Mini Review - Journal of Clinical Ophthalmology (2021) Volume 5, Issue 6
Drusen and pigmentary abnormality: Two ways leading to AMD progression.
Shoji Notomi*
Department of Ophthalmology, Faculty of Medical Sciences, Kyushu University, Higashi-ku, Fukuoka, Japan
- Corresponding Author:
- Dr. Shoji Notomi
Department of Ophthalmology,
Kyushu University,
3-1-1 Maidashi,
Higashi-ku Fukuoka, 812-8582,
Japan.
E-mail: noutomi.shouji.926@m.kyushu-u.ac.jp
Accepted date: 17 November, 2021
Citation: Notomi S. Drusen and pigmentary abnormality: Two ways leading to AMD progression. J Clin Ophthalmol. 2021;5(6):482-485.
Abstract
Drusen are known to be the hallmark that predicts progression of Age-related Macular Degeneration (AMD). It is well studied that large drusen over 125 μm, pigmentary abnormality, and medium drusen 63-125 μm provide an increased risk of AMD progression toward late AMD, namely neovascular AMD and geographic atrophy, especially in Western populations. There are several racial and ethnic differences well recognized in AMD. The prevalences of drusen are lower in Asia than those in Western countries. In contrast, neovascular AMD is more frequently seen rather than atrophic dry AMD in Asians. Here we review the current understanding and epidemiology of drusen and other retinal/choroidal features predicting their progression to late AMD and also summarize our recent study. Thick choroid may be a unique feature of part of AMD, which is often found in Asians. To be noted, pigmentary abnormality rather than soft drusen was more associated with “pachychoroid”, which is accompanied by the dilated choroidal vessels and their hyperpermeability, leading to the formation of PCV, a special type of macular neovascularization commonly seen in Asians.
Keywords
Age-related macular degeneration, Retinal pigment epithelium, Pachychoroid, Choroidal neovascularization.
Introduction
Age-related Macular Degeneration (AMD) is the leading cause of visual impairment affecting individuals over the age of 50 worldwide. It is well known that the existence of macular drusen indicates an increased risk for progression to late AMD, neovascular AMD (nAMD) and geographic atrophy [1,2]. Choroidal Neovascularization (CNV) occurs as a creation of new vessels originating from the choroidal vasculature in nAMD, whereas a progressive loss of retinal tissue including photoreceptors, RPE and choriocapillaris leads to the central vision loss in geographic atrophy. Histologically, drusen were observed as the extracellular amorphous debris accumulating between the basolateral basement membrane of Retinal Pigment Epithelium (RPE) and the underlying choroid [3,4]. Of note, the size and number of drusen has been shown to be predictive factors in AMD progression. The Age-Related Eye Disease Study (AREDS), major clinical trials sponsored by the National Eye Institute, showed that large drusen >125 μm and pigmentary abnormality provide a marked increased risk of 5- year rate of developing advanced AMD [1]. Additionally, further analyses clarified the risk of medium drusen 63-125 μm as well; medium drusen alone, without pigmentary abnormality, provided an increasing risk of progression to late AMD [2,5]. In contrast, drusen <63 μm indicated no clinically relevant increased risk of late AMD so that they were considered as normal aging processes. Hence, medium drusen were defined as early AMD while pigmentary abnormality and/or large drusen were intermediate AMD [2].
Literature Review
Several studies have indicated that there are lower frequencies of drusen in Asians compared with Western populations [6]. Indeed, racial and ethnic differences are well recognized in AMD epidemiology [7]. For instance, the prevalence of nAMD in Asian populations is apparently more frequent than those of Western populations [8,9]. Instead, the atrophic/dry form of AMD, drusen and geographic atrophy, is more often seen in Western populations. Furthermore, Polypoidal Choroidal Vasculopathy (PCV), a special type of CNV in AMD, is known to show a higher frequency in Asians compared with Western populations [9,10]. As mentioned, drusen size and/or pigmentary abnormality constitute the threshold to diagnose AMD stages [1,5,11,12]. However, most of previous studies in Asian cohorts did not refer to medium drusen so that there was limited information on medium drusen and AMD development in Asians [13-15].
Yasuda, et al. reported the 9-year incidence of late AMD in The Hisayama Study, a Japanese population-based cohort study on the residents in Hisayama town [14]. Of 1401 individuals enrolled, 121 with soft drusen and 69 with pigmentary abnormality were observed. Among those, 8 of 121 (6.6%) with soft drusen and 3 of 69 (4.3%) with pigmentary abnormality developed late AMD. In 2014, Sasaki, et al. investigated the incidence of nAMD in the fellow eye of patients with unilateral AMD who visited their hospital. They reported that pigmentary abnormality was associated with the occurrence of PCV, suggesting that pigmentary abnormality may be the early sign of PCV, probably unique to Asian [16]. Later, such pigmentary abnormality was characterized as a part of disease, the so-called pachychoroid.
Central Serous Chorioretinopathy (CSC) and a part of AMD have been considered to be the Pachychoroid Spectrum Disease (PSD), which involves dilated choroidal vessels and their congested circulation. Since “pachy-” means “thick” in greek, the thickness of subfoveal choroid was considered to be one of the hallmarks representing PSD. It is hypothesized that the enlarged vessels and their vascular hyperpermeability imply that abnormal choroidal circulation might cause subsequent RPE changes in PSD [17]. Warrow, et al. first reported nine cases in which pigment epitheliopathy was associated with thick choroid similar to CSC but they lack an appearance of subretinal fluid, thereby termed “Pachychoroid Pigment Epitheliopathy (PPE)” [18]. The idea of a new etiology pachychoroid has further driven a hypothesis that CNV beneath the RPE might be associated with thick choroid, which was termed “Pachychoroid Neovasculopathy (PNV)” [19]. It was previously known that long-standing CSC could be associated with Type 1 (sub-RPE) neovascularization [20]. Hence, it is speculated that chronic RPE changes and longstanding serous pigment epithelium detachment might induce neovascularization beneath the RPE. These observations have led to the concept that PSD involves PPE as well as the development of Type 1 Neovascularization (PNV), ultimately leading to PCV [21].
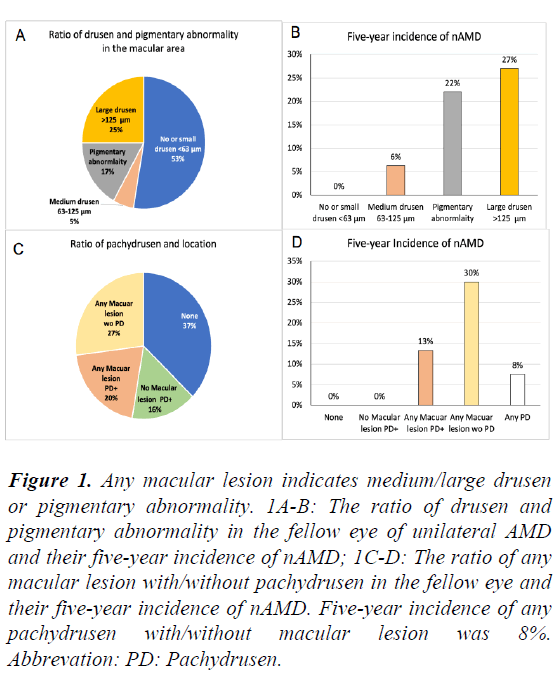
Indeed, Sasaki, et al. have clearly shown that pigmentary abnormality was more likely seen with thick choroid in a Japanese population-based study [22]. Hence, pigmentary abnormality in association with pachychoroid in Asian has been of interests regarding the progression to nAMD. We also investigated 5-year incidence of nAMD in the fellow eye in 296 unilateral AMD patients. In the consecutive retrospective cases, we found 110 eyes with drusen <63 μm, 66 eyes with either medium drusen or pigmentary abnormality, and 74 eyes with large drusen [23]. The other 46 eyes presented no drusen/ pigmentary abnormality in the macular area but were accompanied by pachydrusen outside the macular area. We investigated their fate in AMD progression especially nAMD for 5 years. There were 16 eyes with medium drusen only whereas 50 eyes had pigmentary abnormality without large drusen [23] (Figure 1A). According to the classification by Ferris, et al. in 2013, medium drusen are defined as a sign of “early AMD”, whereas pigmentary abnormality and/or large drusen constitute “intermediate AMD” [12].
Previous studies in Asians described the number of eyes with large drusen but not medium drusen [13-16,24,25]. Recently, the prevalence of medium drusen has been reported to be 136 of 901 (15.1%) in a Japanese cohort (22). In our study, only one eye from 16 eyes with medium drusen developed nAMD (6.3%) while 11 of 50 (22.0%) with pigmentary abnormality presented nAMD within 5 years (Figure 1A). As expected, 20 of 74 (27.0%) eyes with large drusen >125 μm developed nAMD [23] (Figure 1B). Among 20 nAMD-developed cases from large drusen, the proportion of their first eyes’ AMD subtype was 14 typical AMD and 6 RAP, but no PCV. In contrast, about 11 cases that developed nAMD from pigmentary abnormality, in their first eye included 7 typical AMD as well as 4 PCV [23]. Taken together, only large drusen developed RAP and pigment abnormality may become PCV consistent with the previous report [16].
We also compared the subfoveal choroidal thickness based on drusen size/location and pigmentary abnormality. Mean subfoveal choroidal thickness in eyes with pigmentary abnormality was (352 ± 82 μm) much larger than those with drusen <63 μm (276 ± 88 μm) or large drusen (246 ± 96 μm) [23]. Our results were consistent with the population-based study surveyed by Sasaki, et al. that eyes with pigmentary abnormality were more likely accompanied by thick choroid than those having drusen in the macula [22]. Collectively, these results indicated that an accumulation of soft drusen is associated with thin atrophic choroid while RPE degeneration may be accompanied by thick exudative choroid. Such pigmentary abnormality with thick choroid might be considered to be PPE. If so, our data indicates that PPE may proceed to nAMD in Asians.
We also investigated the number of pachydrusen, a newly suggested type of drusen that differ from typical soft drusen on the macula [26]. Unlike typical soft drusen, pachydrusen have an irregular outer contour and show a scattered wide distribution throughout the posterior pole. Of note, pachydrusen are associated with thick choroid and often found especially in Asian eyes [26-28]. It is interesting whether this type of drusen can be associated with the progression of AMD or not. In our study, we found in total 106 eyes with pachydrusen in the fellow eye among 297 eyes. Among 106 eyes, we identified 46 eyes with pachydrusen that did not have any drusen >63 μm or pigmentary abnormality in the macular area [23] (Figure 1C). The mean subfoveal choroidal thickness of those 46 eyes was larger than those without pachydrusen in parallel with previous studies in Korea [24,25].
Importantly, the fate of AMD progression obviously depended on their location. Eyes with pachydrusen did develop nAMD only when they were accompanied by either drusen or pigmentary abnormality in the macula, whereas pachydrusen alone (outside the macular area) did not develop nAMD in our study. Of 60 eyes with pachydrusen with any macular lesion such as medium/large drusen or pigmentary abnormality, 8 (13.3%) developed nAMD [23] (Figure 1D). Of 80 eyes with any macular lesion but not with pachydrusen, nAMD cases had occurred (30.0%) [24]. Since any macular lesion “without pachydrusen” showed a higher incidence of nAMD occurrence than those with both any macular lesion and pachydrusen, pachydrusen themselves did not seem to be a factor positively associated with nAMD occurrence. Our results of pachydrusen were in line with some previous studies although other researchers reported that nAMD incidence of pachydrusen was equivalent to those of soft drusen [25,29-31]. The role of pachydrusen in AMD progression is still elucidative [32].
Figure 1: Any macular lesion indicates medium/large drusen or pigmentary abnormality. 1A-B: The ratio of drusen and pigmentary abnormality in the fellow eye of unilateral AMD and their five-year incidence of nAMD; 1C-D: The ratio of any macular lesion with/without pachydrusen in the fellow eye and their five-year incidence of nAMD. Five-year incidence of any pachydrusen with/without macular lesion was 8%. Abbrevation: PD: Pachydrusen.
Conclusion
In conclusion, pigmentary abnormality, without drusen, may develop nAMD. Such a condition could be associated with pachychoroid pathology. There were limitations in studies in which only subfoveal choroidal thickness was used to determine the pachychoroid. Since choroidal thickness can be affected by age, axial length, etc., comprehensive analyses on the dilated vein structure “pachyvessels” and their vascular permeability will be required to further study the pathology underlying the exudative choroid and the RPE.
References
- Age-related eye disease study research group. The Age-related eye disease study severity scale for age-related macular degeneration: AREDS report no. 17. Arch Ophthal. 2005; 123:1484-98.
- Ferris FL, Davis MD, Clemons TE, et al. A simplified severity scale for age-related macular degeneration: AREDS report No. 18. Arch Ophthalmol. 2005; 123:1570-4.
- Curcio CA, Millican CL. Basal linear deposit and large drusen are specific for early age-related maculopathy. Arch Ophthalmol. 1999; 117:329-39.
- Spaide RF, Curcio CA. Drusen characterization with multimodal imaging. Retina. 2010; 30:1441-54.
- Klein R, Klein BE, Knudtson MD, et al. Fifteen-year cumulative incidence of age-related macular degeneration: The beaver dam eye study. Ophthalmology. 2007; 114:253-62.
- Klein R, Klein BE, Linton KL. Prevalence of age-related maculopathy: The beaver dam eye study. Ophthalmology. 1992; 99:933-43.
- Bressler SB, Munoz B, Solomon SD, et al. Racial differences in the prevalence of age-related macular degeneration: The salisbury eye evaluation project. Arch Ophthalmol. 2008; 126:241-5.
- Klein R, Klein BE, Knudtson MD, et al. Prevalence of age-related macular degeneration in 4 racial/ethnic groups in the multi-ethnic study of atherosclerosis. Ophthalmology. 2006; 113:373-80.
- Coscas G, Yamashiro K, Coscas F, et al. Comparison of exudative age-related macular degeneration subtypes in Japanese and French patients: Multicenter diagnosis with multimodal imaging. Am J Ophthalmol. 2014; 158:309-18.
- Fujiwara K, Yasuda M, Hata J, et al. Prevalence and risk factors for polypoidal choroidal vasculopathy in a general Japanese population: The hisayama study. Semin Ophthalmol. 2018; 33:813-819.
- Age-related eye disease study research group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with Vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001; 119:1417-36.
- Ferris III FL, Wilkinson CP, Bird A, et al. Clinical classification of age-related macular degeneration. Ophthalmology. 2013; 120:844-51.
- Kawasaki R, Wang JJ, Ji GJ, et al. Prevalence and risk factors for age-related macular degeneration in an adult Japanese population: The funagata study. Ophthalmology. 2008; 115:1376-81.
- Yasuda M, Kiyohara Y, Hata Y, et al. Nine-year incidence and risk factors for age-related macular degeneration in a defined Japanese population: The hisayama study. Ophthalmology. 2009; 116:2135-40.
- Nakata I, Yamashiro K, Nakanishi H, et al. Prevalence and characteristics of age-related macular degeneration in the Japanese population: The nagahama study. Am J Ophthalmol. 2013; 156:1002-9.
- Sasaki M, Kawasaki R, Uchida A, et al. Early signs of exudative age-related macular degeneration in Asians. Optom Vis Sci. 2014; 91:849.
- Matsumoto H, Mukai R, Hoshino J, et al. Choroidal congestion mouse model: Could it serve as a pachychoroid model?. Plos one. 2021; 16:e0246115.
- Warrow DJ, Hoang QV, Freund KB. Pachychoroid pigment epitheliopathy. Retina. 2013; 33:1659-72.
- Pang CE, Freund KB. Pachychoroid neovasculopathy. Retina. 2015; 35:1-9.
- Fung AT, Yannuzzi LA, Freund KB. Type 1 (sub-retinal pigment epithelial) neovascularization in central serous chorioretinopathy masquerading as neovascular age-related macular degeneration. Retina. 2012; 32:1829-37.
- Cheung CM, Lee WK, Koizumi H, et al. Pachychoroid disease. Eye. 2019; 33:14-33.
- Sasaki M, Ito Y, Yamasaki T, et al. Association of choroidal thickness with intermediate age-related macular degeneration in a Japanese population. Ophthalmol Retina. 2021; 5:528-35.
- Notomi S, Shiose S, Ishikawa K, et al. Drusen and pigment abnormality predict the development of neovascular age-related macular degeneration in Japanese patients. Plos one. 2021; 16:e0255213.
- Lee J, Choi S, Lee CS, et al. Neovascularization in fellow eye of unilateral neovascular age-related macular degeneration according to different drusen types. Am J Ophthalmol. 2019; 208:103-10.
- Lee J, Kim M, Lee CS, et al. Drusen subtypes and choroidal characteristics in asian eyes with typical neovascular age-related macular degeneration. Retina. 2020; 40:490-98.
- Spaide RF. Disease expression in nonexudative age-related macular degeneration varies with choroidal thickness. Retina. 2018; 38:708-16.
- Cheung CM, Gan A, Yanagi Y, et al. Association between choroidal thickness and drusen subtypes in age-related macular degeneration. Ophthalmol Retina. 2018; 2:1196-205.
- Takahashi A, Hosoda Y, Miyake M, et al. Clinical and genetic characteristics of pachydrusen in eyes with central serous chorioretinopathy and general Japanese individuals. Ophthalmol Retina. 2021; 5:910-17.
- Fukuda Y, Sakurada Y, Yoneyama S, et al. Clinical and genetic characteristics of pachydrusen in patients with exudative age-related macular degeneration. Sci Rep. 2019; 9:1-7.
- Kim KL, Joo K, Park SJ, et al. Progression from intermediate to neovascular age‐related macular degeneration according to drusen subtypes: Bundang AMD cohort study report 3. Acta Ophthalmol. 2021.
- Teo KY, Cheong KX, Ong R, et al. Macular neovascularization in eyes with pachydrusen. Sci Rep. 2021; 11:1-8.
- Baek J, Lee JH, Jung BJ, et al. Morphologic features of large choroidal vessel layer: Age-related macular degeneration, polypoidal choroidal vasculopathy, and central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2018; 256:2309-17.