 3 to 10). Low samples of inulin (13.50 ± 0.40 mg/g of dry matter) were found, with a medium degree of polymerization of 14.5 [17].
Fructans are natural carbohydrates from reserves found in numerous plants, particularly in the Compositae family. Fructans are formed by fructose polymers derived from the sucrose molecule. Fructans have different chain structures and lengths and a wide variety of glycosidic bonds and fructosyl residues; They are soluble in water and are not reducing sugars. There is no single way to classify fructans, which has created some confusion. Fructans are carbohydrate reserves that contain up to 70 fructose units attached or not to a terminal sucrose molecule, they can have a linear or branched structure linked by fructose fructose bonds [18-20].
Goto et al. demonstrated that the oligosaccharides present in the yacon have fructose residues linked by beta glucosidic bonds (2-1) with a terminal sucrose unit and carrying one of the inulin type [21].
Asami et al. observed the fluctuation of the content of fructans in the tuberous roots of yacon during their development and storage. The average degree of polymerization of these fructans increased linearly during or unfolding, after harvest decreased, while the free fructose, sucrose and glucose contents increased [22].
In behavioral studies of reserve carbohydrates in yacon tuberous roots, I observe an expressive decrease in the content of fructans of 101.3 mg/g in the newly exposed root, at 84.31 mg/g on the second day of sun exposure, stabilizing for this period. This is due to enzymatic degradation of these compounds, which is the same when the root is stored at low temperatures [23].
Inulin is an energy reserve carbohydrate present in more than 36,000 plant species, first isolated in 1804, from the Inula helenium species, by a German scientist named Rose. In 1818, Thomson, a British scientist, gave him the current name (Stephen y Phillips, 2016).
Inulin is made up of fructose molecules linked by β-(2 → 1) fructosyl-fructose bonds, the term "fructans" being used to refer to this type of compound.
Fructose chains have the particularity of terminating in a glucose unit linked by an α-(1,2) bond (residue - Dlucopyranosyl), as in sucrose, but also the terminal monomer of the chain may correspond to a residue of β-Dfructopyranosyl. The species with the highest inulin content store it in the underground part of the plant. Other species (for example in the Gramineae family) have high fructan contents in their aerial parts, but with low extraction performance at the industrial level.
There are few appropriate species to obtain erectiles at the industrial level, at the beginning of this decade, inulin was obtained from two species: the pataca (Helianthus tuberosus) and the chicory (Cichorium intybus), the latter being the industrial source more common [24-27].
Inulin is a non-digestible carbohydrate that is present in many vegetables, fruits and cereals. At present, at the industrial level it is extracted from the root of chicory (Cichorium intybus) and is widely used as an ingredient in functional foods.
Inulin and its derivatives (oligofructose, fructooligosaccharides) are generally called fructans, which are basically made up of linear chains of fructose [28].
Inulins are fructan-type carbohydrates, fructose polymers, linked by β-type glycosidic bonds (2-1). Inulins have a degree of polymerization greater than 10 units, they are considered prebiotics, since they are not digestible by the human digestive tract, they have bifidogenic character (they stimulate the growth of bifidobacteria) and also, when consumed frequently, they favor the absorption of minerals such as calcium, they contribute to the health and well-being of the colon through the strengthening of its epithelium and prevent colorectal pathologies such as cancer [29-33].
Inulin-type fructans from natural foods have lower caloric values due to the β (2,1) bonds that link the fructosylfructose molecules. These configurations b of glycosidic bonds are resistant to enzymatic hydrolysis in the human gastrointestinal tract. Thus, fructans are fermented by saccharolytic Bifidobacteria, resulting in final products, such as short chain carboxylic acid (mainly acetate), propionate and butyrate. These inulin-type fructans and oligofructose have been classified as prebiotics and they are promising as functional foods because they are able to stimulate the growth and/or activity of the bacteria of the intestinal flora [20,34].
In dietary terms, fructan has a low energy value (1.5 kcal/g), which becomes relevant for patients with DM1 and those with excessive obesity. This supports the possibility that this functional food can exert lipid-lowering effects, reducing the risk factors of atherosclerotic cardiovascular disease in diabetic patients [29,35].
Methods
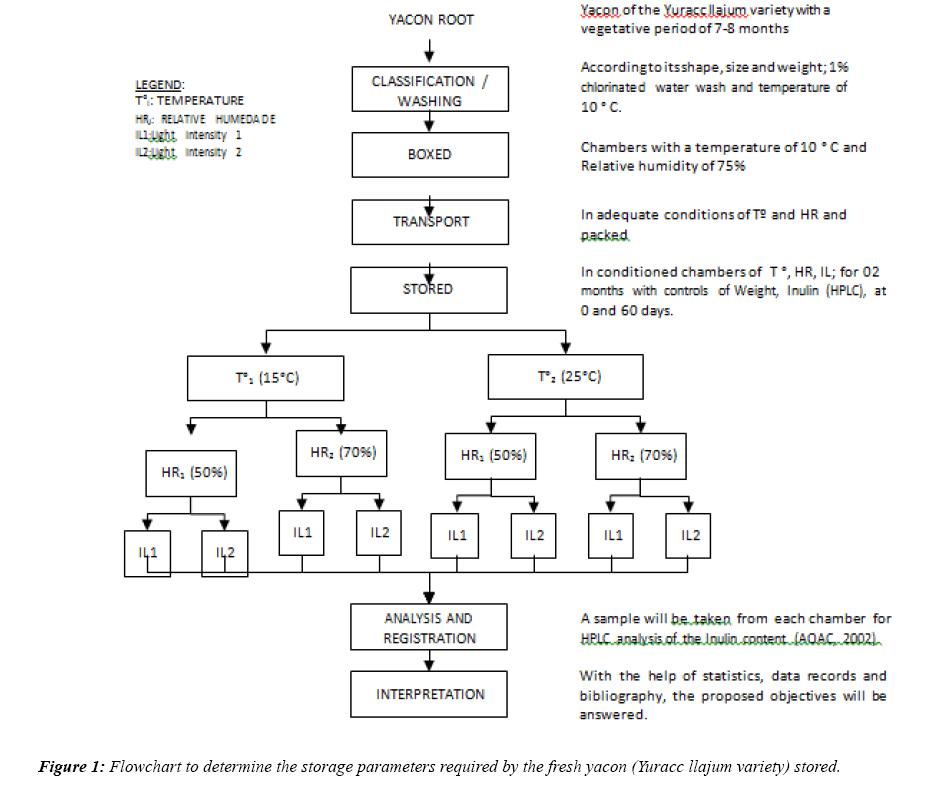
To achieve the stated objectives, the research was developed following the flow chart step by step, which leads to the experimental design, which is detailed in Figure 1. To select the temperature, relative humidity and intensity of storage light that retains inulin in amounts similar to that harvested.
From Figure 1, since the storage operation, 08 controlled storage conditions were created. This allowed the use of CLIMATIZED CAMERAS (with record of temperature, relative humidity and light intensity, for each condition); Stored samples of fresh yacon were extracted every 30 days, according to the analysis to be performed, the weight was recorded and analyzed.
The brix grades, the inulin content (which were sent to the laboratory). The data was processed having the 8 storage conditions and for three repetitions, according to the design matrix; To facilitate the analysis, the calculations were made on the EXCEL electronic sheet and the statistical package statgraphics centurion.
Experimental design and design matrix
It was carried out with a factorial design 23, which allowed determining the storage parameters required by the fresh yacon (Yuracc llajum variety), so that it retains inulin in amounts similar to that harvested, in total there were 08 different conditions that were generated from of factorial design 23, considering 03 factors (temperature, relative humidity and light intensity) with two levels each, all with three repetitions, as detailed in Table 1.
| Temperature | T°1 (25°C) | T°2 (15°C) | ||||||
|---|---|---|---|---|---|---|---|---|
| Relative Humedade | HR1 (50%) | HR2 (70%) | HR1 (50%) | HR2 (70%) | ||||
| Light Intensity (lumen) | 300 | 400 | 300 | 400 | 300 | 400 | 300 | 400 |
| Respuesta Rep. 1 | Y1111 | Y1121 | Y1211 | Y1221 | Y2111 | Y2121 | Y2211 | Y2221 |
| Respuesta Rep. 2 | Y1112 | Y1122 | Y1212 | Y1222 | Y2112 | Y2122 | Y2212 | Y2222 |
| Respuesta Rep. 3 | Y1113 | Y1123 | Y1213 | Y1223 | Y2113 | Y2123 | Y2213 | Y2223 |
Legend: Tºi=Storage Temperature; HRj=Relative Humidity; ILk=Intensity of Light; Yijkl=Dependent variable (Inulin).
Table 1: Structure for experimental design.
Independent variables:
Storage Temperature (Tº):
T°1: 15°C
T°2: 25°C
Relative Storage Humidity (HR):
HR1: 50%
HR2: 70%
Light intensity:
IL1: Light intensity 1 (300 lumen)
IL2: Light intensity 2 (400 lumen)
Dependent variables: Amount of inulin.
Results
For the selection of the temperature, relative humidity and intensity of storage light that manifests a high inulin content in the stored yacon root, we proceeded to evaluate and analyze statistically by means of a factorial arrangement 23, in order to determine what the factors are that influence the variation of the FOS and inulin and to carry out the selection the process of optimization of the factors that allow to obtain a maximum Inulin content or a minimum percentage variation of the inulin content was carried out, with respect to the start as detailed below:
Equation 1: Regression equation that allows to obtain the variation of inulin content
Inulin=228,128 -13,2768*T – 2,90708* HR – 0,756875* 1L + 0,241617*T*
Discussion
Table 2 shows the variation of inulin content, obtained in each sample of fresh stored yacon, determined at the beginning and end of storage, which led to the variation of the content of this component in the yacon root stored under the Eight storage conditions.
| Temperature (ºC) | 25 | 15 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Relative Humedade (%) | 50 | 70 | 50 | 70 | |||||
| Light Intensity (lm) | 300 | 400 | 300 | 400 | 300 | 400 | 300 | 400 | |
| Almac./Día | C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 | |
| INULIN (g/100 g) | 0 | 21,66 | 40,27 | 33,32 | 27,95 | 37,66 | 26,33 | 35,96 | 32,77 |
| 20,97 | 36,79 | 32,71 | 27,76 | 36,48 | 26,56 | 36,31 | 33,10 | ||
| 21,26 | 37,86 | 32,96 | 27,86 | 37,04 | 26,43 | 36,16 | 33,01 | ||
| 60 | 6,26 | 11,17 | 10,18 | 13,29 | 16,98 | 14,14 | 14,31 | 15,16 | |
| 7,85 | 11,16 | 10,95 | 13,34 | 17,22 | 12,34 | 14,34 | 15,19 | ||
| 7,38 | 11,18 | 10,57 | 13,38 | 17,12 | 13,04 | 14,43 | 15,17 | ||
| Variacion INULIN (g/100 g) | 15,40 | 29,11 | 23,14 | 14,66 | 20,68 | 12,19 | 21,65 | 17,61 | |
| 13,12 | 25,64 | 21,76 | 14,42 | 19,26 | 14,22 | 21,97 | 17,91 | ||
| 13,88 | 26,68 | 22,40 | 14,48 | 19,92 | 13,39 | 21,72 | 17,83 | ||
| Porcentaje (%) | 71,09 | 72,27 | 69,45 | 52,44 | 54,91 | 46,30 | 60,21 | 53,74 | |
| 62,58 | 69,67 | 66,51 | 51,94 | 52,80 | 53,55 | 60,51 | 54,12 | ||
| 65,30 | 70,48 | 67,95 | 51,96 | 53,79 | 50,65 | 60,08 | 54,03 | ||
| Porcentaje promedio (%) | 66,32 | 70,81 | 67,97 | 52,12 | 53,83 | 50,17 | 60,27 | 53,96 | |
Table 2: Results of the variation of the inulin content of the fresh yacon root (yuracc llajum variety) stored in the 08 chambers.
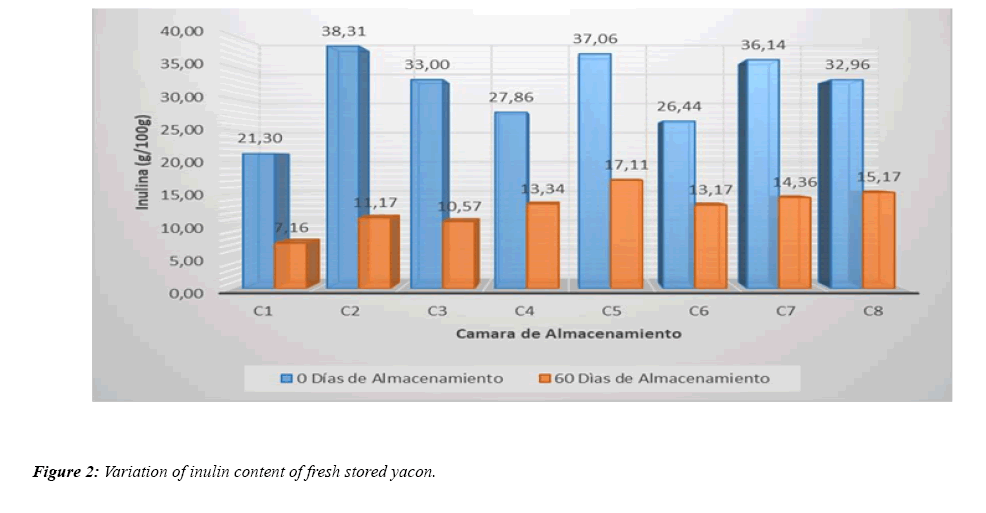
From the results obtained, we observe that during the storage (60 days) of the roots of fresh yacon (variety yuracc llajum), all samples suffer a decrease in Inulin content. Note that, the yacon stored in chamber two (C2: Tº=25; HR=50; IL=400) has the highest percentage of variation of Inulin (70.81%) and the yacon stored in chamber six (C6: Tº=15; HR=50; IL=400) presents less variation (50.17%), all with respect to the initial Inulin content of each yacon (yuracc llajum variety) stored, which is illustrated in Figure 2.
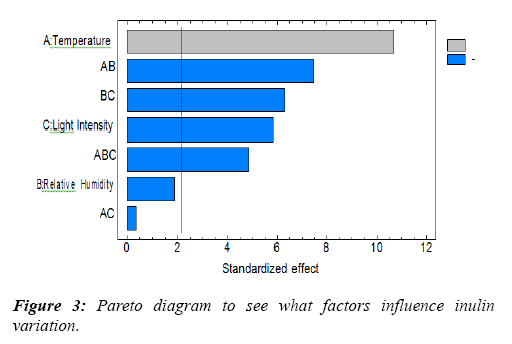
Table 3, indicates the Analysis of Variance (ANOVA) of the influence of the factors on the variation of INULIN, this allowed to observe the factors under study that influence the variation of the inulin content, as well as the statistical significance of each effect comparing its average square against an estimate of the experimental error. In this case, five of the effects have p-values below 0.05, indicating that they are significantly different from zero to 95.0% confidence level, these factors being the ones that significantly influence the variation of the fresh yacon inulin (yuracc llajum variety) stored, at an R-Square statistic of 95,1124%. What is also stated with the Pareto diagram (Figure 3).
| Fuente | Suma de Cuadrados | Gl | Cuadrado Medio | Razón-F | Valor-P |
|---|---|---|---|---|---|
| A:Temperature | 569,888 | 1 | 569,888 | 114,30 | 0,0000 |
| B: Relative Humedade | 17,4251 | 1 | 17,4251 | 3,49 | 0,0826 |
| C: light intensity | 170,827 | 1 | 170,827 | 34,26 | 0,0000 |
| AB | 279,006 | 1 | 279,006 | 55,96 | 0,0000 |
| AC | 0,738504 | 1 | 0,738504 | 0,15 | 0,7061 |
| BC | 197,973 | 1 | 197,973 | 39,70 | 0,0000 |
| ABC | 117,528 | 1 | 117,528 | 23,57 | 0,0003 |
| bloques | 5,03481 | 2 | 2,5174 | 0,50 | 0,6142 |
| Error total | 69,8055 | 14 | 4,9861 | ||
| Total (corr.) | 1428,23 | 23 |
R-square=95,1124 percent, R-square (adjusted by g.l.)=92.9741 percent.
Table 3: Analysis of variance with the most influential effects on the variation of the inulin content of the yacon (variety yuracc llajum) stored.
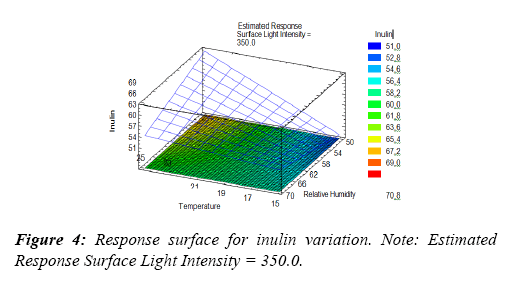
The regression analysis for the inulin content data allowed to obtain the regression equation that has been adjusted to the data, this allowed obtaining a variation of the Inulin content as well as obtaining the response surface (Equation 1). Variable values are specified in their original units. To evaluate this function, the response surface plot was made (Figure 4) which indicates that the blue zone shows the least variation of fructooligosaccharides after 60 days of storage and is the area where the conservation of inulin, that is, in greater quantity being almost equal to the first day of storage.
To better confirm what was shown by the response surface, the Response was optimized and the factors in which the smallest variation of inulin can be obtained or also referred to as the conservation of inulin in greater quantity, obtaining the results shown in Table 4. We can observe that a temperature of 15°C, a relative humidity of 50% and a light intensity of 400 lumen allow us to obtain an optimal variation of 50.1667%, which represents a conservation of 13.17 g/100 g of 26.44 g/100 g of inulin as the largest amount and conserved in chamber six (C6: T=15°C; RH=50%; IL=400 lumen), (Table 2 and Figure 2).
| Factor | low | high | Optimum |
|---|---|---|---|
| Temperature | 15,0 | 25,0 | 15,0 |
| Relative Humedade | 50,0 | 70,0 | 50,0 |
| light intensity | 300,0 | 400,0 | 400,0 |
Goal: minimize INULIN, Optimal value=50.1667%.
Table 4: Optimization of the response for the smallest variation of inulin.
Similar values were found by Asami et al. who indicates that the inulin content gradually decreased to 45% during 02 weeks of storage at 25°C. Likewise Chasquibol et al., Seminario and Valderrama indicates that oligofructan content after one week in storage at room temperature may decrease by 30 to 40%. So we can say that when storing fresh food at a temperature (15°C) considered low, relative humidity considered low (50%) and a light intensity of 400 lumen, the loss of inulin reaches a 50.1667% variation in comparison at the beginning, that is to say that the fresh yacon can be preserved in six (C6: T°=15°C; RH=50%; IL=400 lumen), thus ensuring durability of the product with respect to its inulin content [1,22,36-38].
Conclusion
A storage chamber with a temperature of 15°C, relative humidity of 50% and light intensity of 400 lumen, in a controlled way, allows a variation of 50.16% in the content of Inulin within 60 days of storage of the fresh yacon (yuracc llajun) stored.
References
- Seminario J, Valderrama M. El yacon: fundamentos para el aprovechamiento de un recurso promisorio. Lima - Perú: Centro Internacional de la Papa (CIP), Universidad Nacional de Cajamarca, Agencia Suiza para el Desarrollo y la Cooperación (COSUDE) 2003.
- Mayta P, Payano J, Peláez J, et al. Efecto hipoglicemiante de la raız del Smallanthus sonchifolius en adultos jóvenes clınicamente sanos (estudios preliminares). II. Paper presented at the Simposio Latinoamericano de Raıces y Tuberculos 2001.
- Fukai. Seasonal fluctuations in fructans content and related activities in yacon (Polymnia sonchifolius). Soil Sci Plant Nutr 1997:43, 171-177.
- Lupetti KO. Análise de imagem em química analítica: empregando metodologias simples e didáticas para entender e prevenir o escurecimento de tecidos vegetais. Química Nova, São Carlos. 2005:28:548-554.
- COUNCIL NR. Lost crop of the Incas: Little Known Plants of the Andes with Promise for Worldwide Cultivation 1989.
- Rivera D, Manrique I. Zumo de Yacón. Ficha Técnica. Centro Internacional de la Papa (CIP) 2005.
- Lachman L. Saccharides of yacon [Smallanthus sonchifolius (Poepp. et Endl.) H. Robinson] tubers and rhizomes and factors affecting their content. Plant soil environment, Czech Republic. 2004:50:383-390.
- Manrique I, Párraga A. Conservación y uso de la biodiversidad de raíces y tubérculos Andinos: Una década de investigación para el desarrollo (1993-2003). Jarabe de yacón: principios y procesamiento. Lima: Centro Internacional de La Papa. 2005:40.
- Valentová, y Ulrichová, J. Smallanthus sonchifolius and Lepidium meyenii–prospective Andean crops for the prevention of chronic diseases. Biomedical Papers, Czech Republic 2003:147;119-130.
- Butler G, Rivera D. Innovations in peeling technology for yacon. Project Report International Potato Center. 2004.
- Cabello C. Extração e pré-tratamento químico de frutanos de yacon, Polymnia sonchifolia. Ciência e Tecnologia de Alimentos. 2005:25:202-207.
- Grau A, Rea J. Yacón. Smallanthus sonchifolius. Hermann & Heller (editors) Andean root and tubers: Ahipa, arracacha, maca and yacón.( IPGRI Rome.) 1997:199-240.
- Olvera C, Castillo E, López-Munguía, A. Fructosiltransferasas, fructanas y fructosa. Biotecnología 2007:14, 327-345.
- Ribeiro RCLF. Distribuição, aspectos estruturais e funcionais dos frutanos, com ênfase em plantas herbáceas do cerrado. Revista Brasileira Fisiologia Vegetal 1993;5;203-208.
- Capito S. Raiz tuberosa de yacón (Polyminia sonchifolia): caracterização química e métodos de determinação de frutanos (CG e CLAE-DPA). Universidade Estadual de São Paulo. 2001.
- Goto K, Fukai K, Hikida J, et al. Isolation and structural analysis of oligosaccharides from yacon (Polymnia sonchifolius) Biosci Biotechnol Biochem 1995:59:2346–2347.
- Ohyama T, Yasuyoshi S, Ikarashi T, et al. Composition of storage carbohydrate in tuber roots of yacon (Polymnia sonchifolius). Soil Sci. Plant Nutr. 1990:36:167–171.
- Book G. Compendium of chemical terminology. Int Union of Pure and Appl Chem 2014:528.
- Pedreschi R, Campos D, Noratto G, et al. Andean Yacon Root (Smallanthus sonchifolius Poepp. Endl) Fructooligosaccharides as a Potential Novel Source of Prebiotics. Journal of Agricultural and Food Chemistry 2003:51:5278-5284.
- Roberfroid. Concepts in functional foods: the case of inulin and oligofructose. The Journal of Nutrition 1999;129;1398S-1401S.
- Goto K, Fukai K, Hikida J, et al. Isolation and Structural Analysis of Oligosaccharides from Yacon (Polymnia sonchifolia). Bioscience, Biotechnology, and Biochemistry 1995:59:2346-2347.
- Asami T, Minamisawa, K, Tsuchiya T,et al. Fluctuation of oligofructan contents in tubers of yacon (Polymnia sonchifolia) during growth and storage. Japanese Journal of Soil Science and Plant Nutrition (Japan) 1991.
- Vilhena. Ciclo de cultivo e técnicaspós-colheita de yacon (Polymnia sonchifolia Poep. Endl.) em função do conteúdo de frutose total nos órgãos subterrâneos. São Paulo, Botucatu: Tese (Doutorado em Agronomia) - Universidade Estadual de São Paulo, Botucatu 1997.
- Flickinger EA, Loo JV, Fahey GC. Nutritional Responses to the Presence of Inulin and Oligofructose in the Diets of Domesticated Animals: A Review. Critical Reviews in Food Science and Nutrition. 2003:43:19-60.
- Waterhouse AL, Chatterton NJ. Glossary of fructan terms. Science and technology of fructans 1993;1-7.
- Flamm G, Glinsmann W, Kritchevsky, D. Inulin and Oligofructose as Dietary Fiber: A Review of the Evidence. Critical Reviews in Food Science and Nutrition 2001:41:353-362.
- VanLoo J, Coussement P, De Leenheer L, et al. On the presence of Inulin and Oligofructose as natural ingredients in the western diet. Critical Reviews in Food Science and Nutrition. 1995;35:525-552.
- Madrigal L, Sangronis E. La inulina y derivados como ingredientes claves en alimentos funcionales. Archivos Latinoamericanos de Nutrición 2007;57:4.
- Oliveira M, Nishimoto E. Avaliação do desenvolvimento de plantas de yacon (Polymnia sonchifolia) e caracterização dos carboidratos de reservas em HPLC. Brazilian Journal of Food Technology 2004:7:215-220.
- Garca AC, Mateos BJ, Díaz EL, et al. Alimentos funcionales: Aproximación a una nueva alimentación. Dirección General de Salud Pública y Alimentación, editor. Madrid: Instituto de Nutrición y Transtornos Alimentarios 2008:238.
- PoolZobel B, VanLoo J, Rowland I, et al. Experimental evidences on the potential of prebiotic fructans to reduce the risk of colon cancer. British Journal of Nutrition 2007:87(S2),S273-S281.
- PoolZobel BL, Sauer J. Overview of Experimental Data on Reduction of Colorectal Cancer Risk by Inulin-Type Fructans. The Journal of Nutrition. 2007:137(11):2580S-2584S.
- Rafter J, Bennett M, Caderni G, et al. Dietary symbiotic reduce cancer risk factors in polypectomized and colon cancer patients. The American Journal of Clinical Nutrition. 2007:85;488-496.
- Santana I, Cardoso MH. Raiz tuberosa de yacon (Smallanthus sonchifolius): potencialidade de cultivo, aspectos tecnológicos e nutricionais. . Ciência Rural 2008;38:898-905.
- Stanley J, Goddio F, Jorstad T, Schnepp G. Submergence of ancient Greek cities off Egypt's Nile Delta-A cautionary tale. GSA TODAY 2004;14:4-10.
- Chasquibol N, Aguirre R, Bravo M. Estudio químico y nutricional de las variedades de la raíz de la Polymnia sonchifolia "yacon". Revista Peruana de Química e Ingeniería Química. 2002:5:37-42.
- Alfaro C, Melgarejo VS El yacón: una nueva alternativa en la prevención y el tratamiento de la salud. 2005.
- Stephen AM, Phillips GO. Food polysaccharides and their applications: CRC Press. 2016.