Research Article - Journal of Industrial and Environmental Chemistry (2022) Volume 6, Issue 6
West assiut power plant revers osmoses rejected water environmental impact reduction
Adel M. Kamal El-Dean*1, Abdel-Aal M. Gaber1, H. Manaa Hassanien2, Ramy M. Dissoky2
1Department of Chemistry, Assiut University, Assiut, Egypt.
2Department of Chemistry, West Assiut Combined Cycle Power Plant (WACCPP), Assiut, Egypt.
- Corresponding Author:
- Adel MKD
Chemistry Department
Faculty of Science, Assiut University
Assiut 71516, Egypt
E mail: gaber@aun.edu.eg
Received: 10-Nov-2022, Manuscript No. AAIEC-22-34831; Editor assigned: 11-Nov-2022, PreQC No. AAIEC-22-34831(PQ); Reviewed: 21-Nov-2022, QC No AAIEC-22-34831; Published: 23-Nov-2022, DOI:10.35841/aaiec-6.6.129
Citation: Adel MKD et al. West assiut power plant revers osmoses’ rejected water environmental impact reduction. J Ind Environ Chem. 2022;6(6):129
Abstract
As there are a highly increase in the interest in environmental issues in recent years, and in accordioning to this desire for reduction of the pollution load to the environmental, the problem of the ROC amount that be released to the neighbor environment of the West Assiut Combined Cycle Power Plant (WACCPP), needs to be solved. The main goal of this article is the trials that will reduce volume of ROC effluent that seeps to the environment, and also those will improve its quality will be studied.
Keywords
Reverse Osmoses, Concentrate, Total dissolved Salts, Electrocoagulation, Deep well, Evaporation Pond.
Introduction
Water is being used for electric power generation and different purposes nowadays from traditional sources as Rivers and, non-traditional water sources such as seawater, excessively hard or brackish groundwater, poorer quality surface waters, and wastewater. All of these sources commonly require treatment with high quality technologies before use. In West Assiut Combined Cycle Power Plant (WACCPP) groundwater is used as the main source for demineralized water production across Revers Osmoses treatment units, for this purpose, 37 wells were drilled there. In this paper we will examine the RO rejected water (wastewater of RO). The chemical composition of Reverse Osmosis Concentrate (ROC) or reject is a reflective for the raw water source chemistry, RO pretreatment, and the mode of RO system operation. For inland brackish water RO plants, the concentration and type of different ions vary depending on the process chemicals and the constituents of the different geological regions and layers containing water and owing to differences in the solubility of minerals. ROC represents 10-30% of the feed water flow for brackish and underground waters, salts in ROC could be concentrated for 4 to7 times [1,2]. The viable options for ROC management often dictate whether or not a project progresses past the planning stages, therefore, it may be the most critical consideration in many situations.
Reverse Osmoses Reject Characterization
ROC disposal environmental adverse effects and its associated costs are reduced by reducing the volume and/ or by diminishing the pollutant load of the concentrates. Converting it from a waste to a resource through treatment and beneficial use minimize both environmental impacts and costs. The physical parameters and chemical composition of the concentrate is presumably related to feed water characteristics and variable plant operational parameters such as membrane selection and flux. Depending on regional geochemistry, groundwater RO plants concentrate dissolved minerals such as calcium, magnesium, sodium, potassium, chloride, sulfate, silica, fluoride, nitrate and iron. The data had shown a concentration factor (CF) of about 4 for most constituents, but some parameters do not follow this pattern as: total iron (CF=2.3); calcium (CF=6.6); magnesium (CF=0.1). The average chemical composition of brines produced from inland brackish water RO plants on Australia and the Arabian Peninsula (Oman, United Arab Emirates, and Saudi Arabia) have been compiled as shown in (Table 1). The data indicate that plants in this region tend to produce more concentrated brines with respect to conductivity and total dissolved solids [3].
| Item | pH | EC X100 µS/cm |
T H ppm CaCO3 | T Alk. ppm CaCO3 | SiO2 ppm |
Fe ppm | Mn ppm | Ca ppm | Mg ppm | Na ppm | K ppm |
SO4 ppm |
Cl ppm | NO3 ppm |
PO4 ppm |
Tem H. ppm CaCO3 | HCO3 alk. ppm CaCO3 | TDS X 100 ppm |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Australia | 8 | 104 | 1650 | 1480 | 133 | 0.09 | 0.01 | 650 | 5.7 | 2180 | 10.4 | 453 | 3080 | 29.1 | 1.04 | 1480 | 1480 | 74.4 |
| Arab. Peni. | 8 | 1274 | 4041 | 945 | — | 65.5 | 22.6 | 923 | 510 | 6206 | 264 | 4197 | 8946 | 143 | — | — | — | 803 |
Table 1. ROC variety parameters’ concentrations.
The water recovery of seawater reverse osmosis (SWRO) systems varies between 40% and 50%, this means that 60–50% of the feed stream is wasted as concentrate. While brackish water reverses osmosis (BWRO) recovery is from 75% to 85%, but reduced to only 50–60% due to scaling or energy saving considerations [2], [23] and [20] and [8]. The amount of contaminants could increase by 4-10 times and potentially reaching toxic level in the ROC concentrate, these contaminants areas; nitrate, naturally occurring radioactive materials, arsenic, and other heavy metals introduced by natural dissolution of rocks, and agricultural runoff, so it requires treatment before beneficial use or discharge [4,5,6].
ROC treatment
The RO concentrate treatment is considered the key barriers that need to be overcome for a broader use of desalination processes in inland applications and includes; treatment, disposal and beneficial uses of ROC. RO feed water pretreatment (physio-chemically pretreatment), quality improvement, feed water pH adjusting, membrane scale control and, using RO new technologies, are used for improve water recovery consequently, RO permeate volume increase. Also, reducing the ROC volume to be disposed is by; revers osmoses units permeate production improvement, concentrate treatment for improvement water recovery when pass in another RO unit, and reduction of contaminants before beneficial uses or discharge. Technologies have been proposed for ROC treatment, that could be viable options to be used to diversify available freshwater resources, provide newly recognized value and thus facilitate the more expensive means for treatment of ROC for beneficial use. ROC volume reduction is by; intermediate physio-chemically treatment, using a second reverse osmosis stage, new technologies (non-traditional reverse osmosis (VSEP & EMS), electrodialysis (ED), electrodialysis reversal (EDR) Zero Liquid Discharge Technologies (ZLDT)) [7,8]. Disposal methods depend on concentrate quantity and quality, permitting requirements, geography and geology (e.g., accessibility to different receivers, appropriate geological formation for deep well injection, land availability), costs, and potential environmental impacts. Traditional concentrate disposal methods as evaporation, discharge to surface water, discharge to municipal treatment plants, and deep well injection, also, controlled by environmental impacts, lack of dilution of the receiving water bodies, and by the required physical footprint. As there is no one treatment method fits all scenarios, however, the more that the reject volume can be reduced, the better the choices for final disposal. ROC of brackish water desalination may be disposed to some of the common methods adopted for disposal as; evaporation ponds, deep wells, disposal to sanitary sewers, storm drains, irrigating salt tolerant species, mechanical evaporation, carting away from site by vendors. Direct discharge in place of the ROC source is by blending with power plant cooling water, and land applications (evaporation ponds, deep well injection, infiltration, constructed wetlands and injection to petroleum old wells (used as disposal wells) [9,10]. Beneficial use of the ROC represents a more concentrate disposal and treatment as a sustainable alternative to the traditional disposal and treatment options, and the concentrate becomes a resource rather than a pollutant, as long as there is a value for this recovered resource. The viability of beneficial and nontraditional uses of concentrate are depended on a number of critically important site-specific factors, including costs, climate, markets, regulatory issues, and ecological risks. There are a lot of technologies for beneficial reuse of ROC, converting it from a waste to a resource minimize both costs and environmental impacts. Also, ROC byproducts are studied, recovering commercial byproducts from ROC would be the optimum treatment option, as it solves the environmental problem of concentrate disposal, as well as the economic profitability of reverse osmosis is improved at the same time. Generally, all these methods need to consider the environmental impacts, costs, complexity of permitting and regulations, site requirements and footprint, energy use, reliability, ease of implementation, and operation of the processes involved. In recent years there a lot of researches about finding substance that could absorb salts and minimize the TDS, they use aluminum electrodes in a process called electrocoagulation (EC), they found that the TDS removal is about 90%, and factors affect the removal of salts process are current density (I), reaction time (RT), pH, temperature of the solution (T), distance between the electrodes (IED) and stirrer speed (rpm)[11,12,13].
Environmental impact
The environmental impacts associated with concentrate disposal are related to its elevated salinity, ion imbalancerelated toxicity as well as toxicity due to elevated concentrations of anthropogenic contaminants. The disposal of untreated concentrate from high-pressure membrane plants presents a number of environmental issues; It can potentially cause severe damages to marine, freshwater and terrestrial environments, each receiving environment is unique. The untreated ROC has impacts other than direct changes in salinity, it was reported that the high salinity associated with reject brine discharges has detrimental effects on the aquatic life, sea grass structure and vitality as well as the quality of the seawater available for desalination in the area [14]. It adversely affects the water and sediment quality of receiving water bodies, reduce dissolved oxygen levels impairs marine life as well as functioning and intactness of coastal ecosystems, also the indigenous aquatic species in the area of discharge vary in their susceptibility to deleterious effects [15], Many aquatic organisms are highly sensitive to variations in salinity, as cell dehydration occurs with increased salinity, and as salinity rises the number and diversity of species falls. High salinity also causes ion imbalance – triggered toxicity to aquatic flora and fauna. Disposal into open land, unlined ponds or pits have significant environmental impacts, potential for polluting the underground water (increase hardness) resources, risk of soil salinization and profound effects on subsurface soil physical properties [16,17]. Water logging, formation of crusts and reduce soil permeability causing great reduction in infiltration rate thereby preventing plants or crops from accessing enough water for good growth. While specific constituents, such as heavy metals, can present significant environmental concerns, which was confirmed hydrochemically and chemically for the raw water used in WACCPP [18,19].
Specification of West Assiut Combined Cycle Power Plant (WACCPP)
West Assiut power plant is divided into two stages the simple cycle and the combined cycle, (Table 2) shows the main specifications. The simple cycle consists of 8 similar gas turbine units each with a combustion chamber contains14 cans, with a maximum power of 125 MW, with a total maximum power 1000 MW, each unit is equipped with wet-de NOX system. The combined cycle consists of 2 similar units each unit is composed of 4 heat recovery steam generators (HRSG) (each is on one gas turbine, and with a 210 m3/hr steam production capacity and the steam have a 540ºC) with a maximum power of 2X250 MW =500MW. And with a total power for both stages 1500MW. Burn of natural gas as the main fuel and light distillate oil as the alternative fuel.
| Description of Simple Cycle Stage’s Constituents | |
|---|---|
| Item | Description |
| Simple cycle | Two Modules of gas turbines, contains 8 gas turbine units, power capacity. |
| Power capacity | 125 MW/hr for each gas turbine |
| Simple cycle Modules | Each 4 gas turbine units composes one module |
| Module 1 | Contains U1A, U1B, U1C, U1D. |
| Module 2 | Contains U2A, U2B, U2C, U2D. |
| Exhaust gases temp. | 590ºC |
| Water uses | 176 ton/hr injection waters for d-NOx, Makeup water for closed cooling system, Washing water for the turbine and compressor offline washing |
| Description the constituents of combined cycle stage’s | |
| Combined cycle | Two Modules of HRSGs, one on each module of simple cycle |
| Power capacity | 250 MW/hr for each module |
| Combined cycle 1 | Composed of 4 HRSGs one on each gas turbine in the simple cycle module 1 |
| Combined cycle 2 | Composed of 4 HRSGs one on each gas turbine in the simple cycle module 2 |
| Exhaust gases temp. | 150 ºC |
| Water source | 37 wells were dogged around the power plant site, gathered in 20 stations, 17 doubled wells and 3 are singled. Each well’s deep reach to 200m and the beginning of water surface is at 80m from land surface. |
| Water uses | Makeup water for closed thermal cycle, Makeup water for closed cooling system, Washing water for the HRSGS offline washing |
| Steam ton/hr | 840 ton / hr for each combined |
| Steam pressure bar | 116 |
Table 2. Description of WACCPP’s Constituents.
Water treatment in west assiut combined cycle power plant
Water treatment depends on the purpose for which water is used and its source. In UEEPC, power plants use the demineralized water for different purposes such as: Steam generation, cooling, sealing, de- NOx, washing and so on. In old plants the traditional treatment means used is; clarifiers, gravity filters, activated carbon filters for pre-treatments and ion exchangers for desalination. In the modern power plants; self-cleaning filters and ultra-filtrations systems are used for pre-treatments and reverse osmosis and electrodialysis for demineralization. Water treatment reverse osmosis units that fed by a ground water as a raw water from, result in a huge amount of the reject water that is normally viewed as a severe environmental threat.
West assiut power plant location
Assiut is the largest town in upper Egypt and lies about 234 miles south of Cairo. City of Assiut is located at 27°11՝00̏N, 31°10՝00̏E and spread across 26,000km2. West Assiut power plant as shown in image (1c), is located at the northern west of Assiut city in Assiut governorate in Upper Egypt, and allocated from Assiut city by 25km and at about 5km from Bany Ghalleb and about 3.43km from the nearest village Jhdum as in Image (1). The site located between the petroleum company in the north and cement company in the south as shown in image (1b) at the coordinates shown in (Table 3), it is on a 33.6 acres area, the whole WACCPP view as in Image (2), also wells are used as a raw water source.
| point | A | B | C | D |
|---|---|---|---|---|
| Easting | 30.59-53.21“ E | 30.59-47.47“ E | 31.0-5.20“ E | 31.0-9.78“ E |
| Northing | 27.11-3.83“ N | 27.10-48.71“ N | 27.10-44.17“ N | 27.10-59.50“ N |
Table 3: Description of WACCPP’s location.
Experimental
Sampling
Water samples from raw water tank in WACCPP and ROC water sample were withdrawn and analyzed using standard procedures for determining its physical and chemical constituents. The sample’s bottles were prepared according the ASTM standards and the water samples were taken under almost careful conditions.
Instrumentation
The samples were analyzed for determining its content of; PH, hardness, alkalinity, TDS. The measurements were conducted according the standard specification, where; potential of Hydrogen (pH) and Electrical Conductivity (EC), were measured by HANA (HI9811-5) instrument directly in the field. While the total hardness was analyzed by the volumetric method [20].
Laboratory work
In this work the ROC solution was subjected to an electrocoagulation by applying a potential about 12 volt and 3.3 A, in a container with 600 ml volume, the applied potential is on 2electrodes (Al & stainless steel) for a 4h the pH, TDS, and T were tapped, then the reduction in the TDS was calculated at 25ºC.
Water Samples Analysis’ Results
Physical specifications results
Raw and RO reject water TDS change with hypochlorite and SMBS dosing (with controlling residual chlorine at RO inlet does not exceed 0.0 ppm) as shown in (Table 4).
| Item/ SMBS p/p stroke % |
Dosing rate of SMBS l/h | NaOCl dosing p/p stroke | Raw water’s TDS (ppm) | Reject’s TDS (ppm) |
|---|---|---|---|---|
| 0.0 | 0.0 | 0.0 | 2713 | 6512 |
| 30 | 1.8 | 100 | 2797 | 6714 |
| 100 | 6 | 100 | 2833 | 6801 |
Table 4: Effect of RCl2 and SMBS on the ROC’s TDS.
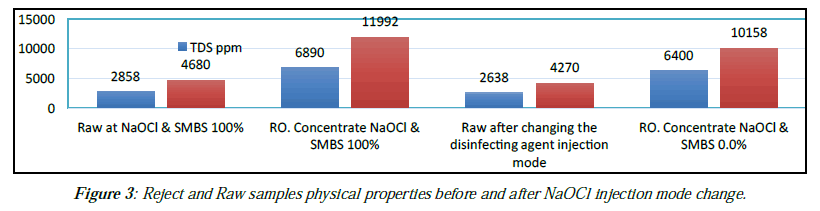
Physical and chemical analysis results of raw and rejected water samples are as in (Tables 5 and 6). Reject and Raw samples physical properties before and after NaOCl injection mode change (Figure 1), (Table 5).
| Item | pH | TDS ppm | EC µs/ cm | T. Alk ppm | TH. ppm |
|---|---|---|---|---|---|
| Raw at NaOCl & SMBS 100% | 7.25 | 2858 | 4680 | 250 | 705 |
| RO. Concentrate NaOCl & SMBS 100% | 7.75 | 0986 | 11992 | 413.7 | 1883 |
| Raw after changing the disinfecting agent injection mode | 7.38 | 2638 | 4270 | 302 | 569 |
| RO. Concentrate NaOCl & SMBS 0.0% | 7.65 | 6400 | 10158 | 502 | 1537 |
Table 5: Reject and Raw samples physical properties before and after NaOCl injection mode change.
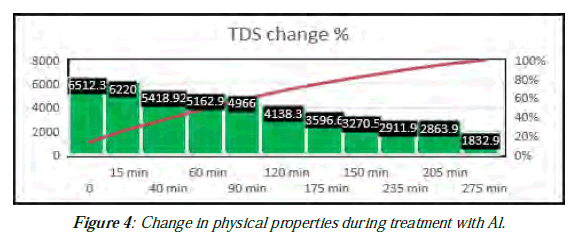
| Time min | 0.0 | 15 min | 40 min | 60 min | 90 min | 120 min | 150 min | 175 min | 200min | 220 min | 240 min |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TDS | 6890 | 6730 | 6015 | 5865 | 5850 | 5057 | 4147 | 4690 | 3872 | 3966 | 2665 |
| PH | 7.762 | 7.34 | 7.01 | 6.89 | 6.75 | 6.16 | 5.939 | 6.225 | 6.23 | 6.31 | 7.37 |
| Temp | 27.9 | 29.1 | 30.5 | 31.8 | 33.9 | 36.1 | 38.4 | 40.2 | 42.6 | 43.1 | 47.7 |
| TDS at 25ºC | 6512.3 | 6220 | 5418.92 | 5162.9 | 4966 | 4138.3 | 3270.5 | 3596.6 | 2863.9 | 2911.9 | 1832.9 |
| Red % | 0.0 | 4.49 | 16.8 | 20.72 | 23.74 | 36.45 | 49.78 | 44.8 | 56.02 | 55.3 | 71.85 |
Table 6: Change in physical properties during treatment with Al.
Results and Discussion
In our way to treat the high TDS in ROC, as the main goal is changing the concentrate from a waste to a resource through treatment and beneficial use, which minimize both costs and environmental impacts. And as the RO permeate increasing, ROC volume reduction will lead to a reduction in the volume of wastewater to waste water treatment plant and finally reduction of the volume of treated waste water needs to be discharged. We found that the following means could be used to treat ROC (240m3/h) in WACCPP as an inland case.
Quality improvement by reduction of TDS in RO’s feed water (treatment scenario) which could increase the RO units permeate, consequently decreasing the RO reject water volume, that was obtained by changing the disinfection mode from continuous to a shock mode; as by injecting the disinfecting agent (NaOCl) on a shock mode the amount of Na+ and Cl- ions added Eqs. (1,2), where, HOCl, and OCl- is referred to as free available chlorine or free residual chlorine, expressed as ppm Cl2.
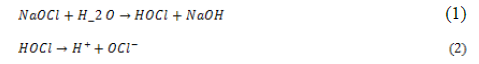
And consequently SMBS (Na, HSO3 and SO4), commonly used for removal of free chlorine, when dissolved in water, sodium bisulfite (SBS) is formed from SMBS, then SBS reduces hypochlorous acid Eqs. (3,4):
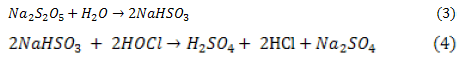
Finally, a significant reduction in TDS of both RO feed and RO reject water by about 7% as appears in (Tables 4,5) and (Figure 1).
RO concentrate disposal (direct disposal scenario), as disposal methods depends on concentrate quantity and quality, permitting requirements, geography and geology, costs, and potential environmental impacts, after all these issues were taken in consideration;
a. Evaporation ponds were used; by constructing an evaporation ponds by a total capacity reach to 500000m3 as a traditional ROC disposal method Image (3).
b. Deep well injection was used; by digging 5 deep wells, especially after add on the combined cycle extension.
c. By RO concentrate volume reduction (ROC volume reduction scenario) in WACCPP is by:
d. As by change the fuel used from heavy oil, which requires a 175m3 (21.8 m3/h for each unit) service water for de- NOx, as the main fuel in combustion in the gas turbines in the simple cycle, to the natural gas, which does not require this quantity, consequently, led to a reduction in ROC by about 105m3.
e. As by change the fuel used from heavy oil to natural gas as the main fuel, the heavy fuel oil washing water (about 35m3/h) was not going be used ever again as the fuel water treatment units not used again, this will lead to a reduction in ROC by about 21m3. Then the total reduction in demineralized water is by about 210m3 due to using natural gas instead of HFO which led to a reduction in the ROC by about 126m3, i.e. reduced to about the half of the total amount of the planned ROC effluent [21-23].
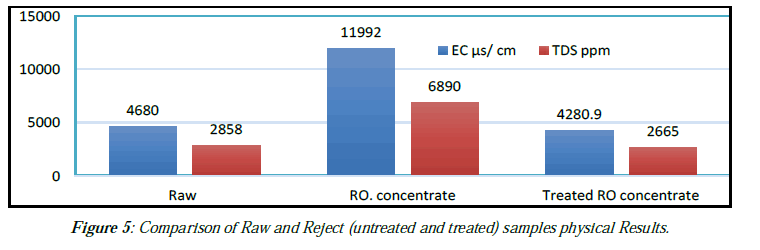
Experimental work for ROC quality improvement in the laboratory using Al electrodes for TDS reduction from ROC in a process called electrocoagulation (EC) was succeed to reduce the TDS from 6512.3 to 1832.9 ppm by about 71.85 % (25 ºC) and even decreased less than that for feed water (2638 ppm) as shown in (Figure 2,3,4,5,6 ) and (Table 5,7).
| Item | pH | EC μs/ cm | TDS ppm | T. Alk ppm | TH. ppm |
|---|---|---|---|---|---|
| RO. concentrate | 7.75 | 11992 | 0986 | 413.7 | 1883 |
| Raw | 7.25 | 4680 | 2858 | 250 | 705 |
| Treated RO concentrate | 7.37 | 4280.9 | 2665 | 150 | 650 |
Table 7: Comparison of Raw and Reject (untreated and treated) samples physical.
Conclusion
In accordance to the desire for reduction of the pollution load to the environmental, the ways used to reduce the amount of ROC that seeps to the neighbor environment of the WACCPP are; the managing the injection of disinfecting agent resulted in increases of the RO permeate consequently, reduces the rejected water (ROC). While the use of natural gas reduced the ROC by about 126 m3i.e., to half of the total rejected water (240m3), also, the quality improvement treatment process as electrocoagulation treatment process for ROC using Al electrodes led to reduction of TDS to less than that for RO feed water.
Recommendations
As the ways of reduction of ROC water environmental impact are the improvement of its quality or decrease its volume, so we suggest the following recommendations:
Stop using NaOCl as disinfecting agent and use O3 or UV rays instead, consequentially, the SMBS will stop also, which will reduce the amount of TDS so, increase the production of RO units and decreases the ROC amount.
Reuse the blowdown water of the steam boilers as raw water, where its amount reaches to 50m3/h with TDS reach to a 50ppm, as by mixing this amount (50m3) of low TDS water with a bout 300m3 (2858 ppm) the TDS of the 350m3 that will used as feed water per hour will be reduced to about 2457ppm which less than that for raw water used, consequently increases the production of RO units and decreases the ROC amount.
Re-introduce the produced ROC after reducing its TDS to less than 6000ppm by mixing with raw water, to a reverse osmosis’s units from the second RO line (contain 5 units).
On the long run they should works on to dispose ROC to the municipal sewar production (outlet) which will benefits from one side, in the reduction of microorganisms in the effluent to the surface water, and its TDS also will be reduced on the other side.
References
- Abou Zakhem B, Hafez R. Heavy metal pollution index for GW quality assessment in Damascus Oasis, Syria Environ Ear Scien, 2015;73-10: 6591-600.
- Ahmed M, Shayya WH, Hoey D, et al. Brine disposal from reverse osmosis desalination plants in Oman and the United Arab Emirates. Desalina, 2001;133(2):135-47.
- Al-Raad AA, Hanafiah MM, Naje AS et al. Treatment of saline water using electrocoagulation with combined electrical connection of electrodes. Proces. 2019;7:242.
- APHA (American Public Health Association), American water works association (AWWA) and Water Environmental Federation (WEF). Standard methods for the examination of water and wastewater, 2017;23nd Edition.
- ASCE (1990). Agricultural Salinity Assessment and Management. Manual No. 71. American Society of Civil Engineers. [NRC. 2004]. “Review of the Desalination and Water Purification Technology Roadmap.” The National Research Council. The National Academies Press.
- Ayers RS, Westcot DW. Water quality for agriculture, Food and Agriculture Organiz of the U.N. Rome. 1985.
- Carollo. Water Desalination Concentrate Management and Piloting” South Florida Water Management District. 2009 . pw://Carollo/Documents/Client/FL/SFWMD/8178A00/Deliverables/Final Report.doc.
- Cress B. Las Animas Water Treatment Plant. 1999. Available at: www2.hawaii. edu/nabil/lasanima.htm.
- Dohare DE, Trivedi ER. A review on membrane bioreactors: an emerging technology for industrial wastewater treatment. Interna J Emer Techno Advan Engin J. 2014;4(12):226-36.
- Gaber AM, Kamal-El-Dean AM, Manaa HH. et al. Hydrochemical classification of groundwater in west assiut combined cycle power’s area, Assiut, Egypt, Egypt Sug J. 2020;14:51-77.
- Gorenflo A, Brusilovsky M, Faigon M, et al. High pH operation in seawater reverse osmosis permeate: First results from the world’s largest SWRO plant in Ashkelon. Desal. 2007;203:82-90.
- Janpoor F, Torabian A, Khatibikamal V. Treatment of laundry waste-water by electrocoagulation, J Chem Technol Biotechnol. 2001;6(8):1113–20.
- Katzir L, Volkmann Y, Daltrophe N. et al. WAIV e wind aided intensified evaporation for brine volume reduction and generating mineral byproducts. Desal and Water Treat. 2010;13:63-73.
- Khan SJ, Murchl D, Rhodes M et al. Management of concentrated waste streams from high-pressure membrane water treatment systems. Criti Revi Environ Sci and Tech. 2009;39(5):367-415.
- Lattemann S, Hopner T. Environ. impact and impact assessment of seawater desalination. Desal. 2008;220:1-15.
- Manaa HH, Gaber AM, Bady M et al. Evaluation of groundwater suitability for different applications in the area of West Assiut Power Plant, Egypt. Intern J Environ Sci Techno. 2022;19:3031–44.
- Mohamed AMO, Maraqa M, AL Handhaly J. Impact of land disposal of reject brine from desalination plants on soil and groundwater. Desalina. 2005;182:411-33.
- Owen G, Bandi M, Howell JA. Churchouse SJ Economic assessment of membrane processes for water and waste water treatment. J Membr Sci. 1995;102:77-91.
- Sánchez-Lizaso JL, Romero J, Ruiz J, et al. Salinity tolerance of the Mediterranean seagrass Posidonia oceanica: recommendations to minimize the impact of brine discharges from desalination plants, Desalination. 2008;221:602–7.
- Sethi S, Walker S, Drewes JE. Existing & emerging concentrate minimization & disposal practices for membrane systems. Fla. Water Resour J. 2006;38-48.
- Sposito G. The chemistry of soils. 1989; New York: Oxford University Press.
- Xu P, Cath TY, Robertson AP, et al. Critical review of desalination concentrate management, treatment and beneficial use. Environ Enginee Sci. 2013;30(8):502-14.
- Younos T. Environmental issues of desalination. J Contemp Water Res Educ. 2005;132:11-8.