Research Article - Biomedical Research (2017) Health Science and Bio Convergence Technology: Edition-II
The effects of Scutellariae radix extract gargling solution on the prevention of periodontal disease
Yu-Rin Kim1 and Seoul-Hee Nam2*
1Department of Dental Hygiene, Silla University, Busan, Republic of Korea
2Department of Dental Hygiene, College of Health Science, Kangwon National University, Samcheok-si, Republic of Korea
- *Corresponding Author:
- Seoul-Hee Nam
Department of Dental Hygiene, College of Health Science
Kangwon National University, Republic of Korea
Accepted date: May 17, 2017
Abstract
This study was to evaluate the effect of Scutellariae radix extract on periodontal disease. A 500 g of Scutellariae radix was added to 70% methanol and extracted for 3 h, followed by filtration and freeze drying. The freeze dried specimen was added to 10% Dimethyl Sulfoxide (DMSO) solution, and the diluted solution was used in this study, which was conducted after obtaining the approval of the IRB of Inje University (No. 2-1041024-AB-N-01-20140514-HR-055-04; approval date: 2014-08-27). The study enrolled patients without systemic disease or drawbacks with data collection. They visited the M Dentist Office located in Busan from September 2015 to January 2016. They were equally divided into three groups. The 10 subjects were placed in 0.9% saline (JW Pharmaceutical Corporation) gargle group, 10 subjects were placed in 0.005% chlorhexidine (Bukwang Pharmaceutical Co., Ltd.) gargle group, and 10 subjects were placed in 10% Scutellaria root gargle group. The periodontal disease variables for the groups prior to the gargle treatment were the same. Each group gargled a 15 ml solution for 1 min and expectorated for 1 min until there was no gargle solution left. The bacterial count was calculated by using a phase contrast microscope and the O’Leary index. As seen with the naked eye, the O’Leary index was reduced in the order of saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group, and the gap among the groups was distinct (p<0.005). However, for the active bacteria, Spirochaeta, the chlorhexidine gargle group and Scutellaria root gargle group showed a similar effect, and their gap was not statistically significant (p>0.005). Since many side effects are reported on the chemical solution, chlorhexidine, substituting it with natural Scutellaria root gargle solution will enable a safe and efficient oral care.
Keywords
Chlorhexidine, O'Leary index, Periodontitis, Scutellaria radix, Spirochaeta
Introduction
Periodontal disease occurs to 90% of adults and it is a threat to oral health because it can cause loss of function. Its morbidity rate and severity are increasing steadily and mostly high during and after late-middle-aged adults. Periodontal disease refers to an inflammation in the dental supporting apparatus due to bacteria. In addition, the bacterial composition in the dental plaque of a healthy gingiva and periodontal disease gingiva are different [1].
Studies on periodontal disease were mainly focused on controlling its progress by efficiently removing the dental plaque [2]. Dental plaque is formed when microorganisms adhere to the dental surface and constitute either a homotypic bond or a heterotypic bond between the cells, along with the production of an extracellular matrix. Dental plaque can inhibit external materials from entering, as its extracellular matrix forms a thick layer, and its resistance to antibiotics and antibacterial substances are high, as compared to bioaerosol due to genetic morphism through cell-to-cell interaction, thereby making it difficult to remove [3]. The deposition and maturity of the dental plaque determine the severity of the periodontal diseases. Therefore, maintaining a good oral hygiene and managing the dental plaque regularly are necessary for the prevention of periodontal diseases [4]. In particular, chlorhexidine was discovered in the late 1940s, and it was used diversely in gynecology, urology, and ophthalmology. It has been used for various purposes (e.g., treatment for skin infection or burn, and sterilization of surgical instruments before surgery) [5], since it has antimicrobial activity against gram-positive bacteria, gramnegative bacteria, yeast, fungi, and anaerobic species [6]. It has been recommended for the chemical dental plaque control of handicapped patients, who are having difficulties with tooth brushing, and for circumstances where the jaw fracture is fixed, including after periodontic surgery and after fixation of the orthodontic appliances [7]. In particular, it selectively retrains Streptococcus mutans, which can be used for periodontal disease treatment and dental caries prevention purposes. However, its bitter taste makes it difficult to be used as a gargle solution for children, and it cannot be used long term, since it can proliferate on the dental surface and cause tongue discoloration [8]. The discoloration can take place in both hard tissue and soft tissue, and it mainly occurs on the dental surface with its severity, while the intensity varies according to the eating habit and tooth brushing pattern of the patients. While the mechanism of discoloration has not yet manifested, Rolla et al. [9] mentioned that the denaturation of chlorhexidine, formaldehyde, and tannic acid is due to the dark brown discoloration caused by the deposition of ferrous and sulfur ions, as well as the denaturation of protein inside the dental plaque and acquired films. Jansen [10] stated that the dental surface is pigmented by the ionic bond between pigment anion and chlorhexidine cation. Low-concentration chlorhexidine, zinc, lanthanum, fluoric, and oxidant were suggested as an attempt to minimize the discoloration [11-13]; however, a gargle solution made with natural Korean medicine extracts are actively being experimented due to the side effects caused by chemical agents. Psoralea corylifolia [14] and Erythrina variegata [15] have excellent antimicrobial effects. Meanwhile, Curcuma aromatica [16] and Asarum sieboldii Miquel [17] have been reported to have an antibiotic effect against S. mutans. For this reason, a gargle solution composed of natural extracts has an excellent antibacterial activity. It is also considered as an effective oral care method. In particular, the Scutellaria root (Scutellaria radix) is a perennial herbaceous plant that is used as a medicine and grown all over the country. Its main constituents are flavonoids, such as baicalein, wogonin, and skullcap flavone I and II [18]. Furthermore, it is known for its ability to inhibit contact hypersensitivity, as well as its antioxidant, antimicrobial, antiviral liver function protection, anti-inflammatory, anticonvulsant, antihyperlipidemic, and anti-anxiety effects [19]. Although many studies were conducted on Scutellaria root extract due to its excellent antibacterial activity [20-22], there are only a few reports regarding its use in dentistry. There are only a few studies conducted on periodontal disease, which is closely related to dental plaque adhesion, as well as oral bacteria count and activity, whereas a gargle solution composed of natural extracts is under active investigation. In addition, the clinical data obtained by the clinical application is deemed insufficient.
Therefore, this clinical study aims to assess the potential of Scutellaria root extract in order to prevent periodontal disease with its antimicrobial activity, and investigate its effect on the progress of periodontal disease.
Materials and Methods
Scutellaria radix extract
A 500 g of Scutellaria radix was purchased from Busan Hyundai herb merchandise. A 100 g of crushed lotus leaves was diluted 1:10 in 70% methanol and extracted for 3 h under 65°C heating mantle. The extract was filtered 3 times with a filter paper (Advantec No. 2, Toyo, Japan). An aspirator (A-3S, EYELA Co., Japan) and a freeze dryer (Ilshin Lab. Co., Korea) were used to concentrate and freeze dry the Scutellaria root extract. The freeze dried specimen was dissolved and diluted with 10% dimethyl sulfoxide (DMSO) and stored at -20°C.
Study subjects
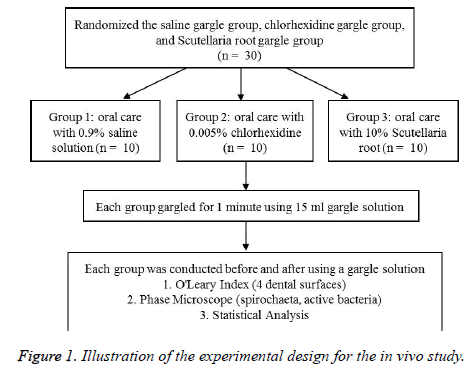
This study was conducted after obtaining the approval of the IRB of Inje University (No. 2-1041024-AB-N-01 – 20140514- HR-055-04; approval date: 2014-08-27). The study enrolled patients who visited the M Dentist Office located in Busan from September 2015 to January 2016. A dental hygienist with more than 10 y’ experience informed the patients about the objectives of the study, and among the 36 patients who filled out the questionnaire, only 30 of them who had no limitations in the data collection were selected. Each of the 30 patients was randomly assigned to three groups. The 10 patients in the 0.9% saline (JW Pharmaceutical) gargle group, 10 patients in the 0.005% chlorhexidine (Bukwang Pharmaceutical Co., Ltd.) gargle group, and the remaining 10 patients in the 10% Scutellaria root gargle group were checked for periodontal disease variables before and after the gargle process that included a one-minute gargle with 15 ml of designated solution, and expectorated until there was no gargle solution left. The overall design of the experiment proceeds is the same as Figure 1.
Clinical application
The O’Leary index was measured in the saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group. Meanwhile, the Spirochaeta (spiro) and active bacteria count were assessed by the phase microscope. The X1 group gargled for 1 min by using 15 ml of 0.9% saline, the X2 group gargled for 1 min by using 15 ml of 0.005% chlorhexidine, and the X3 group gargled for 1 min by using 15 ml of 10% Scutellaria root. The microbe count was carried out again after the subjects expectorated until there was no gargle solution left.
O’Leary index
O'Leary et al.’s dental plaque test (O’Leary index) was carried out. We discolored all teeth in the oral cavity with a dental surface discoloration agent and calculated the level of adherence (%) by using the plaque control score (O'Leary index), which gives 1 point if the dental plaque adheres to the dental surfaces of 4 teeth (mesial, efferent, facies, and lingual) and 0 if not [23].
Phase microscope
A sterilized probe was used to confirm the type of microbes. The subgingival and supragingival dental plaque of the left and right post-lingual surface of the mandible was collected and smeared on a slide. After applying a drop of saline, the slide was mounted with a slide cover without making any bubbles, and then it was observed by using a phase microscope (DCS6002, Doctor prevent Co., Seoul, Korea) by a factor of 4,300. The movement of the microbes were monitored without staining, and they were differentiated to active bacteria and inactive bacteria. The relative increase of Spirochaeta may influence the progression of periodontal disease.
Statistical analysis
The statistical analysis of the saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group was conducted by using the IBM SPSS ver. 21.0 (IBM Co., Armonk, NY, USA). The O’Leary index and Spirochaeta count, which was quantified by a program that studies microbes observed with a phase microscope, proceeded to oneway ANOVA test and Tukey’s post hoc test under p=0.05 significance level in order to test the significance among the groups.
Results
Comparison of the O’Leary index
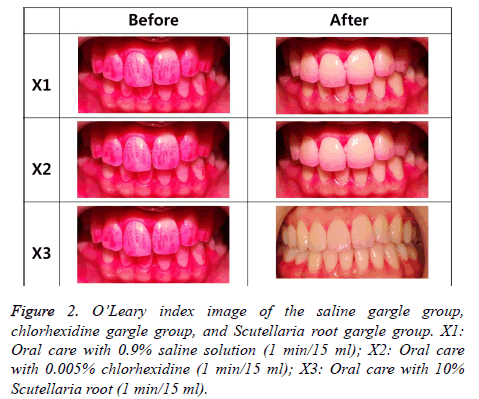
The result of the O’Leary index before using a gargle solution was high in all groups, while it was reduced in the order of saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group after using a gargle solution (Figure 2). As shown in Table 1, there was a distinct difference between the chlorhexidine gargle group and the Scutellaria root gargle group (p<0.05).
| N | Before | After | ||||
|---|---|---|---|---|---|---|
| M ± SD | p-values | M ± SD | p-values | |||
| O'Leary index | X1 | 10 | 96.00 ± 8.43 | 0.054 | 87.60 ± 11.01a | 0.000* |
| X2 | 10 | 77.80 ± 37.17 | 56.60 ± 23.99b | |||
| X3 | 10 | 68.40 ± 18.93 | 22.00 ± 17.39c | |||
Table 1. Comparison of the O’Leary index.
Change in phase microscope image
The active bacteria count before using a gargle solution was high in all groups, and it was reduced in the order of saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group after using a gargle solution (Figure 3). As shown in Table 2, the chlorhexidine gargle group and Scutellaria root gargle group showed similar antimicrobial effects against active bacteria and Spirochaeta (p<0.05).
| N | Before | After | ||||
|---|---|---|---|---|---|---|
| M ± SD | p-values | M ± SD | p-values | |||
| Micro_a | X1 | 10 | 88.00 ± 7.89 | 0.202 | 68.00 ± 32.93a | 0.004* |
| X2 | 10 | 86.00 ± 14.29 | 37.00 ± 23.94bc | |||
| X3 | 10 | 75.00 ± 24.49 | 26.40 ± 21.18bc | |||
| Spiro | X1 | 10 | 6.00 ± 5.16 | 1 | 4.00 ± 5.16Ma | 0.007* |
| X2 | 10 | 6.00 ± 8.43 | 0.00 ± 0.00bc | |||
| X3 | 10 | 6.00 ± 6.22 | 0.00 ± 0.00bc | |||
Table 2. Bacterial count before and after the use of a gargle solution of the saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group.
Discussion
Pathogenic bacterium produces many kinds of diseases, and it causes pain and loss of teeth. In particular, periodontitis is a mixed infection of multiple bacteria strains that is characterized by gingivitis due to the disposition of the dental plaques. It has progressed into damaging the dental supporting apparatus and loss of teeth when inflammation is spread by the reactions in the periodontal tissues [24]. Although various physical methods are used to manage dental plaque, the measures conducted by the experts are short term and limited. Tooth brushing is a self-care method that has been proven in many studies; however, its efficacy depends on a person’s motivation [25], and it will be laborious to a person with limited hand movement. Chemical dental plaque care is used in order to make up for these limitations, and the gargle solution, in particular, is easy and convenient to use. Various gargle solutions in the overseas market were assessed for their antibacterial activity on dental plaques, and their efficacy has been proven in both laboratory and clinical studies. Upon the assessment of the marketed products with the main ingredients of chlorhexidine, essential oil, fluoric amine, and fluoric tin, each product had different antibacterial activity on dental plaque [26,27]. This study compared the periodontal disease variables among the saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group. As a result of this study, the O’Leary index after the use of a gargle solution was reduced in the order of saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group. The gap among the three groups was statistically significant and distinct between the chlorhexidine gargle group and the Scutellaria root gargle group. The number of active bacteria after using a gargle solution, as seen in the phase microscope, was reduced in the order of saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group. The objectives of periodontal disease treatment are to cease the progress of inflammation. There is still an ongoing debate as to whether it can be cured completely [28]. Gargle solution and antibiotics are generally used to decrease pathogenic bacteria and help prevent secondary infection after surgical intervention. However, antibiotics, antimicrobials, and anti-inflammatory agents are applied in a limited manner, since they account for side effects, such as manifestation of resistant bacteria, anaphylaxis, gastrointestinal disturbance, and dental discoloration [29]. The limitations of this study are that Scutellaria root gargle group did not have a significant effect on active bacteria and Spirochaeta, but they can be used to prevent and treat periodontal disease more effectively by using natural ingredients without the side effects.
In order to overcome the side effects of the conventional products, investigations of the natural ingredients for their application in the prevention and treatment of dental disease are actively being made [30,31]. Liquorice [32] is known to effectively diminish bacteria that can cause periodontal disease and dental caries. Moreover, Their et al. has reported the efficacy of corn extract in periodontitis [33], dental plaque index decreasing effect and gingiva index decreasing effect [34].
Zea mays L. and Magnolia extract complex have shown more effective antimicrobial effects against germs that can cause periodontal disease, as compared to using its ingredient separately [35]. This study used Scutellariae radix, the root of Scutellariae baicalensis with its periderm removed [36], of which the extract was reported to have an anti-inflammatory effect and the ability to enhance the cytological function of gingiva fibroblasts [37]. For the antibacterial activity, it was reported that Scutellaria root extract has an antimicrobial effect against Streptococcus mutans, which usually causes dental caries [20], and an antibiotic effect against Pseudomonas aeruginosa [21]. The antibacterial activity of Scutellaria root extract was also reported [22]. This study was conducted in order to obtain fundamental data regarding the clinical application of Scutellariae radix extract and its efficacy.
This study clinically applied and comparatively analysed the saline gargle group (X1), chlorhexidine gargle group (X2), and Scutellaria root gargle group (X3). As a result, the O’Leary index was reduced in the saline gargle group, chlorhexidine gargle group, Scutellaria root gargle group, and the gap among the groups was distinct (p<0.005). The gap between the chlorhexidine gargle group and Scutellaria root gargle group was not statistically significant, which means that their effects are similar against the active bacteria and Spirochaeta, (p>0.005), although, the three groups showed a significant difference in the phase microscope image.
Therefore, Scutellaria root extract can help prevent the progression of periodontal disease by inhibiting the growth of pathogenic bacteria, and it is capable of dental plaque generation care. Although the chlorhexidine gargle group and Scutellaria root gargle group did not show any significant effect on active bacteria and Spirochaeta, periodontal disease will be more thoroughly prevented and treated by using nature-derived ingredients that do not have any anticipated side effects without using chemical ingredients that consist of chlorhexidine, which accounts for many side effects. This safe ingredient is expected to replace the conventional oral hygiene products.
Conclusion
In conclusion, all periodontal disease variables have decreased after using the gargle solution in the order of saline gargle group, chlorhexidine gargle group, and Scutellaria root gargle group. The three groups did not show any significant difference in the O’Leary index, active bacteria, and Spirochaeta prior to the use of the gargle solution; however, after using the gargle solution, the three groups showed a significantly different efficacy in the O’Leary index. In addition, the chlorhexidine gargle group and Scutellaria root gargle group showed a similar efficacy on the bacteria. Dental plaque care and oral care that reduce bacterial activity are critical to preventing periodontal diseases. Therefore, it is better to use the natural Scutellaria root extract that shows a similar effect but with less side effects than the commonly used chlorhexidine in dental practice.
Conflict of Interest
The authors report no conflicts of interest related to this study. The author does not have any financial interest in the companies whose materials are included in the article.
References
- Oh HY, Kim CH, Park YH, Lim SH, Kim J. The comparison on periodontal attitude and oral health promotion behavior by dental hygiene process applies. J Korean Soc Dent Hyg 2012; 12: 861-870.
- Min EG, Kim YH, Kum SI, Han YH. Inhibition of growth and collagenase activity of the extract from Salvia miltiorrhiza against microorganisms causing periodontal diseases. Kor J Microbiol 2004; 40: 111-114.
- Marsh PD. Dental plaque as a biofilm and a microbial community-implications for health and disease. BMC Oral Health 2006; 6: 14.
- Greenstein G, Polson A. The role of local drug delivery in the management of periodontal diseases: a comprehensive review. J Periodontol 1998; 69: 507-520.
- Nielsen ML, Raahave D, Stage JG, Justesen T. Anaerobic and aerobic skin bacteria before and after skin disinfection with chlorhexidine: an experimental study in volunteers. J Clin Pathol 1975; 28: 793-797.
- Hennessey TS. Some antibacterial properties of chlorhexidine. J Periodontal Res 1973; 12: 61-67.
- Tonelli PM, Hume WR, Kenney EB. Chlorhexidine: a review of the literature. J West Soc Periodontol Periodontal 1983; 31: 5-10.
- Fardal O, Turnbull RS. A review of the literature on use of chlorhexidine in dentistry. J Am Dent Assoc 1986; 112: 863-869.
- Rolla G, Loe H, Schiott CR. The affinity of chlorhexidine for hydroxyapatite and salivary mucins. J Periodontal Res 1970; 5: 90-95.
- Jensen JE. Binding of dyes to chlorhexidine treated hydroxyapatitie. Scand J Dent Res 1977; 85: 334-340.
- Dolles OK, Eriksen HM, Gjermo P. Tooth staining during 2 years use of chlorhexidine and fluoride-containing dentifrices. Scand J Dent Res 1979; 87: 268-274.
- Eriksen HM, Solheim H, Nordbo H. Chemical plaque control and prevention of extrinsic tooth discoloration in vivo. Acta Odontol Scand 1983; 41: 87-91.
- Ellingsen JE, Rolla G, Eriksen HM. Extrinsic dental stain caused by chlorhexidine and other denaturing agents. J Clin Periodontol 1982; 9: 317-322.
- Shan L, Yang S, Zhang G, Zhou D, Qiu Z, Tian L, Yuan H, Feng Y, Shi X. Comparison of the inhibitory potential of Bavachalcone and corylin against UDP-glucuronosyltransferases. Evid Based Complement Alternat Med 2014; 2014: 958937-959002.
- Djeussi DE, Sandjo LP, Noumedem JA, Omosa LK, T Ngadjui B, Kuete V. Antibacterial activities of the methanol extracts and compounds from Erythrina sigmoidea against Gram-negative multi-drug resistant phenotypes. BMC Complement Altern Med 2015; 15: 453-459.
- Jeon WY, Lee MY, Shin IS, Jin SE, Ha H. Curcuma aromatica Water Extract Attenuates Ethanol-Induced Gastritis via Enhancement of Antioxidant Status. Evid Based Complement Alternat Med 2015; 2015: 582496-582502.
- Yu HH, Seo SJ, Kim YH. Effect of Asarum sieboldii extracts on the growth, acid production, adhesion, and insoluble glucan synthesis of Streptococcus mutans. Korean J Orient Physiol Pathol 2003; 17: 666-671.
- Hanasaki Y, Ogawa S, Fukui S. The correlation between active oxygens scavenging and antioxidative effects of flavonoids. Free Radic Biol Med 1994; 16: 845-850.
- Shang X, He X, He X, Li M, Zhang R. The genus Scutellaria an ethnopharmacological and phytochemical review. J Ethnopharmacol 2010; 128: 279-313.
- Moon YH, Lee YH, Min BS, Bae KH, Antibacterial constituents from Scutellariae Radix against Streptococcous mutans OMZ176. J Kor Phar 1997; 28: 99-103.
- Choi OK, Kim YS, Cho GS, Sung CK. Screening for antimicrobial activity from korean plants. J Kor Food Sci Nutr 2002; 25: 300-306.
- Lee CK, Seo JJ. Antimicrobial activity of Whangkumtang extract and Scutellariae Radix extract on the Food-Borne pathogens. J Kor Soc Food Sci Nutr 2005; 34: 1606-1610.
- Lang NP, Tonetti MS. Periodontal risk assessment (PRA) for patients in Supportive Periodontal Therapy (SPT). Oral Health Prev Dent 2003; 1: 7-16.
- Pradeep AR, Raj S, Aruna G, Chowdhry S. Gingival crevicular fluid and plasma levels of neuropeptide Substance-P in periodontal health, disease and after nonsurgical therapy. J Periodontal Res 2009; 44: 232-237.
- Barnett ML. The role of therapeutic antimicrobial mouthrinses in clinical practice: control of supragingival plaque and gingivitis. J Am Dent Assoc 2003; 134: 699-704.
- Gunsolley JC. Clinical efficacy of antimicrobial mouthrinses. J Dent 2010; 38: 6-10.
- Shapiro S, Giertsen E, Guggenheim B. An in vitro oral biofilm model for comparing the efficacy of antimicrobial mouthrinses. Caries Res 2002; 36: 93-100.
- Sreenivasan PK, Gaffar A. Antibacterials as anti-inflammatory agents: dual action agents for oral health. Antonie Van Leeuwenhoek 2008; 93: 227-239.
- Minn WK, Lee MS. Effects of ascorbic acid and unsaponifiable fractions of Zea Mays L. on the experimentally induced periodontitis in rats. J Periodontal Implant Sci 1988; 18: 6-23.
- Jeon ES, Yoon SH, Han MD. Antimicrobial activity of Streptococcus mutans by herbal medicine extracts. J Dent Hyg Sci 2002; 2: 31-38.
- Yoon HJ, Ha MO, Lee KH, Park YN, Cho MJ. Effects of medicinal plants against Streptococcus sobrinus B13 in hexane fraction. J Dent Hyg Sci 2007; 7: 197-200.
- Gafner S, Bergeron C, Villinski JR, Godejohann M, Kessler P, Cardellina JH, Ferreira D, Feghali K, Grenier D. Isoflavonoids and coumarins from Glycyrrhiza uralensis: antibacterial activity against oral pathogens and conversion of isoflavans into isoflavan-quinones during purification. J Nat Prod 2011; 74: 2514-2519.
- Thiers H, Jouanneteau, Zwingelstein. Unsaponifiable oil of corn germ; its therapeutic indications. Presse Med 1958; 66: 1293.
- Machuca G, Valencia S, Lacalle JR, Machuca C, Bullon P. A clinical assessment of the effectiveness of a mouthwash based on triclosan and on Zea mays L used as supplements to brushing. Quintessence Int 1997; 28: 329-335.
- Kim T, Choi EJ, Chung CP, Han SB, Ku Y. Antimicorbial effect of Zea Mays L. and Magnoliae cortex extract mixtures on periodontal pathogen and effect on human gingival fibroblast cellular activity. J Periodontal Implant Sci 2002; 32: 249-255.
- Kimura Y, Okuda H, Arichi S. Studies on scutellariae radix; XIII. Effects of various flavonoids on arachidonate metabolism in leukocytes. Planta Med 1985; 51: 132-136.
- Chung CP, Park JB, Bae KH. Pharmacological effects of methanolic extract from the root of Scutellaria baicalensis and its flavonoids on human gingival fibroblast. Planta Med 1995; 61: 150-153.