Research Article - Biomedical Research (2017) Volume 28, Issue 10
Tunicamycin suppresses renal cell carcinoma cells growth and metastasis through regulation of ERK/AKT signaling pathway
Yiping Jia1, Yuan Yang2 and Yan Luo3*
1Department of Ultrasound, No. 4 West China Teaching Hospital, Sichuan University, Chengdu, PR China
2Department of Microbiology, West China School of Preclinical and Forensic Medicine, Sichuan University, Chengdu, PR China
3Department of Ultrasound, West China Hospital, Sichuan University, Chengdu, PR China
- *Corresponding Author:
- Yan Luo
Department of Ultrasound, West China Hospital
Sichuan University, PR China
E-mail: luoyanprof@163.com
Accepted date: March 2, 2017
Abstract
Renal Cell Carcinoma (RCC) is a higher malignant degree tumor in urinary system. Evidences have suggested that tunicamycin can inhibit many human cancer growth and metastasis. The purpose of this study is to investigate the role of tunicamycin in the progression of RCC cells and RCC mouse model. Results showed that tunicamycin significantly suppressed ACHN and RuCa cells growth, migration and invasion in vitro. We demonstrated that tunicamycin treatment arrested cell cycle of ACHN and RuCa cells. In addition, results indicated that tunicamycin treatment induced apoptosis and down-regulated ERK and AKT expression and phosphorylation levels in ACHN and RuCa cells. Mechanism research showed that overexpression of ERK increased AKT expression and phosphorylation levels and canceled Tunicamycin-down-regulated AKT expression and phosphorylation levels in ACHN and RuCa cells. In addition, overexpression of ERK inhibited Tunicamycin-suppressed growth and aggressiveness of ACHN and RuCa cells. Further, in vivo assays showed that Tunicamycin markedly inhibited tumor growth in ACHN-bearing mouse model via increasing apoptosis. In conclusion, these results indicate that tunicamycin could inhibit growth of RCC cells both in vitro and in vivo via ERK/AKT signal pathway, suggesting tunicamycin may be an efficient anti-cancer agent for RCC therapy.
Keywords
RCC, Tunicamycin, Metastasis, Apoptosis, ERK/AKT
Introduction
Renal Cell Carcinoma (RCC) is originated from urinary tubular epithelium and RCC represents 80-90% in human malignant tumors [1,2]. Among urologic neoplasm, the incidence of RCC is the third highest and growing at percent of 6.5 average year perannumpa, and the majority of RCC patients eventually develop metastatic RCC [3,4]. Pathological classification of treatment and prognosis of kidney cancer has important guiding significance for patients with RCC [5,6]. The performance of the RCC is various and mainly presents hematuria, flank pain and swelling for clinical patients [7,8]. RCC is highly aggressive and generally resistant to chemotherapy, radiotherapy, and hormone therapy compared to that many other malignancies [9,10]. Therefore, new therapeutic alternatives and elaboration of molecular mechanism are needed in order to provide more effective treatments of malignancy of RCC.
Tunicamycin is nucleotide antibiotic produced by Streptomyces lysosuperficus. A large numbers of reports have suggested that Tunicamycin can be widely suppressed human cancer cells growth and metastasis [11,12]. Evidences have showed that inhibition of N-linked glycosylation by tunicamycin induces Ecadherin- mediated cell-cell adhesion and inhibits cell proliferation in undifferentiated human colon cancer cells [13]. Huong et al. have showed that proteasome inhibitor-I can enhance tunicamycin-induced chemosensitization of prostate cancer cells through up-regulation of NF-kappaB and CHOP expression [14]. Further, tunicamycin sensitizes human melanoma cells to tumor necrosis factor-related apoptosisinducing ligand-induced apoptosis by up-regulation of TRAILR2 via the unfolded protein response [15]. However, no reports have investigated the therapeutic role of Tunicamycin on RCC in mouse model.
In this study, we analysed the inhibitory effects of Tunicamycin on RCC cells in RCC cells and RCC mouse model. We investigated the potential signal mechanism mediated by Tunicamycin in breast cancer cells. We also identified apoptosis rate of CRC cells were induced by Tunicamycin. Results presented that Tunicamycin may be an efficient anticancer agent for the treatment of RCC via regulation of ERK/AKT signaling pathway.
Materials and Methods
Ethic statement
This study was performed by the recommendations in the Guide for the Care and Use of Laboratory Animals of Sichuan University. All animals’ protocols were experimented in accordance with National Institutes of Health and approved by Committee on the Ethics of Animal Experiments Defence Research.
Cells and reagents
ACHN and RuCa were purchased from American Type Culture Collection (ATCC). All tumor cells were cultured in DMEM (Sigma-Aldrich) medium (Gibco, CA, USA) supplemented with 10% Fetal Serum (FBS) (Invitrogen, CA, USA). All cells were cultured in a 37°C humidified atmosphere of 5 % CO2.
Flow cytometry
Apoptosis of ACHN and RuCa cells were evaluated using Annexin V-fluorescein isothiocyanate (Annexin V-FITC) and Propidium Iodide (PI) apoptosis detection kit (Becton- Dickinson, CA, USA). ACHN and RuCa cells were isolated form Simvastatin- or PBS-treated mice and suspended with Annexin V-FITC and PI according to the manufacturer instructions. Fluorescence was measured with a FACS scan flow cytometry (Becton-Dickinson, San Jose, CA, USA).
MTT assays
ACHN and RuCa cells were transfected by pvector or pAKT and culture in 96-well plates for 48 h. Growing ACHN and RuCa cells were treated by 10 mg/ml Tunicamycin for 48 h. After incubation, 20 μl of MTT (10 mg/ml) in PBS solution was added to each well, the plate was further incubated for 4 h. Most of the medium was removed and 100 μl of DMSO was added into the wells to solubilize the crystals. The OD was measured by a BIO-RAD (ELISA) reader at wavelength of 450 nm.
Endogenous expression of ERK
ACHN and RuCa cells were cultured until 90% confluence and the media was then removed from culture plate. ACHN and RuCa cells were transfected by lentivirus-ERK (pERK) or lentivirus-vector (Control) by using lipofectamine 2000 (Sigma-Aldrich). Stable ERK-overexpressed ACHN and RuCa cells were treated by Tunicamycin (10 mg/ml) for 24 hours to analyse the purpose protein expression levels determined by Western blot.
Cell invasion and migration assays
Sable AKT-overexpressed ACHN and RuCa cells were cultured with Tunicamycin (5 mg/ml) or PBS for 48 hours. Migration and invasion of ACHN and RuCa cells was performed in a 6 well culture plate with chamber inserts (BD Biosciences). For migration assays, 1 × 104/well concentration of the ACHN and RuCa cells were placed into the upper chamber. For invasion assays, ACHN and RuCa cells (1 ×104/ well) were placed into the upper chamber with the Matrigelcoated membrane. Migration and invasion of ACHN and RuCa cells were counted in at least three randomly stain-field microscope every membrane.
Real-time quantitative PCR (RT-qPCR)
Total mRNA was isolated from ACHN and RuCa cells by using RNA Easy Mini Extract Kit (Sigma-Aldrich, Gaithersburg, USA). Expressions of Bcl-2, P53, Caspase-3 and Bad in ACHN and RuCa cells were calculated by using RTqPCR kit with β-actin expression as an endogenous control. All procedures were performed according to the manufacturer’s instructions. All the primers were synthesized by Invitrogen (Invitrogen, Shanghai, China). Relative mRNA expression levels were determined by 2-ΔΔCt. The final results were presented as the n-fold manner compared to β-actin.
Analysis of cells cycle
To analyse the effects of Tunicamycin (10 mg/ml) on the cell cycle stage of ACHN and RuCa cells, flow cytometry was performed. Exponentially, culturing ACHN and RuCa cells or ERK-overexpressed ACHN and RuCa cells were treated with Tunicamycin (10 mg/ml) for 48 h. Cells were washed and trypsinized and rinsed with Phosphate-Buffered Saline (PBS). All cells were fixed in 75% ice-cold ethanol for 5 minutes and then washed with PBS three times. The fixed cells were washed with RNase A (20 μg ml/l, Fermentas) and stained propidium iodide (20 μg ml/l, Sigma-Aldrich) for 10 min at 37°C. The percentages of cells in G0/G1 and S phase were analysed using BD FACS Calibur (Becton Dickinson, NJ, USA).
Western blotting
ACHN and RuCa cells were treated by Tunicamycin (10 mg/ml) and harvested by scraping and lysed in RIPA buffer followed homogenized at 4°C for 10 min. Protein were analysed by SDS-PAGE assays followed transfer membrane. Protein were incubated with rabbit anti-human ERK (1: 500, ab17942, Abcam), pERK (1: 500, ab79483, Abcam), AKT (1: 500, ab64148, Abcam), pAKT (1: 500, ab81283, Abcam) or β- actin (1: 500, ab32572, Abcam) for 12 hours at 4°C. Horseradish peroxidase (HRP)-conjugated secondary antibody (Bio-Rad, Hercules, CA, USA) was used at a 1:5,000 dilution and detected using a Western Blotting Luminol Reagent. The results were visualized by using chemi-luminescence detection system (Amersham Biosciences, Piscataway, NJ).
Animal study
Male Balb/c (8 weeks old, body weight: 30-35 g) mice were purchased from Slack co., LTD (Shanghai, China). Mice were maintained at a 12 h light/dark cycle with free access to diet and water. Experimental mice were implanted ACHN cells (5 × 107) into groin and were divided into two groups (n=50 in each group). Treatments were initiated on day 5 after tumor implantation (diameter: 5-8 mm). Tumor-bearing mice were intravenously injected Tunicamycin (10 mg/kg) as PBS as control. The treatment was continued 10 times once time a day. The tumor volumes were calculated according to previous study [16].
TUNEL analysis
For analysis the apoptosis of myocardial cells, the terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick end labeling (TUNEL) assay (Biotool) were used to detect TUNEL-positive cells. Tumor cells from experimental mice after treated by Tunicamycin or PBS were fixed with 4% paraformaldehyde solution for 60 minutes at 4°C. Subsequently, cells were incubated with equilibration buffer for 30 minutes at 4°C. The tumor sections were then incubated with 50 μl of the reaction mixture at 37°C for 60 minutes and washed 3 times with PBS. The cells nuclei were stained with 4', 6-Diamidino-2-Phenylindole (DAPI) for 60 minutes at 4°C. Finally, tumor tissues images were captured with a ZEISS LSM 510 confocal microscope at 488 nm.
Immunohistochemistry
Tumors from xenografted mice were fixed by using formaldehyde (10%) followed with embed in paraffin. Immunohistochemical staining was performed by an avidinbiotin- peroxidase technique. Paraffin-embedded tissue sections were prepared and epitope retrieval was performed for further analysis. The paraffin sections were subjected with hydrogen peroxide (3%) for 10~15 minutes, which subsequently were blocked by a regular blocking solution for 10~15 minutes 37°C. Finally, the sections were incubated with biotinylated goat anti-human ERK (1: 500, ab17942, Abcam) and AKT (1: 500, ab64148, Abcam), respectively, at 4°C for 12 h. Samples were washed with PBS three times and relative expression of proteins was analysed by chemi-luminescence detection system (Amersham Biosciences, Piscataway, NJ).
Statistical analysis
Results are calculated as mean ± SEM. Significance was established with the SPSS statistical (SPSS, USA) and GraphPad Prism 5 software (GraphPad Software, USA), Pearson's correlation coefficient or a two-tailed Student's t-test. The data are presented as the mean and SEM of three independent experiments. Differences were considered significant at *P<0.05, **P<0.01.
Results
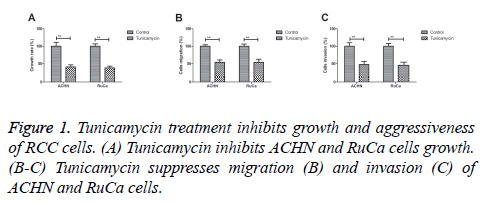
Tunicamycin treatment inhibits growth and aggressiveness of RCC cells
We first investigated the inhibitory effects of Tunicamycin treatment on growth and aggressiveness of RCC cells. As shown in Figure 1A, tunicamycin treatment significantly inhibited ACHN and RuCa cells growth. We found that Tunicamycin treatment inhibited migration and invasion of ACHN and RuCa cells (Figures 1B and 1C). These results suggest that Tunicamycin treatment can inhibit growth and aggressiveness of RCC cells.
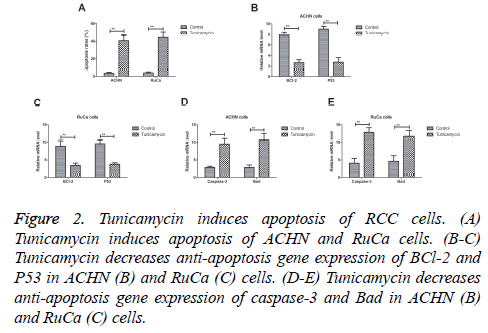
Tunicamycin induced apoptosis of RCC cells
We analysed the efficacy of Tunicamycin on apoptosis of CRC cells in vitro assay. Results demonstrated that Tunicamycin induced apoptosis of ACHN and RuCa cells (Figure 2A). Western blot demonstrated that Tunicamycin decreased antiapoptosis gene expression of BCl-2 and P53 in ACHN and RuCa cells (Figures 2B and 2C). As shown in Figures 2D and 2E, we found that Tunicamycin treatment increased proapoptosis gene expression levels of caspase-3 and Bad levels in ACHN and RuCa cells. These results indicate that tunicamycin can induce apoptosis of RCC cells.
Figure 2: Tunicamycin induces apoptosis of RCC cells. (A) Tunicamycin induces apoptosis of ACHN and RuCa cells. (B-C) Tunicamycin decreases anti-apoptosis gene expression of BCl-2 and P53 in ACHN (B) and RuCa (C) cells. (D-E) Tunicamycin decreases anti-apoptosis gene expression of caspase-3 and Bad in ACHN (B) and RuCa (C) cells.
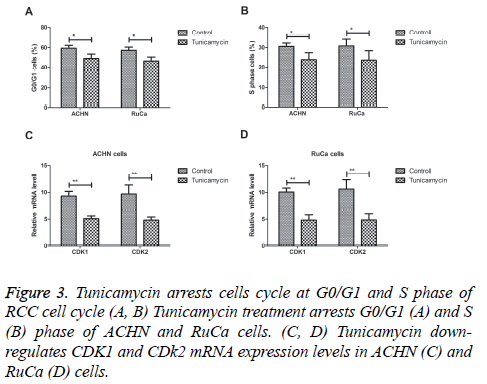
Tunicamycin arrests cells cycle of RCC cell cycle
We next analysed the efficacy of Tunicamycin for cell cycle in vitro. Our results showed that Tunicamycin treatment arrested G0/G1 and S phase of ACHN and RuCa cells (Figures 3A and 3B). Results showed that Tunicamycin down-regulated CDK1 and CDk2 mRNA expression levels in ACHN and RuCa cells (Figures 3C and 3D). These results indicate that Tunicamycin can arrest cells cycle of RCC cell cycle through downregulation of CDK1 and CDk2 expression levels.
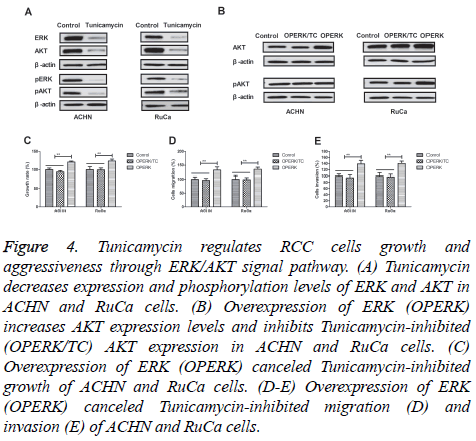
Tunicamycin regulates RCC cells growth and aggressiveness through ERK/AKT signal pathway
In order to analyse the potential mechanism of Tunicamycinmediated growth and aggressiveness in RCC cells, we investigate ERK/AKT signal pathway in RCC cells. We found that Tunicamycin treatment decreased expression and phosphorylation levels of ERK and AKT in ACHN and RuCa cells (Figures 4A and 4B). We observed that overexpression of ERK (OPERK) increased AKT expression levels and inhibited Tunicamycin-inhibited (OPERK/TC) AKT expression in ACHN and RuCa cells (Figure 4C). We found that overexpression of ERK abolished Tunicamycin-inhibited growth, migration and invasion of ACHN and RuCa cells (Figures 4D and 4F). Collectively, these results suggest that tunicamycin can regulate RCC cells growth and aggressiveness through ERK/AKT signal pathway.
Figure 4: Tunicamycin regulates RCC cells growth and aggressiveness through ERK/AKT signal pathway. (A) Tunicamycin decreases expression and phosphorylation levels of ERK and AKT in ACHN and RuCa cells. (B) Overexpression of ERK (OPERK) increases AKT expression levels and inhibits Tunicamycin-inhibited (OPERK/TC) AKT expression in ACHN and RuCa cells. (C) Overexpression of ERK (OPERK) canceled Tunicamycin-inhibited growth of ACHN and RuCa cells. (D-E) Overexpression of ERK (OPERK) canceled Tunicamycin-inhibited migration (D) and invasion (E) of ACHN and RuCa cells.
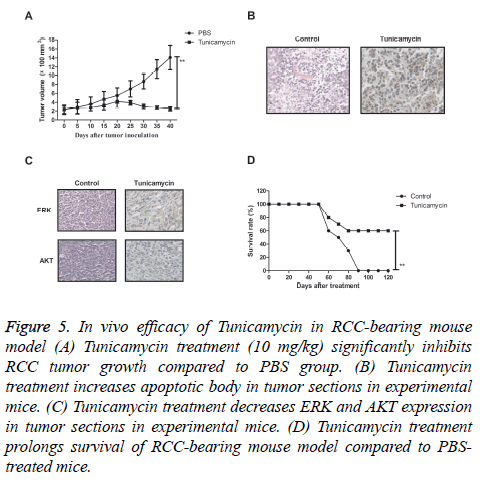
Tunicamycin inhibits tumor in RCC-bearing mouse model
We further the in vivo effects of Tunicamycin on growth in ACHN-bearing mice. As shown in Figure 5A, we found that Tunicamycin treatment (10 mg/kg) significantly inhibited RCC tumor growth compared to PBS group. We observed that Tunicamycin treatment increased apoptotic body in tumor sections in Tunicamycin-treated mice (Figure 5B). Immunohistochemistry assays showed that Tunicamycin treatment decreased ERK and AKT expression levels in tumor tissues compared to PBS-treated tumors (Figure 5C). We also observed that Tunicamycin treatment prolonged survival of RCC-bearing mouse model compared to PBS-treated mice (Figure 5D). These results suggest that Tunicamycin treatment can significantly inhibited RCC growth in vivo.
Figure 5: In vivo efficacy of Tunicamycin in RCC-bearing mouse model (A) Tunicamycin treatment (10 mg/kg) significantly inhibits RCC tumor growth compared to PBS group. (B) Tunicamycin treatment increases apoptotic body in tumor sections in experimental mice. (C) Tunicamycin treatment decreases ERK and AKT expression in tumor sections in experimental mice. (D) Tunicamycin treatment prolongs survival of RCC-bearing mouse model compared to PBStreated mice.
Discussion
The global incidence of RCC is increasing annually and the causes are multifactorial [17]. Though multiple new targets have been identified, and therapeutic agents are in varying stages of development, the targeted therapy remains a promising treatment modality in human cancer [18,19]. Hopefully, the approval of newer agents will translate into more accessible and efficacious options for patients and oncologists. Currently, previous study has reported the efficacy of Tunicamycin for the treatment of human cancer [11,20]. Research also has showed that Tunicamycin presents antitumor efficacy by killing tumor cells growth and enhancing apoptosis of tumor cells [21,22]. Findings have showed that Tunicamycin significantly inhibited growth and aggressiveness of RCC cells. We have found that Tunicamycin treatment induced apoptosis and arrested cell cycle of ACHN and RuCa cells, which contributed to long-term survival rate of RCCbearing mouse model after treatment with Tunicamycin.
Ferraro et al. have indicated that EZH2 is regulated by ERK/AKT and targets integrin alpha2 gene to control epithelial-mesenchymal transition and anoikis in colon cancer cells [23]. Unfolded protein response is required in nu/nu mice microvasculature for treating breast tumor with Tunicamycin, which strongly supported tunicamycin's potential for a powerful glycotherapeutic treatment of breast cancer in the clinic [24]. In this study, we reported the efficacy of Tunicamycin for the treatment of RCC. Findings have indicated that Tunicamycin induced apoptosis of RCC cells via increasing pro-apoptosis gene expression. In addition, blockage of ERK/Akt signaling pathway promoted human breast cancer cells apoptosis and death through reactive oxygen speciesdependent down-regulation [25]. Furthermore, inhibition of prostate cancer proliferation is paralleled by Erk/Akt inactivation and reduced Bcl-2 and bad phosphorylation [26]. Our results showed that Tunicamycin treatment decreased Bcl-2 and bad expression, which led to inhibition of growth of RCC cells through promoting apoptosis.
In conclusion, observed activity warrants further evaluation of the therapeutic effects of Tunicamycin in RCC cells and mouse model. Our present study demonstrates that up-regulation of expression of apoptosis contributes to inhibition of RCC growth, down-regulation of the ERK/AKT signaling pathways, as well as inhibition of migration and invasion, arresting cell cycle in RCC cell.
References
- Nguyen TK, Louie AV. Synchronous oligometastatic non-small cell lung cancer and isolated renal cell carcinoma: a case report and literature review. Cureus 2015; 7: 366.
- Zhang L, Xu B, Wang Y, Liu C, Lu K. Advanced renal cell carcinoma associated with von Hippel-Lindau disease: A case report and review of the literature. Oncol Lett 2015; 10: 1087-1090.
- Brugarolas J. Renal-cell carcinoma--molecular pathways and therapies. N Engl J Med 2007; 356: 185-187.
- Ather MH, Nazim SM. Impact of Charlsons comorbidity index on overall survival following tumor nephrectomy for renal cell carcinoma. Int Urol Nephrol 2010; 42: 299-303.
- Smaletz O. Current management and future directions in the treatment of advanced renal cell carcinoma-a Latin American perspective: 10 years in review. International Braz J Urol 2015; 41: 835-843.
- Li C, Dong H, Fu W, Qi M, Han B. Thyroid-like follicular carcinoma of the kidney and papillary renal cell carcinoma with thyroid-like feature: comparison of two cases and literature review. Ann Clin Lab Sci 2015; 45: 707-712.
- Mucciardi G, Gali A, DAmico C, Muscara G, Barresi V, Magno C. Transitional cell carcinoma of the renal pelvis with synchronous ipsilateral papillary renal cell carcinoma: case report and review. Urol Case Rep 2015; 3: 93-95.
- Kong Y, Wang J, Li H, Guo P, Xu JF, Feng HL. Pathological clavicular fracture as first presentation of renal cell carcinoma: a case report and literature review. Cancer Biol Med 2015; 12: 409-412.
- Iacovelli R, Alesini D, Palazzo A. Targeted therapies and complete responses in first line treatment of metastatic renal cell carcinoma. A meta-analysis of published trials. Cancer Treat Rev 2014; 40: 271-275.
- Iacovelli R, Palazzo A, Trenta P. Management of metastatic renal cell carcinoma progressed after sunitinib or another antiangiogenic treatment. Am J Clin Oncol 2014; 37: 611-615.
- Ling YH, Li T, Perez-Soler R, Haigentz M. Activation of ER stress and inhibition of EGFR N-glycosylation by tunicamycin enhances susceptibility of human non-small cell lung cancer cells to erlotinib. Cancer Chemother Pharmacol 2009; 64: 539-548.
- Shiraishi T, Yoshida T, Nakata S. Tunicamycin enhances tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in human prostate cancer cells. Cancer Res 2005; 65: 6364-6370.
- de Freitas JC, Silva Bdu R, de Souza WF, de Araujo WM, Abdelhay ES, Morgado-Diaz JA. Inhibition of N-linked glycosylation by tunicamycin induces E-cadherin-mediated cell-cell adhesion and inhibits cell proliferation in undifferentiated human colon cancer cells. Cancer Chemother Pharmacol 2011; 68: 227-238.
- Huong PT, Moon DO, Kim SO. Proteasome inhibitor-I enhances tunicamycin-induced chemosensitization of prostate cancer cells through regulation of NF-kappaB and CHOP expression. Cell Signal 2011; 23: 857-865.
- Jiang CC, Chen LH, Gillespie S. Tunicamycin sensitizes human melanoma cells to tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis by up-regulation of TRAIL-R2 via the unfolded protein response. Cancer Res 2007; 67: 5880-5888.
- Bai FL, Yu YH, Tian H. Genetically engineered Newcastle disease virus expressing interleukin-2 and TNF-related apoptosis-inducing ligand for cancer therapy. Cancer Biol Ther 2014; 15: 1226-1238.
- Petejova N, Martinek A. Renal cell carcinoma: Review of etiology, pathophysiology and risk factors. Biomedical papers of the Medical Faculty of the University Palacky, Olomouc, Czechoslovakia 160: 183-194, 2016.
- Zhang GS. New targets for molecular therapy of acute leukemia: a single-hit or multiple-hit strategy against signaling pathway. Zhonghua Yi Xue Za Zhi 2005; 85: 437-438.
- Gahrton G. New therapeutic targets in multiple myeloma. Lancet 2004; 364: 1648-1649.
- Han X, Zhang X, Li H, Huang S, Zhang S. Tunicamycin enhances the antitumor activity of trastuzumab on breast cancer in vitro and in vivo. Oncotarget 2015; 6: 38912-38925.
- Giordano E, Davalos A, Nicod N, Visioli F. Hydroxytyrosol attenuates tunicamycin-induced endoplasmic reticulum stress in human hepatocarcinoma cells. Mol Nutr Food Res 2014; 58: 954-962.
- Carlisle RE, Brimble E, Werner KE, Cruz GL, Ask K. 4-Phenylbutyrate inhibits tunicamycin-induced acute kidney injury via CHOP/GADD153 repression. PLoS One 2014; 9: 84663.
- Ferraro A, Mourtzoukou D, Kosmidou V. EZH2 is regulated by ERK/AKT and targets integrin alpha2 gene to control epithelial-mesenchymal transition and anoikis in colon cancer cells. Int J Biochem Cell Biol 2013; 45: 243-254.
- Banerjee A, Lang JY, Hung MC. Unfolded protein response is required in nu/nu mice microvasculature for treating breast tumor with tunicamycin. J Biol Chem 2011; 286: 29127-29138.
- Kim TH, Woo JS, Kim YK, Kim KH. Silibinin induces cell death through reactive oxygen species-dependent downregulation of notch-1/ERK/Akt signaling in human breast cancer cells. J Pharmacol Exp Ther 2014; 349: 268-278.
- Caraglia M, Marra M, Leonetti C. R115777 (Zarnestra)/Zoledronic acid (Zometa) cooperation on inhibition of prostate cancer proliferation is paralleled by Erk/Akt inactivation and reduced Bcl-2 and bad phosphorylation. J Cell Physiol 2007; 211: 533-543.