Research Article - Journal of Mental Health and Aging (2022) Volume 6, Issue 1
The association between cognitive reserve and depressive mood in older inpatients: Gender and age differences.
Alessandra Coin1*, Alessandro Bubola1, Enzo Manzato1, Giuseppe Sergi1, Caterina Trevisan1, Maria Devita2, Marianna Bizzotto3
1Geriatrics Division, Department of Medicine (DIMED), University of Padua, Italy
2Department of General Psychology (DPG), University of Padua, Italy
3Geriatrics Division, Dell’Angelo Hospital, Mestre-Venezia, Italy
- *Corresponding Author:
- Alessandra Coin
Geriatrics Division, Department of Medicine
(DIMED), University of Padua, Italy
E-mail: alessandra.coin@unipd.it
Received: 27-Nov-2021, Manuscript No. AAJMHA-21-15832; Editor assigned: 30-Nov-2021, PreQC No. AAJMHA-21-15832 (PQ); Reviewed: 15-Dec-2021, QC No. AAJMHA-21-15832; Revised: 29-Dec-2021, Manuscript No. AAJMHA-21-15832 (R); Published: 05-Jan-2022, DOI:10.35841/aajmha-6.1.101
Citation: Coin A, Bubola A, Manzato E, et al. The association between cognitive reserve and depressive mood in older inpatients: Gender and age differences. J Ment Health Aging. 2022; 6(1):101.
Abstract
Background: Evidence shows that Cognitive Reserve (CR) plays a protective role in neurological and acute/chronic diseases. Its effect on depressive mood, however, is still unclear, and is an issue that may be relevant in a hospital setting, since depressive mood has been associated with worse clinical outcomes. We aimed to explore the relationship between CR and depressive mood in older inpatients, including evaluation of gender- and age-related differences. Methods: This cross-sectional study involved 86 inpatients aged ≥65 years with a Mini-Mental State Examination ≥ 15. Participants underwent a multidimensional evaluation that included measuring CR with the Cognitive Reserve Index questionnaire (CRIq), which is divided into three sections (Education, Working Activity and Leisure Time). Depressive mood was assessed with the 15-item Geriatric Depression Scale (GDS-15). The relationship between CR and GDS was evaluated by linear regressions adjusted for potential confounders. Results: Significant associations between CR and GDS scores were found in the total sample (β=-0.03 [SE=0.02], p=0.047), especially in men (β=-0.05 [SE=0.02], p=0.005) and in individuals aged<85 years (β=-0.07 [SE=0.03], p=0.01). Of the individual CRI sections, CRI-Leisure Time was associated with GDS in the total sample (β=-0.03 [SE=0.01], p=0.03), in men (β=-0.03 [SE=0.01], p=0.03), and in participants <85 years (β=-0.05 [SE=0.02], p=0.01). Conclusions: CR seems to be inversely associated with depressive mood in older inpatients. This relationship is particularly marked for CR related to Leisure Time, which may be modifiable even in older age through social support and interaction.
Keywords
Cognitive reserve, Depressive mood, Age differences, Gender differences, Older inpatients.
Introduction
Mood disorders are frequent among older individuals and may therefore be regarded as an important public health problem [1]. In particular, depressive mood in the elderly has been found to be associated with several chronic diseases, an increased risk of dementia and suicide, and with overall mortality. In fact, the prevalence of depressive mood is higher in older individuals with ongoing health problems, and a recent study showed that almost one third of hospitalized patients were referred or diagnosed with depressive symptoms [2]. During hospitalization, depressive mood is associated with worse medical outcomes and an increased mean time of recovery, and has a negative influence on patients’ overall compliance with medical treatments and their efficacy [2]. It is therefore not only desirable, but also necessary to develop preventive strategies to reduce the risk and incidence of depression in the elderly [3]. Almeida and colleagues [4] identified four main modifiable risk factors in older people’s life-styles that together have a strong influence on the development of depressive symptoms in the subsequent 3-8 years: over/underweight, alcohol abuse, active or passive smoking, and physical inactivity. Physical exercise, in particular, seems to be one of the most promising nonpharmacological treatments of depression. In addition to these factors, education has been found to play an important role in protection from possible cognitive impairment associated with depressive mood, particularly in women [5]. On the other hand, stressful working activities were found to negatively affect depressive symptoms. Overall, these findings suggest that education, and working and leisure activities, which are all proxies of cognitive reserve, may significantly influence the individual’s psycho-affective status. Indeed, individuals with a higher CR seem better able to tolerate possible age-related pathophysiological alterations by exploiting their more effective and flexible cognitive and cerebral networks [6]. Although CR has been widely investigated in the field of dementia research, it is also emerging that CR has protective effects against other medical/psychopathological conditions. Evidence for its effects on depressive mood is, however, still scarce. The aim of the present study was to investigate the association between CR and depressive mood in older inpatients, including the presence of possible gender [7, 8] and age-specific differences. Our hypotheses were that the lower the CR, the severer the depressive mood, and that this relationship could be weaker in females and in the oldest old [9-11].
Materials and Methods
Study design and participants
This cross-sectional study involved patients hospitalized in the Geriatrics Division of the Department of Medicine (DIMED) of the University of Padua, the Ospedale Dell’Angelo (Mestre, Venice), and the Ospedale Sant’Antonio (Padua). A total of 86 inpatients were enrolled consecutively over the period January to September 2019 in accordance with the following inclusion criteria: age ≥ 65 years, Mini-Mental State Examination (MMSE) score ≥ 15, preserved basic sensory/perceptual and intellectual abilities to understand and execute the tests and questionnaires.
Measures
In addition to collecting demographic data from the inpatients, a multidimensional geriatric assessment of their cognitive, functional, clinical and social conditions was carried out by expert physicians in the geriatrics field. These assessments were made before hospital discharge and when the inpatients’ clinical conditions were judged to be stable.
Cognitive function: was assessed with the MMSE, a wellvalidated screening test for identifying cognitive impairment in older adults [12, 13]. Total scores range from a minimum of 0 to a maximum of 30, with <24 indicating the presence of cognitive impairment.
Functional status was assessed with the Activities of Daily Living [14] and Instrumental Activities of Daily Living (IADL) [13] scales.
Number and severity of chronic diseases were assessed with the Cumulative Illness Rating Scale [14], which covers 14 groups of diseases. For the purposes of this study, we considered the index of comorbidities, which are stratified within the scale according to the level of severity: absent (1 point), mild (2 points), moderate (3 points), severe (4 points), extremely severe (5 points).
Nutritional status was assessed with the Mini Nutritional Assessment (MNA), which includes anthropometric measures combined with an evaluation of lifestyle, mobility, drug use, dietary habits, and dependency in eating, and a selfassessment of health and nutritional status. Based on their MNA scores, individuals’ nutritional statuses were classified as “malnourished” (MNA <17), “at risk of malnutrition” (MNA 17-24), or “well-nourished” (MNA>24).
Depressive mood was assessed with the Geriatric Depression Scale (GDS) [14], a series of binary (yes/no) questions excluding somatic symptoms. In this study, we used the shorter, validated 15-item GDS, which is only suitable for patients with MMSE ≥15. A GDS score ≥5 was used to indicate the presence of depressive mood [14].
Cognitive Reserve was assessed with the Cognitive Reserve Index questionnaire (CRIq) [15], which includes 20 items across the three major proxies of CR [16, 17]: CRI-Education (total number of years in education and on other training courses); CRI-Working Activity (all professions carried out from the age of 18 classified into 5 levels with increasing cognitive involvement); and CRI-Leisure Time (cognitively stimulating leisure occupations). CRI-Total scores were calculated as the average of the three sub-scores, standardized and transposed to a scale with a mean of 100 and a standard deviation of 15. These CRI-Total scores were then classified as low (≤ 70), mediumlow (70-84), medium (85-114), medium-high (115-130), or high (≥ 130). Where inpatients had cognitive deficits in memory or attention, their next of kin replied to the questions.
Statistical analyses
Quantitative variables normally distributed were expressed as means ± standard deviation (SD), those non-normally distributed as median and interquartile range (IQR). Categorical variables were expressed as number and percentage (%). Depending on the distribution of the variables, parametric (Student t-test) or non-parametric (Mann-Whitney test) analyses were used to compare individuals by CRI category. The association between CRI (total score and sub-scores) and GDS was assessed with a generalized linear model adjusted for potential confounders, and the strength of the association was expressed as β coefficient and standard error (SE). All analyses were carried out firstly on the total sample, and then stratified by gender and age (using 85 years as cut-off, in accordance with Baltes & Smith’s (2003) aging classification) [18]. A p-value of 0.05 was adopted as the level of statistical significance for the tests. All statistical analyses were performed with SPSS (version 25, IBM).
Results
Our sample consisted of 86 individuals (mean age 85 ± 6 years), comprising 58 women (67.4%) and 28 men (32.6%). Of the total sample, 47.7% had MMSE scores <24, 41.9% had GDS scores indicating depressive mood, and with respect to social status 33.7% lived alone.
The characteristics of the sample as a whole and stratified by gender are shown in Table 1.
| Variables | All (n=86) | Women (n=58) | Men (n=28) | p-value |
|---|---|---|---|---|
| Age* | 84.8 ± 5.97 | 85.34 ± 6.11 | 83.68 ± 5.61 | n.s. |
| GDS | 4.69 ± 3.69 | 4.5 (8-3) | 3 (4.75-1) | 0.03 |
| CRI* | 108.36 ± 28.89 | 103.21 ± 18.62 | 119.04 ± 29.81 | <0.001 |
| CRI Education | 102.16 ± 18.85 | 95 (107-90.75) | 99 (121.50–90.25) | n.s. |
| CRI Working Activity | 88.97 ± 21.28 | 78.5 (92.25-70.75) | 99.5 (107–87.25) | <0.001 |
| CRI Leisure Time* | 127.66 ± 30.29 | 125.02 ± 27.17 | 133.14 ± 135.81 | n.s. |
| MMSE* | 23.67 ± 3.96 | 23.23 ± 3.97 | 24.58 ± 3.83 | n.s. |
| ADL | 3.30 ± 1.89 | 3 (4.25–2) | 4.5 (6–1.25) | n.s. |
| IADL | 2.83 ± 2.17 | 3 (4–1) | 3 (5–1) | n.s. |
| MNA | 19.70 ± 5.12 | 19.75 (24–15) | 23.75 (24–19.13) | 0.02 |
| CIRS | 5.33 ± 2.38 | 5 (7–3) | 5 (6.75–3) | n.s. |
Table 1. Characteristics of the sample as a whole and stratified by gender.
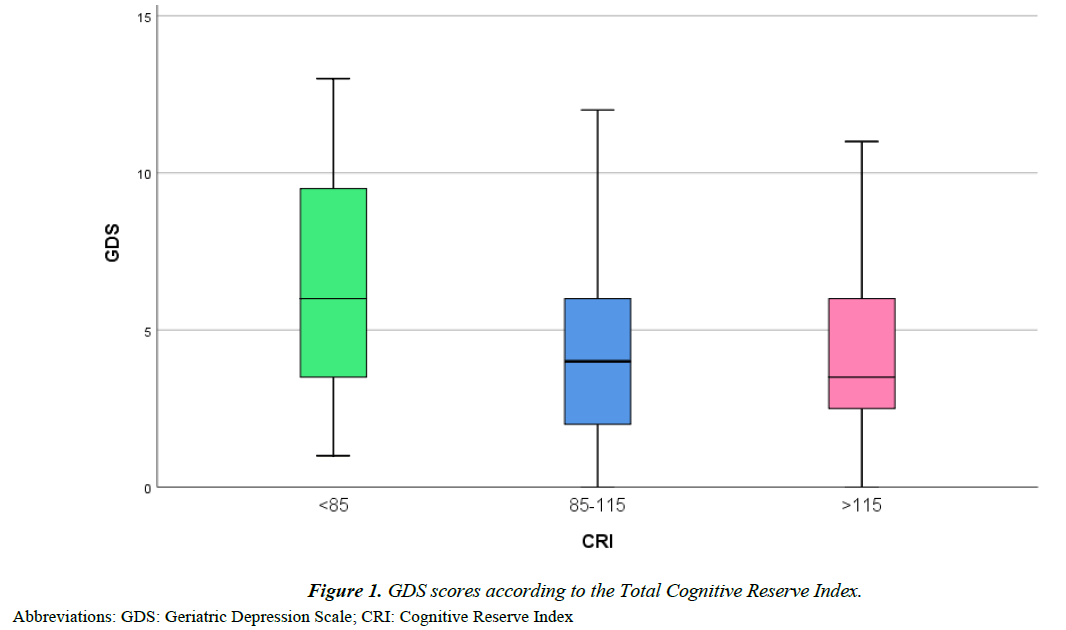
Significant differences were found between men and women for MNA, GDS, CRI-Total score and CRI-Leisure Time, with women having worse results. Analysis of the sample by age (Supplementary Table 1) showed that the only significant difference was in GDS scores, which were higher for individuals aged <85 years than those ≥85 years (median [IQR]: 4.5 (8.75– 3) vs 3 (6–1), p=0.03). Figure 1 reports the GDS scores of the study participants classified by CRI-Total score. Although the differences were not statistically significant, we observed that GDS tended to be higher with a lower CR. The linear regression analysis (Table 2), after adjusting for potential confounders, revealed a significant inverse association between CRI-Total score, whether considered as a continuous or a categorical variable, and GDS. In particular, inpatients with a CRI-Total score <85 had a GDS score that was on average more than 3 points higher than that of individuals with the highest CRI (β=3.34, SE=1.22, p=0.01). This association was more marked among men and those aged <85 years. Regarding the association between the different CRI sub-scores and GDS, we observed a slight but significant indirect association between CRI-Leisure Time and GDS scores. This result was confirmed when all the CRI sub-scores were included in the same model, and supports an independent association between CRI-Leisure Time and GDS. As in the main analysis, this association was more marked among patients aged <85 years. Regarding gender, only men exhibited significant inverse associations between GDS score and CRI-Working Activity, CRI Leisure Time and CRIEducation (marginally). When all CRI sub-scores were included in the same model, only CRI-Leisure Time was found to be independently associated with GDS (Table 3).
| Variables | Age ?85 (n=44) | Age ≥85 (n=42) | p-value |
|---|---|---|---|
| Age* | 80.07 ± 3.05 | 89.76 ± 3.88 | <0.001 |
| GDS | 4.5 (8.75–3) | 3 (6–1) | 0.03 |
| CRI* | 110.55 ± 25.32 | 106.07 ± 22.36 | n.s |
| CRI Education | 95 (103–89.25) | 96 (110.75–92.75) | n.s |
| CRI Working Activity | 86 (98.75–76) | 84.5 (102–71) | n.s |
| CRI Leisure Time* | 130.30 ± 28.38 | 124.9 ± 32.28 | n.s |
| MMSE* | 24.36 ± 3.46 | 22.95 ± 4.34 | n.s |
| ADL | 3.5 (5–2) | 3 (5–2) | n.s |
| IADL | 3 (4-2) | 2 (4.25–1) | n.s |
| MNA | 21 (24–15) | 20 (24–17.75) | n.s |
| CIRS | 5 (7–3) | 5 (7–3) | n.s |
Table 1. (Supplementary)Characteristics of the sample stratified by age.
| All (n=88) | Gender | Age | |||
|---|---|---|---|---|---|
| Women (n=58) | Men (n=28) | <85 years (n=44) | ≥ 85 (n=42) | ||
| CRI Total Score | |||||
| Per each 1-unit increase | - 0.03 (0.02)* | - 0.02 (0.03) | - 0.05 (0.02)** | - 0.07 (0.03)** | - 0.02 (0.02) |
| > 115 | [ref] | [ref] | [ref] | [ref] | [ref] |
| 85 – 115 | 1.38 (0.80) | 0.98 (1.11) | 2.37 (0.93)* | 1.96 (1.06) | 1.06 (1.11) |
| < 85 | 3.34 (1.22)** | 3.10 (1.56)* | 3.93 (2.03) † | 7.38 (1.87)*** | 1.83 (1.50) |
| CRI sub-scores (included in the model separately) | |||||
| CRI Education | - 0.01 (0.02) | 0.014 (0.04) | - 0.05 (0.02) † | - 0.03 (0.03) | - 0.03 (0.03) |
| CRI Working Activity | - 0.02 (0.02) | 0.01 (0.03) | - 0.04 (0.02)* | - 0.04 (0.03) | 0.00 (0.03) |
| CRI Leisure Time | - 0.03 (0.01)* | - 0.03 (0.02) | - 0.03 (0.01)* | - 0.05 (0.02)* | - 0.01 (0.02) |
| CRI sub-scores (all included in the model) | |||||
| CRI Education | 0.00 (0.03) | 0.03 (0.04) | 0.02 (0.05) | 0.00 (0.04) | - 0.04 (0.04) |
| CRI Working Activity | - 0.01 (0.03) | 0.01 (0.03) | - 0.05 (0.04) | - 0.03 (0.03) | 0.02 (0.04) |
| CRI Leisure Time | - 0.03 (0.01)* | - 0.04 (0.02) | - 0.03 (0.01)* | - 0.05 (0.02)* | - 0.01 (0.02) |
Table 3. β coefficients (and standard errors) for the association between Geriatric Depressive Scale and cognitive reserve in the sample as a whole and stratified by gender and by age.
Discussion
The present study showed that a lower CR, especially related to leisure time, was associated with higher depressive mood in older inpatients, and this relationship was stronger in men and those aged <85 years.
In line with previous studies, 42% of our sample of older inpatients had GDS scores indicating the presence of depressive mood, with a higher prevalence among women [19-23] Investigation of depressive mood in patients with different CRI levels showed a trend that, although not significant, suggested that individuals with a lower CRI had a higher GDS. This finding corroborates the findings of Evans et al., who found higher CR scores in healthy individuals compared with people with anxiety and/or depression [24]. Wu et al. showed that CR may be considered a “brain health factor” by modulating the association between cognitive functioning and lifestyle factors, such as physical exercise, smoking, alcohol consumption, dietary patterns, and cognitive and social activities. The same mechanisms may underlie a link between CR and depressive mood [25, 26]. Indeed, Almeida et al. found that individuals engaging in little or no physical activity were more likely to develop depression, as were smokers, alcohol abusers, and over/underweight people [27]. Leisure time activities in the CRIq include physical exercise, and social and entertainment activities, such as church attendance, clubs, theatre, exhibitions, conferences, embroidery, and playing cards. These activities could be enhanced in advanced aged, unlike the other two proxies of CR (education and working activity), which appear to be more static, and which in our sample did not show any strong relationship with depressive mood. Regarding possible gender differences in the association between CR and depressive mood, we found more marked results among men. This could in part be related to the small sample size, and in part to different opportunities in educational and professional fields that, for historical and socio-cultural reasons, could have advantaged older men over older women. In this regard, a longitudinal study assessing gender differences and risk factors for depression over time suggested that working activity for men and social participation for women were preventive factors against the development of depressive symptoms. Our findings show that CRI working activity scores were significantly higher in men than in women, and were inversely associated with GDS only in men.
However, this relationship was no longer significant when CRI-Working Activity was considered together with the other CRI sub-scores in the model. As for the relationship between depressive mood and age, we did not find any significant difference in GDS scores between the youngest and the oldest old individuals. This supports the view that, although older inpatients are at greater risk of developing depression, this disease is not a normal part of aging, but rather a pathological situation needing investigation and treatment. Stratifying by age, we observed that a lower CR, in particular CRI-Leisure Time, was significantly associated with a higher GDS only in those aged <85 years. This finding confirms the importance of intervention in the social aspects of daily life in terms of encouraging a healthy and socially active lifestyle. The evidence that this relationship is no longer apparent in the oldest old is in line with what has been observed in cognitive decay. The CR theory, in fact, suggests that in individuals with a high CR cognitive decline has a later onset, and the diagnostic criteria for dementia manifest later, when the disease is more severe [28-30]. In other words, CR is a dynamic compensation system that makes it possible to counteract the brain damage caused by the disease. However, when the disease exceeds a certain degree of severity, CR runs out of compensation capacity and the disease becomes manifest. This does not mean that in the more advanced age groups no lifestyle interventions should be made. On the contrary, they become even more important in this population, who may benefit from a socioenvironmental context that mitigates loneliness and the sense of abandonment.
Limitations
The main limitations of the present study are certainly the small sample size and its cross-sectional observational design. Furthermore, only GDS was used to evaluate depressive symptoms, and only the MMSE to assess cognition.
Conclusion
The present study suggests that improving CR, especially through fostering a socially active lifestyle, may check the development of depressive symptoms in older individuals. The effect could be less marked in the oldest old, probably due to a lower compensation capacity of CR, and in women. In this regard, the observed gender differences highlight the need for further research on the topic in order to devise personalized preventive interventions.
References
- Nair P, Bhanu C, Frost R,et al. A Systematic Review of Older Adults' Attitudes towards Depression and its Treatment. Gerontologist 2019;22.
- Royal College of Psychiatrists. Who care wins. Improving the outcome for older people admitted to the general hospital: Guidelines for the development of Liason Mental Health Services for oldel people. 2005;
- Almeida OP. Prevention of depression in older age. Maturitas 2014;79(2):136-141.
- Almeida OP, Hankey GJ, Yeap BB, et al. A risk table to assist health practitioners assess and prevent the onset of depression in later life. Prev Med 2013;57(6):878-882.
- Ross CE, Mirowsky J. Sex differences in the effect of education on depression: resource multiplication or resource substitution? Soc Sci Med. 2006;63(5):1400-1413.
- Stern Y. Cognitive reserve in ageing and Alzheimer's disease. Lancet Neurol 2012;11(11):1006-1012.
- Kockler, M., & Heun, R. Gender differences of depressive symptoms in depressed and nondepressed elderly persons. International Journal of Geriatric Psychiatry, 2002;17(1), 65-72
- Laske C, Banschbach S, Stransky E, et al. Exercise-induced normalization of decreased BDNF serum concentration in elderly women with remitted major depression. Int J Neuropsychopharmacol 2010;13(5):595-602.
- Abrams LR, Mehta NK. Changes in depressive symptoms over age among older Americans: Differences by gender, race/ethnicity, education, and birth cohort. SSM Popul Health 2019;7:100399.
- Burke, WJ, Roccaforte WH, Wengel, SP. The short form of the Geriatric Depression Scale: a comparison with the 30-item form. Topics in Geriatrics, 1991;4(3), 173-178.
- Evans IEM, Llewellyn DJ, Matthews FE,et al. Social isolation, cognitive reserve, and cognition in older people with depression and anxiety. Aging Ment Health 2018;6:1-10.
- Folstein MF, Folstein SE, McHugh PR. "Mini Mental State": a practical method for grading the cognitive state of patients for the clnician. J Psychiatr Res 1975;12.
- Karlsson B, Johnell K, Sigstrom R,et al. Depression and Depression Treatment in a Population-Based Study of Individuals Over 60 Years Old Without Dementia. Am J Geriatr Psychiatry 2016;24(8):615-623.
- Katz S, Ford AB, Moskowitz RW, et al. Studies of Illness in the Aged. the Index of Adl: a Standardized Measure of Biological and Psychosocial Function. JAMA 1963;21;185:914-919.
- Linn BS, Linn MW, Gurel L. Cumulative illness rating scale. J Am Geriatr Soc 1968;16(5):622-626.
- Parmelee PA, Thuras PD, Katz IR, et al. Validation of the Cumulative Illness Rating Scale in a geriatric residential population. J Am Geriatr Soc. 1995;43(2):130-137.
- Yesavage JA, Brink TL, Rose TL, et al. Development and validation of a geriatric depression screening scale: a preliminary report. J Psychiatr Res. 1982 -1983;17(1):37-49.
- Nucci M, Mapelli D, Mondini S. Cognitive Reserve Index questionnaire (CRIq): a new instrument for measuring cognitive reserve. Aging Clin Exp Res 2012;24(3):218-226.
- Pereira DS, de Queiroz BZ, Miranda AS, et al. Effects of physical exercise on plasma levels of brain-derived neurotrophic factor and depressive symptoms in elderly women--a randomized clinical trial. Arch Phys Med Rehabil 2013;94(8):1443-1450.
- Pfeiffer E. A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients. J Am Geriatr Soc 1975;23(10):433-441.
- Baltes, P. B., & Smith, J. New frontiers in the future of aging: From successful aging of the young old to the dilemmas of the fourth age. Gerontology, 49(2), 123-135.
- Meeks TW, Vahia IV, Lavretsky H,et al. A tune in "a minor" can "b major": a review of epidemiology, illness course, and public health implications of subthreshold depression in older adults. J Affect Disord 2011;129(1-3):126-142.
- Pilotto A, Ferrucci L, Franceschi M, et al. Development and validation of a multidimensional prognostic index for one-year mortality from comprehensive geriatric assessment in hospitalized older patients. Rejuvenation Res. 2008;11(1):151-161.
- Rodda J, Walker Z, Carter J. Depression in older adults. BMJ 2011;28:343:d5219.
- Lawhorne L. Depression in the older adult. Prim Care 2005;32(3):777-792.
- Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist Autumn 1969;9(3):179-186.
- Stern Y. Cognitive reserve. Neuropsychologia 2009;47(10):2015-2028.
- Vaughan L, Corbin AL, Goveas JS. Depression and frailty in later life: a systematic review. Clin Interv Aging 2015;10:1947-1958.
- Wu YT, Teale J, Matthews FE, et al. Lifestyle factors, cognitive reserve, and cognitive function: results frome the Cognitive Function and Ageing Study Wales, a population-based cohort. Lancet 2016.
- Yang Y. Is old age depressing? Growth trajectories and cohort variations in late-life depression. J Health Soc Behav 2007;48(1):16-32.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref