Case Report - Journal of Clinical Ophthalmology (2022) Volume 6, Issue 5
Rapid and sustained resolution of dysthyroid optic neuropathy with teprotumumab monotherapy
Michael L. Fasano*, Matthew D. Kay
Larkin Eye Institute, 7031 SW 62nd Ave, South Miami, FL 33143, United States
- Corresponding Author:
- Dr. Michael Fasano Larkin Eye Institute South Miami FL 33143 United States E-mail: Mfasano.research@gmail.com
Received: 02-Sep-2022, Manuscript No AACOVS-22-76494; Editor assigned: 08-Sep-2022, PreQC No. AACOVS-22-76494 (PQ); Reviewed: 22-Sep-2022, QC No. AACOVS-22-76494; Revised: 30-Sep-2022, Manuscript No. AACOVS-22-76494 (R); Published: 07-Oct-2022, DOI: 10.35841/aacovs.6.2.577-579.
Citation: Fasano ML, Kay MD.. Rapid and sustained resolution of dysthyroid optic neuropathy with teprotumumab monotherapy. J Clin Ophthalmol 2022;6(5):577-579.
Abstract
Purpose: We describe the first case of a patient who had rapid and sustained resolution of severe dysthyroid optic neuropathy with teprotumumab therapy without prior medical, surgical, or radiotherapeutic intervention.
Observations: An 83 year-old-man with no prior history of thyroid disease and an ocular history of strabismus surgery as a child presented with progressive visual loss, diplopia, and eyelid swelling. Visual field testing demonstrated double arcuate scotomata denser inferiorly in the left eye (OS) and peripheral depression in the right eye (OD). He was offered treatment options of intravenous steroids, high dose oral steroids, or surgery but refused given concern for side effects. Teprotumumab was initiated with close observation, and he demonstrated improvement of his field defect after one infusion and resolution after three infusions of teprotumumab. He has since completed his 8 dose, 24 week course of teprotumumab and his visual acuity and optic nerve function remain stable.
Conclusion: Teprotumumab may be a safe and effective sole therapeutic agent for rapid and sustained resolution of dysthyroid optic neuropathy. Further studies may be warranted.
Keywords
Dysthyroid optic neuropathy, Thyroid eye disease, Teprotumumab.
Introduction
Vision threatening Thyroid Eye Disease (TED) occurs in the setting of Dysthyroid Optic Neuropathy (DON). Enlargement of intraorbital muscle and fat can compress the optic nerve leading to decreases in visual acuity and fields. Therefore, treatments for DON need to decompress the orbit quickly and efficiently via medical, radiotherapeutic, and/or surgical intervention. Historically, intravenous steroids and orbital decompression have been the mainstay medical and surgical management for DON, respectively.
However, high doses of corticosteroids increase risk of cardiac disease, liver failure, and hyperglycemia. Surgical decompression increases orbital volume thereby reducing pressure on the optic nerve. However, surgery presents its own risks of bleeding, infection, and worsening of diplopia.
Teprotumumab is a human monoclonal IGF-1 receptor antibody that has been demonstrated to effectively treat TED [1]. The drug is dosed every 3 weeks for 8 weeks, starting at 10 mg/kg for the first dose and increased to 20 mg/kg for the subsequent seven doses. The side effects include but are not limited to muscle spasms, hyperglycemia, hearing loss, diarrhea, dry skin and hair loss. Patients with DON were excluded from the trials as it was felt that more emergent intervention was indicated. However, recent reports describe teprotumumab’s success as a treatment for DON [2-4]. At the time of this writing, we herein describe what we believe to be the first case of severe DON that resolved with teprotumumab monotherapy without antecedent steroids, radiation, or surgery. Another case of DON responded to sole teprotumumab therapy has been reported [2].
Case Presentation
We describe a case of an 83-year-old male with no history of dysthyroidism and an ocular history significant for strabismus surgery as a child with longstanding exotropia was referred for neuro-ophthalmologic consultation after a four-month history of progressive visual loss accompanied by bilateral lid swelling, horizontal diplopia, and bilateral eye pain. On presentation, his acuity was 20/25 OD and 20/40 OS with a left afferent pupillary defect. Color vision was 7/8 OD and 2/8 OS.
Exophthalmometry was 19 mm OD and 18 OS at base 98. The optic nerves were sharp and pink with cup-to-disc ratios of 0.3 OD and 0.2 OS. Humphrey visual field demonstrated mild inferior peripheral depression in the right eye and a dense nearly complete inferior altitudinal defect and a moderate superior arcuate scotoma in the left eye (Figure 1A).
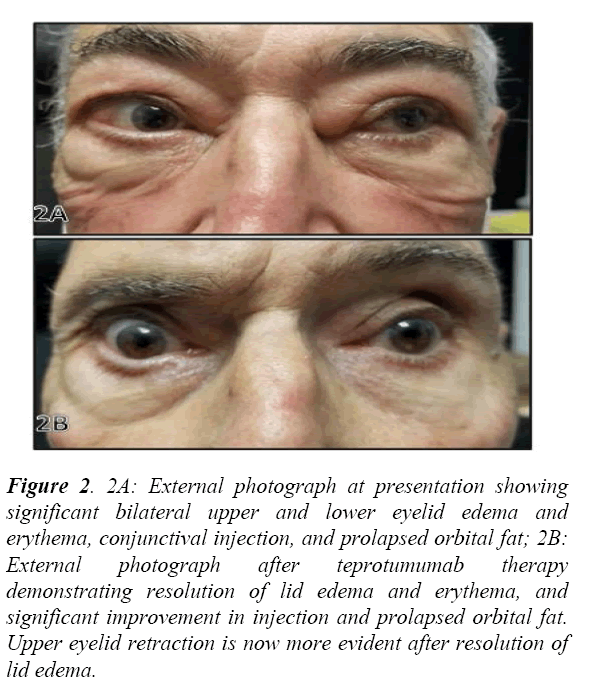
Thyroid eye disease was suspected based on his presentation (Figure 2A), and thyroid function tests and imaging studies were obtained. His MRI Orbits revealed bilaterally symmetric marked thickening and enlargement of the extraocular muscles consistent with TED. He was found to be hyperthyroid with a significantly suppressed TSH and elevated free T3 and free T4. His Thyroid Stimulating Immunoglobulin was elevated to 410 (normal <140% of baseline), and Thyrotropin Receptor Antibody was elevated to 34.37 (normal <2.00 IU/L) diagnostic of Disease. Due to the severity of his visual field defects and decline in acuity attributed to DON, the decision was made to initiate therapy. The patient was offered steroids or surgical decompression but refused both. Therefore, after long discussion, teprotumumab therapy with close serial examinations was initiated. The patient understood the need for surgical decompression of the orbit if persistent or progressive visual loss was noted.
Figure 2: 2A: External photograph at presentation showing significant bilateral upper and lower eyelid edema and erythema, conjunctival injection, and prolapsed orbital fat; 2B: External photograph after teprotumumab therapy demonstrating resolution of lid edema and erythema, and significant improvement in injection and prolapsed orbital fat. Upper eyelid retraction is now more evident after resolution of lid edema.
After his first infusion of teprotumumab subjective visual improvement was noted (Figure 1B). His visual field on the left dramatically improved from a dense inferior arcuate scotoma to mild circumferential peripheral depression. After his third infusion the visual fields normalized OU, his afferent pupillary defect resolved, his color vision was restored, and best corrected visual acuity increased to 20/30. Optic nerve function has remained stable through a complete course of eight infusions. He demonstrates persistent ocular misalignment without diplopia secondary to his history of congenital strabismus in addition to mild lid retraction (Figure 2B).
Discussion
Only a single prior reported case employed teprotumumab as the sole therapeutic agent for DON without utilizing steroids, surgery, and/or radiation therapy prior to initiation of teprotumumab for DON [2]. Contrary to our patient, that case had a mild field defect without other signs of significant optic nerve dysfunction whereas our patient had severe DON including dyschromatopsia, dense double arcuate scotoma, and an afferent pupillary defect. Like the findings in other cases of DON treated with teprotumumab, our patient similarly exhibited a swift response to the therapy demonstrating improvement in visual field testing after one infusion and resolution of visual field defects after three infusions.
The literature lacks significant data for teprotumumab as sole therapy for DON. One study investigated patients with dysthyroid optic neuropathy treated with teprotumumab, however, these patients had failed conventional medical therapy and were not candidates for surgical orbital decompression [3]. A report of two case of DON describes the resolution of optic neuropathy with teprotumumab therapy. These patients had failed prior intravenous corticosteroid therapy with persistent scotomata. They initiated teprotumumab and had complete resolution of pain, lid swelling, and redness after only one infusion with dramatic improvement in diplopia. The second patient did enjoy mild benefit after six weeks of intravenous steroid therapy. However, they had a persistent inferior paracentral hemifield defect. Therefore, teprotumumab was initiated. After two doses they noted complete resolution symptoms and the scotoma on visual field testing [5]. Another case refractory to steroids and radiotherapy describes a similar outcome in which a patient experienced improvement in visual acuity, visual field findings, and afferent pupillary defect after only two infusions of teprotumumab [4]. Interestingly, the patient’s iPhone facial recognition software functioned properly after months of facial incompatibility. These reports of rapid recovery of optic nerve dysfunction are consistent with the data found in the clinical trials of teprotumumab which similarly demonstrate rapid improvement in proptosis, clinical activity score, and visual functioning, although the trials excluded patients with optic nerve dysfunction [1].
Conclusion
As aforementioned, we report the first case of severe DON with an afferent pupillary defect, dense double arcuate scotoma and dyschromatopsia that resolved with teprotumumab monotherapy without previous steroids, radiotherapeutic, or surgical intervention. The patient has remained stable without any signs of optic nerve dysfunction after completion of teprotumumab therapy. Based upon our case and the evolving literature, further studies may be warranted to evaluate teprotumumab’s efficacy as an alternative primary therapy for Dysthyroid Optic Neuropathy (DON) in select cases.
Conflict of Interest Statement
Dr Kay is a paid speaker and consultant for Horizon Therapeutics
References
- Douglas RS, Kahaly GJ, Patel A, et al. Teprotumumab for the treatment of active thyroid eye disease. N Engl J Med. 2020;382(4):341-52.
- Slentz DH, Smith TJ, Kim DS, et al. Teprotumumab for optic neuropathy in thyroid eye disease. JAMA Ophthalmol. 2021;139(2):244-247.
- Sears CM, Wang Y, Bailey LA, et al. Early efficacy of teprotumumab for the treatment of dysthyroid optic neuropathy: A multicenter study. Am J Ophthalmol Case Rep. 2021;23:101111.
- Sears CM, Azad AD, Dosiou C, et al. Teprotumumab for dysthyroid optic neuropathy: early response to therapy. Ophthalmic Plast Reconstr Surg. 2021;37(3S):S157-60.
- Chiou CA, Reshef ER, Freitag SK. Teprotumumab for the treatment of mild compressive optic neuropathy in thyroid eye disease: a report of two cases. Am J Ophthalmol Case Rep. 2021;22:101075.