Case Report - Journal of Nutrition and Human Health (2019) Volume 3, Issue 1
Whipple case presenting with cachexia
Yuksel A1*, Bulent V1, Yaprak OU2, Fazilet U3, Yuksel E4, Adibelli Z5 and Bilgin RR2
1Department of Internal Medicine, Izmir Bozyaka Health Research and Application Center, University of Health Sciences, Izmir, Turkey
2Department of Neurology, Izmir Bozyaka Health Research and Application Center, University of Health Sciences, Izmir, Turkey
3Department of Pathology, Izmir Cigli Health Research and Application Center, Izmir, Turkey
4Department of Clinical Infectious Diseases and Microbiology, Izmir Cigli Health Research and Application Center, Izmir, Turkey
5Department of Radiology, Izmir Bozyaka Health Research and Application Center, University of Health Sciences, Izmir, Turkey
- *Corresponding Author:
- Yuksel A
Department of Internal Medicine
Izmir Bozyaka Health Research and Application Center
University of Health Sciences, Izmir, Turkey
Tel: 00902322505050
E-mail: ayuksel68@gmail.com
Accepted on May 21, 2019
Citation: Yuksel A, Bulent V, Yaprak OU, et al. Whipple case presenting with cachexia. J Nutr Hum Health. 2019;3(1):7-11.
DOI: 10.35841/nutrition-human-health.3.1.7-11
Visit for more related articles at Journal of Nutrition and Human HealthAbstract
Whipple’s disease is a bacterial systemic inflammatory disease is caused by Tropheryma whipplei. Classic Whipple’s disease includes arthralgia, diarrhea, abdominal pain, and weight loss. The diagnosis is confirmed by Periodic Acid-Schiff (PAS) positive macrophages presented in small bowel biopsy. The treatment requires long-term antibiotic therapy and the disease could be fatal if not treated. Whipple’s disease should be considered in patients claiming from diarrhea, abdominal pain and malabsorption either most common seen causes are excluded. In this case report, we will present a fifty-four years old patient who presented with weight loss, arthralgia, and chronic diarrhea.
Keywords
Whipple’s disease, Tropheryma whipplei, Malabsorption.
Introduction
Whipple disease is a rare systemic inflammatory condition clinically presented with diarrhea, weight loss, abdominal pain, and arthralgia [1]. The cause of the disease lasted until the 1950s as a mysterious illness. It was believed as a contagious infectious etiology due to its' response to antibiotics. Whipple's disease was first described in 1907 by George H. Whipple in a 36-year-old male patient with abdominal pain, lymphadenopathy, diarrhea, weight loss, and polyarthritis [2]. The disease mostly affects men above middle age (m/f: 8/1) [3]. Tropheryma Whipple (TW) a gram and PAS-positive bacillus exists % 12-26 higher than the general population in stool specimens of sewer workers [4-6].
Despite the rare occurrence of TW infection, the causative bacteria can be found everywhere in the environment. It is described in aseptic individuals who live in Europe, Africa, and the US. PCR samples are obtained from specific tissues and fluids (small bowel, colon, gastric, blood, synovial fluid, cerebral spinal fluid, dura, muscle, lymph node, brain tissue, thrombus, and vitreous fluid) [7]. Classic Whipple disease’s symptoms are; arthralgia, weight loss, abdominal pain, diarrhea, fever, central nervous system disturbance, malabsorption. Migratory arthralgia is usually seen especially in large joints without deformity and destruction. Gastrointestinal symptoms often occur after joint symptoms. Intermittent diarrhea and colic abdominal pain are prominent gastrointestinal symptoms. Weight loss is also seen in later stages depending on diarrhea [8]. Out of these findings, mediastinal lymphadenopathy, dementia, endocarditis, hyperpigmentation of the skin, pleural effusion and chronic cough may also be seen in the course of the disease. (Greub, 2010). A previous study showed excellent sensitivity and specificity of PCR (Feurle, 2004), and the false positive rate of PCR in the gastrointestinal tract has been reported at 6% (95% confidence interval 5–8%) [9].
Neurological manifestations include dementia, abnormalities of eye movements, involuntary movements, seizures, hypothalamic dysfunction, myelopathy, ataxia, and psychiatric manifestations. Uveitis, retinitis, optic neuritis, and papilloedema may be found. 80% of the reported cases of neuro-Whipple had associated systemic symptoms or signs but many patients are presenting without concurrent intestinal manifestation [10].
Laboratory findings include chronic inflammation and malabsorption-ending anemia, elevated leukocyte and platelet counts, elevated C-reactive protein [CRP], hypoalbuminemia, vitamin deficiency and increased prothrombin time. Whipple disease can be diagnosed by small bowel biopsies with the appearance of PAS-positive macrophages. A positive predictive value of 95% and a negative predictive value of 99.5% were reported for both positive stool and saliva PCR [9,11,12].
All patients must be treated with antibiotics which cross the blood-brain barrier. The treatment with a combination of parenteral penicillin and streptomycin for at least 14 days is appropriate, thereafter cotrimoxazole orally 3 times a day for at least one and probably for two years [10]. Dramatic clinical response is obtained in patients within 7 to 21 days after treatment was initiated [7].
Case Report
A fifty four-year-old car mechanist male patient who admitted to the emergency department with complaints of diarrhea, abdominal pain, weakness and unconsciousness for last ten days. The patient had no significant medical history except epilepsy. The patient has been suffering episodes of diarrhea 5-6 times daily in abundant watery but not bloody or mucous form ongoing about 3-4 days in the last 2-3 months. Body mass index [BMI] decreased from 22.8 to 17.9 in six weeks. There was a pain in the bilateral knee joints and there was no joint swelling or redness. There was the loss of consciousness and sleep problems were intensified during admission.
Physical examinatıon
The patient had a low weighted BMI = 17.9 kg/m2 and the examination of the respiratory and cardiovascular system was normal. There was no defensive or rebound on the abdomen, but there was widespread tenderness. There was no organomegaly. There was an increase of 10 seconds in the gut sounds. TA: 90/60 mmHg, pulse: 89/min, fever: 37.3 C, oxygen saturation: 99%, blood gas examination was normal. The sinus bradycardia detected in the ECG.
Laboratory values
The renal function tests and electrolyte values were within normal limits.As theferritin 287 ng/ml (N: 12-300), iron: 16 μg/dl (N: 70-180), total iron binding capacity 130 μg/dl (N: 259-dl), Hemoglobin [Hb]: 6.9 mm3 (N: 13.6-17.2), findings were consistent with chronic disease anemia. In the perypheral smear WBC: 9600 (68% PNL, 21% Lymphocyte, 6% Monocyte 3% Eosinophil 2% Bazophil) was seen. The patient has hypoalbuminemia (Albumin: 2 g/dL [3.5-5.2] and low 25-OH D vitamine: 3.56 ng/ml [N: 30-150], Folate: 2.55 ng/ml [3.1- 19.9] and vitamin B12: 124 ng/ml [N:200-900] test results has been detected. C-reactive protein elevation was detected when infectious parameters were reviewed [CRP 14.9 mg/L (0-5]).
The stool microscopy revealed 1-2 leukocytes. Direct microscopy of stool and parasite examination were normal. Full urine test included 4-5 leukocytes. There was no reproduction in the stool culture. ARB and TBC culture in 3 phase spontaneous were negative. HIV tests and hepatic viral markers A, B, C were negative.
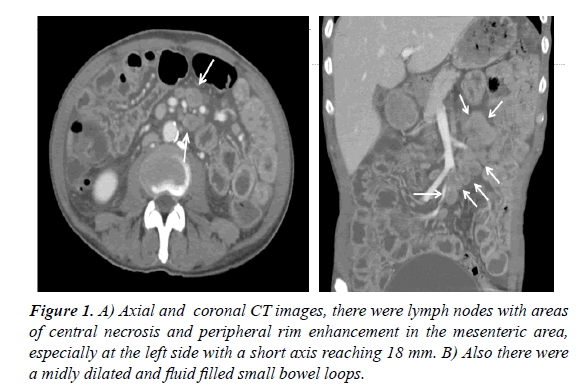
Brucella and typhoid tests were negative. ECG showed sinus bradycardia with flattened QRS and T waves. The cardiothoracic ratio (CTR) and Transthoracic EKO was normal. Abdomen ultrasonography (USG) had shown perihepatic, perisplenic, pericellular and perirenal 4-5 cm free fluid. At the computed tomography (CT) scan, there were hypodense lymph nodes with areas of central necrosis and demonstrated peripheral rim enhancement in the mesenteric area, especially at the left side with a short axis reaching 18 mm (Figure 1A and 1B). Mesenteric fat tissue density had increased and there was fluid in the localization of since it was thought that tuberculous peritonitis might be present with these findings, acidic fluid culture for TBC taken with USG was negative. Adenosine deaminase (ADA) was found to be negative in the acidic fluid sample by paracentesis. Tuberculous peritonitis was excluded from the patient.
Chest CT scan for malignancy showed a 13 mm sized cavitary lesion (sequel bronchotomy) in the right upper lobe and 2 cm bilateral basal pleural effusion. Acid Resistant Bacillus (ARB) in sputum and tuberculosis culture respectively obtained 3 times were negative. The patient was evaluated by Chest and Respiratory Disease specialist and tuberculosis was not considered primarily. The lesion seen on CT was consulted as the area of bronchiectasis. Celiac disease was investigated at the beginning of the non-infectious causes that may lead to malabsorption for the patient. Antitransglyaminase and antigliadin antibodies were negative then the celiac disease was excluded. Colonoscopy of the patient was normal. Acute antral gastritis, duodenitis was detected in the upper gastrointestinal endoscopy and biopsy was taken.
Ciprofloxacin and metronidazole treatment was applied for the urinary tract infection. On the third day of hospitalization, the patient had generalized tonıc clonıc seızure for three times and diagnosed with a status epilepticus. When the first two episodes were responded to the diazepam, the last episode was quite resistant. However, valproic acid 400 mg IV and levetiracetam 1000 mg IV could be administered to control episodes. Levetiracetam 1000 mg and Valproic acid 1000 mg/g were applied in the treatment of maintenance. EEG examination was performed on the patient and reported as a widespread organizational disorder. Meanwhile, the patient's blood valproic acid level was normal. The patient's blood ammonia test was normal and the patient had no history for the liver disease. The findings of the patient's neurology consultation and cranial MRI findings were normal as well.
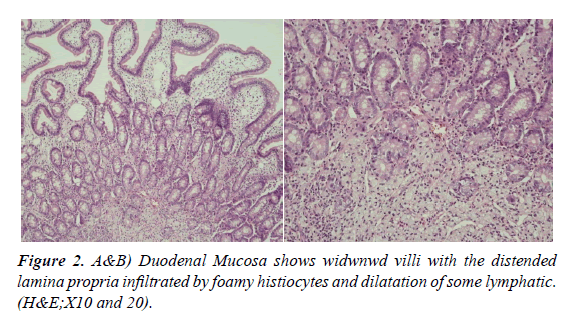
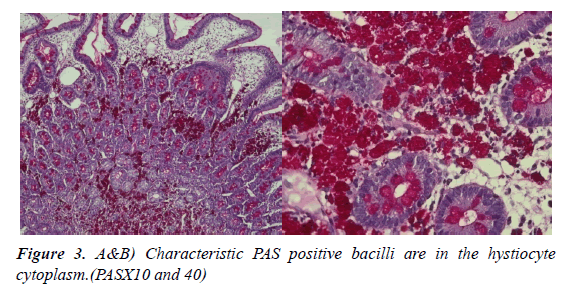
The patient's anemia was linked to the lack of nutrition and normochromic normocytic anemia findings were found. No other etiological investigation was needed with anemia. Acute phase reactors (C-RP), sedimentation, and ferritin] were high and albumin was low. In the blood and stool cultures, which were repeated 3 times, the factor could not be isolated. The duodenal biopsy report obtained by the endoscopic intervention of the patient, histological examination of the duodenal mucosa showed widened villi with the lamina propria infiltrated by immunohistochemical CD68 positive foamy histiocytes, as well as neutrophil-rich inflammatory infiltrate and moderate dilation of some lymphatic vessels (Figure 2A and 2B). On Periodic Acid-Schiff (PAS) staining, the macrophages were filled with diastase-resistant and non-acid-fast stained PAS Positive granular material, due to the presence of bacilliform bodies (Figure 3A and 3B). Histopathological findings may be significant in terms of Whipple's disease.
In order to exclude benign and malignant pathologies of the possible gastrointestinal system, admission to internal medicine intensive care unit (ICU) was carried out because of hypotension, resistant hypokalemia, anemia and recurrent episodes of diarrhea. In ICU, patient’s symptoms were getting better and the vital values were stabilized. The patient’s treatment had been continued on the internal medicine ward. There was no enteropathogenic bacterium in the stool culture sent for the investigation of diarrhea etiology in the examinations sent to the internal medicine service. No parasites were detected in stool. Human immunodeficiency virus (HIV) and other viral markers were negative. Anti-gliadin and anti-endomysium antibodies had sent for celiac disease and they were negative. Malabsorption angle endoscopy was performed. In the antrum, the mucosa was erodible and widely erosive. Biopsies were taken from Antrum. Duodenum was normal, and biopsies were taken from malabsorption angle. The patient had sinusoidal bradycardia onward visits and was consulted for cardiology and theophylline was recommended for treatment. Carbamazepine treatment has discontinued the approval of neurology department because of its' bradycardic side effect.
Although the patient has been using antiepileptic treatment for 13 years, there were many epileptic seizure episodes onward during admission. The patient's antiepileptic treatment was arranged by neurologist due to resistant epilepsy as levetiracetam 2000 mg/ day and valproic acid 1000 mg/day.The patient was evaluated with the lumbar puncture of CSF. No leukocytes were seen, the protein was normal, pandy was negative. The CSF sample for the trophyma whipplei polymerase chain reaction (PCR) was negative. The external center pathology report will be written and the patient was diagnosed with Whipple disease. After the diagnosis of Whipple disease, transesophageal echocardiography was performed to determine the endocarditis of the patient with bradycardia, although it was long and asymptomatic. Since the patient reassessed by cardiology was asymptomatic, follow-up was recommended in terms of bradycardia. Carbamazepine treatment was discontinued due to the risk of bradycardia side effects.
Ceftriaxone was given for 2 weeks for urinary tract infection according to the consultation with infectious diseases, it was suggested to continue treatment with trimethoprim/ sulfamethoxazole (TMP-SMX) for a year. Shortly after the treatment started, the general condition of the patient had improved. Ceftriaxone was given for 2 weeks for urinary tract infection according to the consultation with infectious diseases, it was suggested to continue treatment with trimethoprim/ sulfamethoxazole (TMP-SMX) for a year. Shortly after the treatment started, the general condition of the patient had improved.
Discussion
Our patient was also 54 years old male patient complaining of long-standing arthralgia. He was a car mechanist and was not assessed as a job-related contamination etiology. Classic Whipple disease is a multisystemic disease characterized by arthralgia, weight loss, abdominal pain, and diarrhea. Our patient applied with intermittent diarrhea, colic abdominal pain and weight loss to us. Migratory arthralgia involving large joints occurs in approximately 80% of patients [13,14].
Especially peripheral joints (knees, elbows, ankles) are mostly involved. In our case, there was a complaint of prolonged arthralgia involving bilateral knees. Our patient said that he had applied to different centers before and used various analgesics.
Gastrointestinal symptoms usually occur after the onset of joint complaints. The first symptom usually manifests like diarrhea and colic abdominal pain. Watery diarrhea, steatorrhea, sometimes even severe gastrointestinal bleeding may occur. Acidemia and abdominal distension may occur due to chronic malabsorption in the late period Gunther et al., In our case, gastrointestinal symptoms appeared as watery diarrhea and colic abdominal pain after complaints of arthralgia. The patient had a perihepatic, perisplenic, pericellular and perirenal plastering style in the USG, free-flowing fluid reaching 4.5 cm deep in the deepest line of the intestine, but there was no abdominal distension on physical examination. The patient had a cachectic appearance. Mesenteric and mediastinal lymphadenopathy were reported in about half of cases with Whipple's disease. These patients were searched for lymphoma before the diagnosis. In our patient's bovine cysteine, the mesenteric area, especially the left hemisphere, showed a hypodense (necrotic?) lymphadenopathies in the appearance of a cone, reaching a short axis of 18 mm. Our patient was researched for tuberculosis and the result was negative. Neurological findings of Whipple disease has been reported in about 10-40% of people who are diagnosed with the disease [10].
Dementia and other central nervous system findings can be detected later in the disease. In our patient, only epilepsy was diagnosed neurologically and only could be controlled with tripple antiepileptic therapy. Whipple disease was later investigated for neurological involvement. CSF was performed, CSF PCR was sent for the Trophyeryma Whipplei and the result was negative. Whipple endocarditis was found in a small proportion of patients who are diagnosed with Whipple disease [15]. The patients with Whipple endocarditis do not have classical Whipple disease and do not apply with diarrhea arthralgia [11,16].
In our patient, only sinus bradycardia was detected as cardiac pathology and theophylline were started by cardiology. The carbamazepine used by the patient for epilepsy was also discontinued due to possible bradycardia. Patients who were bradycardia were asymptomatic and follow-up was recommended by cardiology.
Skin hyperpigmentation can be seen in about 40-45% of cases [17]. In our case, there was widespread hyperpigmentation in the skin. Pulmonary involvement may include pleural effusion, chronic cough, interstitial lung disease, and pulmonary hypertension [18]. The chest CT of our patient had pleural effusion up to 2 cm in the bilateral bases. Laboratory findings include chronic inflammation and malabsorption-ending anemia, elevated leukocyte and platelet counts, CRP, hypoalbuminemia, vitamin deficiency and increased prothrombin time. We found anemia, CRP increase, hypoalbuminemia, vitamin deficiency and increased prothrombin time in our case. However, there was no increase in leukocyte and platelet counts. Diagnostic testing of Whipple's disease involves taking small bowel biopsy with upper gastrointestinal endoscopy and showing PAS-positive macrophages in the pathology [4]. In our patient, the upper gastrointestinal system endoscopy was performed and duodenal biopsies were taken to investigate malabsorption angle. While PAS-positive macrophages were seen in the pathology of the biopsies, the patient was diagnosed with Whipple disease.
Adults receiving a classical Whipple disease without neurological involvement were firstly treated with ceftriaxone (2 g IV once daily) or a penicillin (2 MU IV every 4 h) for the first 2 weeks; followed by TMP/SMX (160 mg TMP/800 mg SMX 2 times a day) for 1 year 15. Dramatic clinical response is obtained in patients within 7 to 21 days after treatment has begun [19]. Since our patient was given ceftriaxone 1 x 2 gr IV for 2 weeks due to urinary tract infection during the course of Whipple's disease diagnosis, it was decided to continue treatment with 2x (160 mg TMP/800 mg SMX) for 1 year. It was observed that the clinical condition improved after about 10-15 days after the diagnosis and the treatment was started. Routine blood tests, which all the clinical findings mentioned in the third month. Control examination at the hospital was found to be normalized.
Conclusion
Whipple's disease should be considered in patients with long-term diarrhea, weight loss, and malabsorption findings. While rare etiologic findings are encountered with age-related malabsorption findings, WH should be investigated. It is recommended that endoscopic examination should be taken from the duodenum and examined with PAS stain. Early recognition and dramatic response to treatment of SM/TX reveal once again the importance of this disease in pregnant patients. We think that we will contribute to awareness about the disease in the literature.
References
- Whipple GH. A hitherto undescribed disease characterized anatomically by deposits of fat and fatty acids in the intestinal and mesenteric lymphatic tissues. Bull Johns Hopkins Hosp. 1907;18: 382–391.
- Lepidi H, Fenollar F, Gerolami R, et al. Whipple’s disease: Immunospecific and quantitative immunohistochemical study of intestinal biopsy specimens. Hum Pathol. 2003;34(6): 589-96.
- Marth T. Tropheryma whipplei, immunosuppression and whipple’s disease: From a low-pathogenic, environmental infectious organism to a rare, multifaceted inflammatory complex. Dig Dis. 2015;33(2):190-9.
- Braverman DZ, Dollberg L, Shiner M. Clinical, Histological, and Electron Microscopic Study of Mast Cell Disease of the Small Bowel. Am J Gastroenterol.1985;80(1): 30-7.
- Dutly F, Dutly F, Altwegg M, et al. Whipple’s Disease and “Tropheryma. Society. 2001;14(3): 561–83.
- Weinberger SE, Weiss JW. Weaning from Ventilatory Support. N Engl J Med. 1995;332(6): 388-9.
- Fenollar F, Puechal X, Raoult D. Whipple’s disease. 2007;356(1): 55-66.
- Moos V, Feurle GE, Schinnerling K et al. Immunopathology of Immune Reconstitution Inflammatory Syndrome in Whipple’s Disease. J Immunol. 2013;190(5):2354-61.
- Müller SA, Vogt P, Altwegg M, et al. Deadly carousel or difficult interpretation of new diagnostic tools for Whipple’s disease: Case report and review of the literature. Infection. 2005;33(1):39-42.
- Vital Durand D, Gérard A, Rousset H. Neurological manifestations of Whipple disease. Rev Neurol. 2002;158(10 Pt 1):988-92.
- Fenollar F, Lagier JC, Raoult D. Tropheryma whipplei and Whipple’s disease. J Infection. 2014;69(2):103-12.
- Fenollar F, Raoult D, Rolain J, et al. Value of Tropheryma whipplei Quantitative Polymerase Chain Reaction Assay for the Diagnosis of Whipple Disease: Usefulness of Saliva and Stool Specimens for First‐Line Screening. Clin Infect Dis. 2008;47(5): 659-67.
- Günther U, Moos V, Offenmuller G, et al. Gastrointestinal diagnosis of classical whipple disease: Clinical, endoscopic, and histopathologic features in 191 patients. Med (United States). 2015;94(15): e714.
- Lagier JC, Lepidi H, Raoult D, et al. Systemic tropheryma whipplei: Clinical presentation of 142 patients with infections diagnosed or confirmed in a reference center. Medicine (Baltimore). 2010;89(5):337-45.
- Raoult D, Birg ML, La Scola B, et al. Cultivation of the bacillus of Whipple’s disease. N Engl J Med. 2000;342(9): 620-5.
- Gubler JGH, Kuster M, Dutly F, et al. Whipple endocarditis without overt gastrointestinal disease: Report of four cases. Ann Intern Med. 1999;131(2): 112-6.
- Greub G. A new piece added to the whipple puzzle: Tropheryma Whipplei primary infection with bacteremia and cough. Clin Infect Dis. 2010;51(5): 522-4.
- Edouard S, Stein A, Lions C, et al. Tropheryma whipplei DNA in bronchoalveolar lavage samples: a case control study. Clin Microbiol Infect. 2016;22(10): 875-879.
- Schneider T, Moos V, Loddenkemper C, et al. Whipple’s disease: new aspects of pathogenesis and treatment. Lancet Infect Dis. 2008r;8(3): 179-90.