Research Article - International Journal of Respiratory Medicine (2017) International Journal of Respiratory Medicine (Special Issue 1-2017)
Screening score to identify people prone to respiratory tract infections in the community.
- *Corresponding Author:
- Alexandra Nieters
Center for Chronic Immunodeficiency, Medical Center - University of Freiburg, Germany.
Tel: +49 761 270 78150
E-mail: alexandra.nieters@uniklinik-freiburg.de
Accepted date: July 19, 2017
Abstract
Background: Respiratory tract infections (RTIs) are a major cause of morbidity in society. A proportion of the general population suffers more often from recurrent and severe RTI compared to the vast majority. We aim to identify those in the population at risk for recurrent RTI with the help of a questionnaire-derived score. Method: A population-based study, termed the Airway Infection Susceptibility (AWIS) study, was conducted between December 2011 and March 2013 in South Baden, Germany. From a total of 69, 695 individuals approached via mail 12, 839 individuals in the age range of 18 to 70 years completed the screening questionnaire. A predefined RTI-score was calculated as a weighted sum of relevant questionnaire items. Regression models were used to assess the association of potential risk factors and co-morbidities with the score. The score was prospectively validated among 1459 individuals who reported their monthly RTI prevalence in three consecutive winterspring seasons using a summary measure. Results: Repeated severe or long lasting RTIs during adulthood were reported by 12% and 18% of the study participants. The RTI-score correlated positively with female sex, younger age, and frequent contact to children, smoking status, and obesity as well as a number of co-morbidities, most strongly asthma and chronic obstructive pulmonary disease. The monthly prevalence of most RTI symptom categories in the subsequent infection seasons approximately doubled between the low to medium and quintupled between the low to high baseline score category. Bronchitis was reported by 2.3%, 11.6%, and 19.4% of individuals in the low, medium, and high baseline category, respectively. Furthermore, 33.7% of the high baseline score group compared to only 6.4% of the low score group reported significant infection related impairments of their daily activities. Conclusion: The RTI-score applied in this study helps to identify adults in the community prone to RTI who might benefit from preventive measures.
Introduction
Respiratory tract infections (RTI) are categorised into upper respiratory tract infections (URTI) such as rhinitis/common cold, tonsillitis, pharyngitis/laryngitis, otitis media and sinusitis, and lower RTI (LRTI) comprising acute bronchitis, acute exacerbations of chronic bronchitis, pneumonia and pleurisy. URTI constitute the most frequent acute respiratory illness experienced by humans with an average of 2-4 yearly episodes [1]. Total indirect costs of non-influenza RTI are estimated to exceed US$100 billion annually in the USA. [2].
Variability between individuals in exposure to infectious agents, infectious dose and pathogen virulence are important drivers for differences in frequency of RTI. On the other hand, environmental factors and lifestyle habits are implicated in differences in infectious disease risk. Cigarette smoking appears to be a major risk factor for respiratory tract and other systemic infections [3]. Individuals with young children report more RTI than individuals without [1], and children in the household are an independent risk factor for community acquired pneumonia (CAP) [4]. Intake of dietary or supplemental vitamins was shown to modify risk for RTI with most convincing data supporting a role for vitamin D [5]. Considerable evidence, including a metaanalysis of prospective studies, underlines a positive association of psychological stress with increased susceptibility to URTI [6].
Individuals who suffer from antibody deficiencies are at greater risk of RTI and are likely to develop more severe outcomes of disease [7]. Immunodeficiencies associated with heightened susceptibility to RTI such as common variable immunodeficiency (CVID) and selective antibody deficiencies are comparatively rare in the general population. However, relative specific antibody deficiencies might contribute to interindividual differences in RTI risk [8,9].
Here we ask whether a questionnaire-based score, the RTI score, can predict individuals who suffer more frequent and severe RTIs in an adult community cohort. Furthermore, we aim to find demographic and co-morbidity correlates with recurrent RTI in the general adult population.
Methods
Screening study
The Center for Chronic Immunodeficiency (CCI) at the University Medical Center Freiburg conducted the Airwayinfection susceptibility (AWIS) study between December 2011 and March 2013 in south-western Germany. Individuals aged between 18 and 70 years and resident in South Baden in the counties of Freiburg, Breisgau/Hochschwarzwald and Loerrach were considered eligible. Names and addresses of 69, 695 randomly selected individuals were provided by the communal data processing agency. All individuals received by regular mail an information letter about the aims of the study and the study procedures, a consent form to participate in the study, and a fourpage questionnaire to complete. Completed questionnaires and letters of consent including consent to be contacted for future investigations related to the topic “proneness to RTI” were returned to the CCI by regular mail. Individuals were contacted only once. Individuals who indicated lack of familiarity with the German language were excluded from further analyses.
The study protocol was approved by community officials and the Ethics Committee of the University of Freiburg (Ref. No. 258/11_120365).
Screening questionnaire
A self-administered questionnaire was developed by the CCI. The questionnaire comprised parameters assessing the burden of RTI during the past two years including the frequency and duration of URTI and LRTI, other indicators of proneness to RTI, history of other severe infections, infection-related hospitalizations, enlargement of the spleen, surgery on immune organs, two self-assessment questions, co-morbidities, demographic variables and a list of selected lifestyle modifiers of RTI risk. A translated version of the full questionnaire is presented in Supplementary information 1.
Definition of the respiratory tract infection (RTI) score
The RTI score is defined as a weighted sum of all items in the questionnaire indicative of a putative higher RTI susceptibility such as the frequency, duration and severity of symptoms of reported RTI, type of upper and lower RTI and selected other variables related to immune competence. Both selection of variables and choice of weights was based on the expertise of the authors (HHP and AN) as studied by Goldacker et al. Immune system assessment questionnaire (ISAQ) - a powerful tool to identify patients at risk for immunodeficiency. Poster (P426) ESID 2010 biannual meeting 2010), [10]. The RTI score varies between 0 (no RTI burden) and 50 (maximal RTI burden). The score was applied to identify a high risk group comprising around 5 to 10% of the general population in accordance with previous estimates [11]. Detailed description of the RTI score is provided in the supplementary methods (Supplementary Information 2).
Prospective validation study
Subsamples of AWIS participants were invited to participate in a sub-cohort study. They were requested to fill-in an additional questionnaire (baseline questionnaire) on lifestyle factors and co-morbidities and to complete monthly diaries registering the monthly occurrence and the duration (< 2 weeks,>2 weeks) of RTIs, namely sinusitis, rhinitis, otitis media, pharyngitis/laryngitis, tonsillitis, influenza-like illness, bronchitis, pneumonia, pleurisy and other acute respiratory infections, from the beginning of November to the end of April of the subsequent year. Furthermore, the intake of antibiotics, doctor visits, hospitalization for RTIs and the impact of RTI symptoms on their daily activities were queried. The diaries were distributed and returned in three months periods: November 2012-January 2013; February 2013-April 2013; November 2013-January 2014; February 2014 -April 2014, November 2014-January 2015; February 2015 -April 2015. This subsampling was stratified by the RTI history score, dividing the patients in the AWIS study into putative low risk patients (≤4, n=5834), medium risk patients (>4 and<17, n=5979) and high risk patients (history score ≥ 17, n=1101). Within these three groups 40.95% (2389/5834), 36.44% (2179/5979) and 94.46% (1040/1101) were invited to participate, 34.7% (829/2389), 34.97% (762/2179) and 46.06% (479/1040) of these accepted the invitation. 84.8%, 70.6% and 80.79% of the latter filled in at least one diary, and among these 91.18%, 91.45% and 84.24% completed infection diaries for at least 4 months.
Statistical analysis
Statistical analysis was performed using STATA (version 14 STATSCorp, USA). Response rates and items’ distribution were described by absolute numbers and percentages. Ordinary least squares regression with the logarithm of the score as outcomes and logistic regression with high/low risk status as outcome was used to assess the association of potential risk factors and co-morbidities with the score. The actual transformation of the score was log(base 2) (score+0.5) such that subjects with a score value of 0 were not lost and regression coefficients of 1.0 can be interpreted as doubling of the score, at least approximately. Adjustment was performed with respect to a set of predefined potential categorical confounders. Results from regression analyses are reported by effect estimates with 95% confidence intervals and p-values. For categorical covariates effects are reported in relation to an appropriately chosen reference category, and p-values refer to a test of the global null hypothesis of no differences among the categories. Subjects with incomplete covariate data were excluded from multivariate analyses. The following variable definitions and categorizations were performed as part of the data analysis: age of participants was calculated as difference of study entry date and birth date (both indicated in the questionnaire) and subdivided in categories (<25, 25 ≤ age <35, 35 ≤ age <45, 45 ≤ age <55, 55 ≤ age<65, and ≥ 65). BMI was categorized as <18.5 (underweight), 18.5 ≤ BMI<25 (normal weight), 25 ≤ BMI <30 (overweight) and ≥ 30 (obese). A distinction between “urban, semi-urban and rural” residence was made based on a classification provided by the Ministry of Transport and Infrastructure of Baden-Württemberg. Altitude and population density refers to the municipality level.
In the prospective validation the average monthly prevalence of RTI symptoms and selected other items listed above were computed for subjects with a low, medium and high RTI score. The prevalence was determined for each month and then averaged over the eighteen months period of the 3 winter seasons considered. For each estimate of prevalence, the nominator was the number of subjects responding to the particular item in the respective month. Confidence intervals were determined by using linear regression for the binary indicators at the patient/ month level with month as categorical covariate and robust standard errors to account for averaging over the month specific estimates, intra-individual clustering and deviation from normality.
Results
Characteristics of screening study population
From 69, 695 questionnaires mailed, 12, 839 were completed and returned to the CCI representing an overall response rate of 18.4%. Response increased with age (p<0.001) and women responded at a higher rate than men (21.2% versus 15.7%, respectively (p<0.001)). The study population included more women (n=7.336) than men (n=5.503) and had a mean age of 46.8 years. Further demographic details are presented in Table 1.
| Characteristics | Category | Number (Percentage %) |
|---|---|---|
| Gender | ||
| Female | 7336 (57.14) | |
| Male | 5503 (42.86) | |
| Age (years) | ||
| <25 | 1144 (8.91) | |
| Â ≥ 25, <35 | 1729 (13.47) | |
| Â ≥ 35, <45 | 2295 (17.88) | |
| Â ≥ 45, <55 | 3615 (28.16) | |
| Â ≥ 55, <65 | 2842 (22.14) | |
| Â ≥ 65 | 1197 (9.32) | |
| Missing | 17 (0.13) | |
| Educational level | ||
| None | 68 (.53) | |
| Volks-Hauptschule1 | 2559 (19.93) | |
| Realschule/Mittlere Reife2 | 3894 (30.33) | |
| Abitur3 | 2742 (21.36) | |
| University degree | 3484 (27.14) | |
| Missing | 92 (0.72) | |
| Smoking status | ||
| None | 7509 (58.49) | |
| (Ex-)Smoker | 5323 (41.46) | |
| Missing | 7 (0.05) | |
| BMI (kg/m2) | ||
| <18.5 | 282 (2.2) | |
| ≥18.5, <25 | 6763 (52.68) | |
| Â ≥ 25, <30 | 4117 (32.07) | |
| Â ≥ 30 | 1611 (12.55) | |
| Missing | 66 (0.51) | |
1Secondary general school, represents 9 years of school education.
2Intermediate secondary school, represents 10 years of school education.
3General Higher Education Entrance Qualification represents 12-13 years of school education.
Table 1: Determination of the presence of total iron by ferrozine method.
Non response at item level was mostly very low, not exceeding 5% for most items except the duration variables (ranging from 2.3% to 12.0%), other allergies (6.2%) and medication affecting the immune system (44.5%).
Supplementary information 3 shows the distribution of questionnaire items that participants have reported at least once in the last two years. Common cold/rhinitis was the most frequently reported RTI (94%), followed by influenzalike illness (75%), pharyngitis/laryngitis (48%), (paranasal) sinusitis (32%), bronchitis (31%) and tonsillitis (17%). A long duration of illness (≥ 4 weeks) was most frequently reported for pneumonia, other acute respiratory diseases followed by pleurisy and bronchitis with relative frequencies between 17% and 26%. Antibiotics’ prescriptions within the last two years were reported by 39% of the participants with 5% of them receiving four or more prescriptions. 12% and 18% of the participants reported more than three severe or long lasting RTIs during adulthood, respectively. All queried severe infections outside the respiratory tract were quite rare. Hospitalization due to infection was reported by 9% of the study population. Every fourth individual regarded him/herself as susceptible to RTIs and about a third reported impairment in daily activities - at least “on and off” – as a consequence of RTI.
Distribution of the RTI score
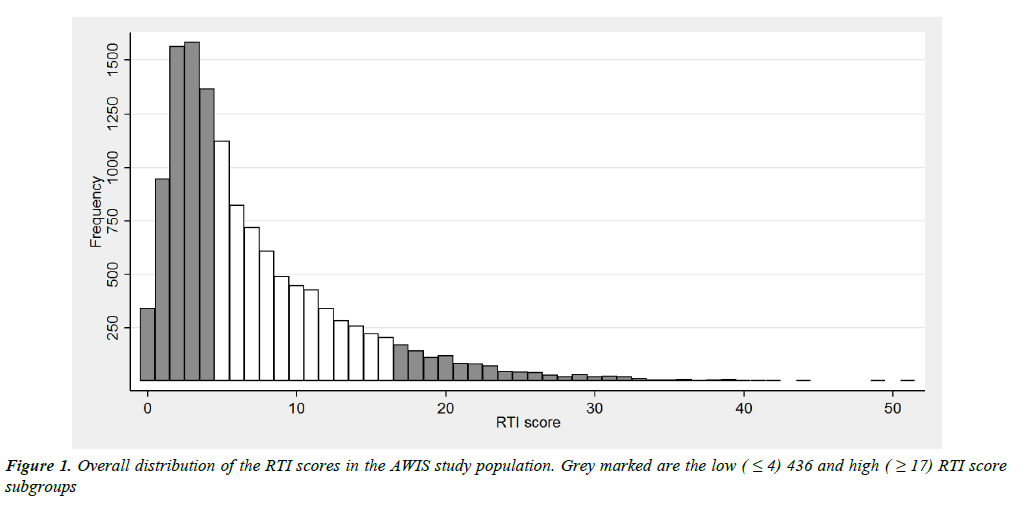
The RTI score has a distinctly right-skewed distribution separating a small fraction of subjects from the main bulk (Figure 1).
According to our aim to identify a high risk group with prevalence between 5% and 10%, we selected a cut point of ≥ 17 resulting in a high risk group representing 8.3% of the study population. Due to the rather poor discrimination among subjects with low scores, we defined a rather large low risk group using a cut point of ≤ 4 comprising 45.2% of the study population. This group is actually characterised by low frequencies for most items, except common cold/rhinitis, influenza-like illness, pharyngitis and laryngitis, whereas prevalence of all items increased markedly in the high score group (Supplementary information 3 and 4).
Association of the RTI score with demographic variables, smoking, and co-morbidities
Our questionnaire queried selected demographic and selected lifestyle variables with possible impact on the overall frequency and severity of RTI. Women and younger individuals had a higher mean RTI score compared to men and older people (meanRTI_ women = 7.51, standard error of the mean (SEM)=0.074; meanRTI__ men = 6.44, SEM = 0.079; meanRTI_s<45 years=7.55, SEM=0.083; mean RTI_ ≥ 45 years = 6.72, SEM=0.072). The direction and magnitude of associations were similar in all models considered (Table 2), demonstrating increased risk of a high RTI score for women, younger people, obese individuals, ever smokers, and study participants with frequent contact to small children. Living in a rural or low populated area, but not at higher or lower altitude, was significantly associated with a tendency to a lower RTI score.
| Items | Category | Number | RTI Score | |||
|---|---|---|---|---|---|---|
| Overall score |  = 4 versus  ≥ 17 | |||||
| Regression coefficient Unadjusted (95% CI) | Regression coefficient Adjusted (95% CI) | OR1 Unadjusted (95% CI) | OR Adjusted2(95% CI) | |||
| Gender | Male | 5505 | 0 | 0 | 1 | 1 |
| Female | 7336 | 0.23 (0.18, 0.27) | 0.26 (0.22, 0.31) | 1.67 (1.46, 1.91) | 1.90 (1.64, 2.19) | |
| P<0.001 | P<0.001 | P<0.001 | P<0.001 | |||
| Age (years) | <25 | 1144 | 0.01 (-0.08, 0.10) | 0.06 (-0.03, 0.15) | 1.08 (0.82, 1.42) | 1.23 (0.92, 1.64) |
| Â ≥ 25, <35 | 1730 | 0 | 0 | 1 | 1 | |
| Â ≥ 35, <45 | 2295 | -0.02 (-0.09, 0.06) | -0.03 (-0.10, 0.05) | 1.01 (0.80, 1.27) | 0.97 (0.76, 1.23) | |
| Â ≥ 45, <55 | 3615 | -0.22 (-0.29, -0.15) | -0.24 (-0.31, -0.17) | 0.69 (0.56, 0.86) | 0.63 (0.50, 0.79) | |
| Â ≥ 55, <65 | 2842 | -0.24 (-0.32, -0.17) | -0.27 (-0.35, -0.20) | 0.80 (0.64, 0.99) | 0.70 (0.56, 0.89) | |
| Â ≥ 65 | 1197 | -0.22 (-0.31, -0.13) | -0.24 (-0.34, -0.15) | 0.80 (0.61, 1.05) | 0.75 (0.56, 0.99) | |
| P<0.001 | P<0.001 | P<0.001 | P<0.001 | |||
| BMI (kg/m2) | <18.5 | 282 | 0.07 (-0.07, 0.22) | -0.03 (-0.18, 0.12) | 1.13 (0.72, 1.78) | 0.95 (0.60, 1.51) |
| Â ≥ 18.5, <25 | 6763 | 0 | 0 | 1 | 1 | |
| Â ≥ 25, <30 | 4117 | 0.06 (0.01, 0.11) | 0.16 (0.11, 0.21) | 1.23 (1.06, 1.42) | 1.45 (1.23, 1.69) | |
| Â ≥ 30 | 1611 | 0.23 (0.16, 0.30) | 0.31 (0.24, 0.38) | 2.06 (1.72, 2.47) | 2.35 (1.94, 2.84) | |
| P<0.001 | P<0.001 | P<0.001 | P<0.001 | |||
| Educational level | None | 68 | 0.01 (-0.29, 0.32) | -0.02 (-0.32, 0.28) | 0.71 (0.28, 1.84) | 0.77 (0.30, 2.03) |
| Volks-Hauptschule3 | 2560 | 0 | 0 | 1 | 1 | |
| Realschule/Mittlere Reife4 | 3894 | 0.03 (-0.03, 0.09) |
-0.03 (-0.09, 0.03) |
0.94 (0.78, 1.12) |
0.89 (0.73, 1.07) |
|
| Abitur5 | 2743 | 0.08 (0.02, 0.15) |
0.02 (-0.05, 0.09) |
0.94 (0.77, 1.14) |
0.95 (0.76, 1.18) |
|
| University degree | 3484 | -0.01 (-0.07, 0.06) |
0.01 (-0.05, 0.08) |
0.80 (0.66, 0.97) |
0.95 (0.78, 1.16) |
|
| p=0.048 | p=0.495 | p=0.196 | p=0.769 | |||
| Smoking status | None | 7510 | 0 | 0 | 1 | 1 |
| (Ex-)Smoker | 5323 | 0.12 (0.07, 0.16) |
0.14 (0.10, 0.18) |
1.48 (1.30, 1.69) |
1.54 (1.34, 1.76) |
|
| P<0.001 | P<0.001 | P<0.001 | P<0.001 | |||
| Contact to children | Never | 1399 | -0.07 (-0.15, 0.00) |
-0.06 (-0.13, 0.01) |
1.01 (0.80, 1.26) |
0.99 (0.79, 1.26) |
| Rarely | 5434 | 0 | 0 | 1 | 1 | |
| Weekly | 2475 | 0.17 (0.11, 0.23) |
0.13 (0.07, 0.19) |
1.49 (1.25, 1.78) |
1.39 (1.16, 1.67) |
|
| Daily | 3490 | 0.33 (0.28, 0.38) |
0.29 (0.23, 0.34) |
1.96 (1.68, 2.30) |
1.88 (1.59, 2.23) |
|
| P<0.001 | P<0.001 | P<0.001 | P<0.001 | |||
| Altitude (meter) | Â = 300 | 8986 | 0 | 0 | 1 | 1 |
| 300-500 | 2445 | -0.02 (-0.07, 0.04) | 0.00 (-0.06, 0.05) | 0.85 (0.71, 1.01) | 0.85 (0.72, 1.02) | |
| >500 | 1410 | 0.00 (-0.07, 0.07) |
0.01 (-0.06, 0.08) |
0.91 (0.73, 1.12) |
0.91 (0.73, 1.13) |
|
| p=0.858 | p=0.962 | p=0.152 | p=0.187 | |||
| Density (habitants/km2) | <200 | 2910 | 0 | 0 | 1 | 1 |
| 200-500 | 3916 | 0.08 (0.02, 0.14) |
0.08 (0.02, 0.13) |
1.24 (1.03, 1.49) |
1.23 (1.02, 1.48) |
|
| 500-1000 | 1995 | 0.05 (-0.02, 0.12) |
0.03 (-0.04, 0.10) |
1.21 (0.98, 1.50) |
1.15 (0.93, 1.44) |
|
| >1000 | 4020 | 0.09 (0.03, 0.15) |
0.08 (0.02, 0.14) |
1.27 (1.06, 1.53) |
1.27 (1.05, 1.54) |
|
| p=0.011 | p=0.029 | p=0.055 | p=0.074 | |||
| Urbanity | Urban | 1195 | 0 | 0 | 1 | 1 |
| Half-urban | 8867 | -0.12 (-0.19, -0.04) |
-0.10 (-0.17, -0.03) |
0.81 (0.66, 1.01) |
0.81 (0.64, 1.01) |
|
| Rural | 2779 | -0.16 (-0.25, -0.08) |
-0.14 (-0.23, -0.06) |
0.75 (0.58, 0.96) |
0.76 (0.58, 0.98) |
|
| p=0.001 | p=0.004 | p=0.071 | p=0.104 | |||
Estimates refer to regression coefficients when using the log(base 2) of the RTI score as outcome and to odds ratios (OR) for the association with the status high RTI score (RTI score equal to or above 17) vs. low score (RTI score equal to or below 4).
1Unadjusted effect estimates as well as effect.
2 Estimates adjusted for age, gender, educational level, BMI and smoking status are presented. Effect estimates with 95% confidence intervalsrefer to an appropriately chosen reference category. P-values refer to a global test of the null hypothesis of no difference between the categories.
3 Secondary general school, represents 9 years of school education.
4Intermediate secondary school, represents 10 years of school education.
5General Higher Education Entrance Qualification represents 12-13 years of school education.
Table 2: Association of demographic and environmental variables with the RTI score.
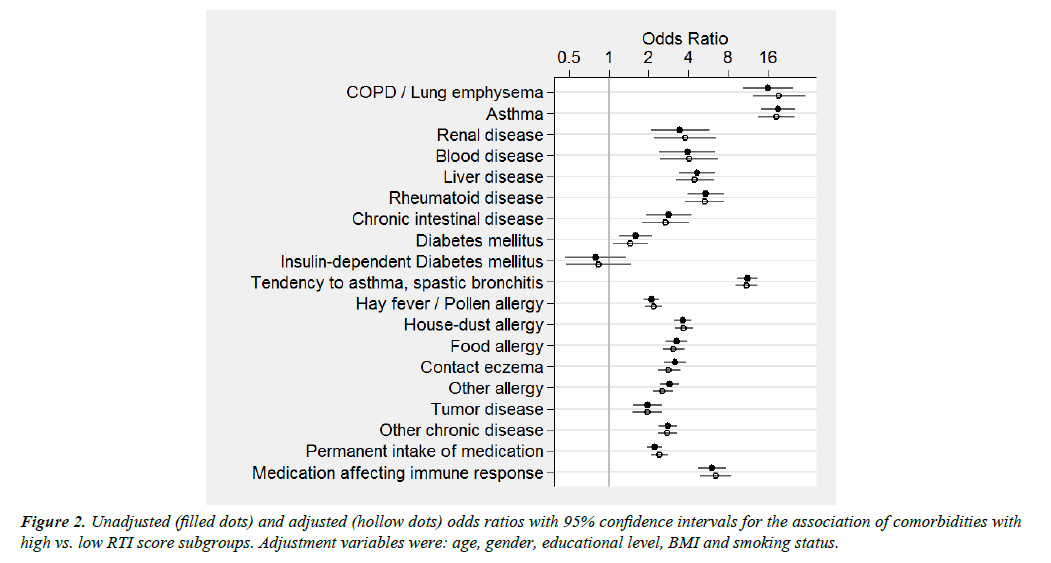
Significant associations (P<0.001) with the RTI score were evident for most co-morbidities queried independent of the type of outcome considered (log-RTI-scores or high vs. low risk) and adjustment for potential confounders (Figure 2 and Supplementary information 5).
The associations of the RTI score with chronic obstructive pulmonary disease (COPD) and asthma were more pronounced than associations with all other individual co-morbidities, whereas a history of diabetes or cancer showed the least marked associations (Figure 2 and Supplementary information 5).
Prospective validation of the RTI score
A subsample of AWIS participants with a low (n=641), medium (n=492), and high (n=326) baseline RTI score completed monthly infection diaries from November 2012 to the end of April 2015. Table 3 describes the average monthly prevalence for all assessed RTI symptoms by baseline RTI score. All queried RTI symptoms as well as RTI related medical consultations and antibiotics intake were reported by a higher number of individuals in the high baseline score group compared to the low score group with the medium score group in between, except for otitis media. Except for common cold and influenza-like illness, the prevalence of all other single symptoms as well as RTI related antibiotics’ use approximately doubled between the low and the medium score category, and quintupled between the low and the high category. More than fivefold more people with a high baseline RTI score reported a moderate to considerable impairment of daily activities in consequence of RTI compared to those with a low baseline RTI score. Overall, in this study population, the baseline RTI score was highly predictive of RTI occurrence in the following three winter-spring seasons.
| Frequency | Low baseline RTI score | Medium baseline RTI score | High baseline RTI score | |||
|---|---|---|---|---|---|---|
| N=641 | N=492 | N=326 | ||||
| Monthly prevalence % | 95%CI | Monthly prevalence % | 95%CI | Monthly prevalence % | 95%CI | |
| Sinusitis | 1.74 | 1.38-2.09 | 9.68 | 6.14-13.22 | 17.79 | 15.45-20.13 |
| Common cold/ Rhinitis | 17.26 | 16.08-18.44 | 35.98 | 31.31-40.65 | 42.42 | 39.28-45.57 |
| Otitis media | 0.45 | 0.23-0.66 | 2.53 | 0.48-4.58 | 2.15 | 1.18-3.12 |
| Pharyngitis / Laryngitis | 4.10 | 3.53-4.67 | 16.82 | 12.85-20.79 | 21.79 | 19.20-24.37 |
| Tonsillitis | 0.76 | 0.46-1.05 | 2.71 | 1.35-4.07 | 3.80 | 2.69-4.92 |
| Influenza-like illness | 6.08 | 5.42-6.73 | 16.22 | 13.20-19.23 | 20.85 | 18.78-22.92 |
| Bronchitis | 2.27 | 1.84-2.71 | 11.59 | 8.22-14.96 | 19.36 | 16.68-22.05 |
| Other acute respiratory infections | 1.07 | 0.79-1.36 | 2.49 | 1.30-3.67 | 4.80 | 3.39-6.22 |
| RTI related physician contact | 3.35 | 2.88-3.81 | 11.72 | 8.50-14.94 | 18.17 | 16.16-20.17 |
| RTI related antibiotics use | 1.25 | 0.98-1.52 | 5.27 | 3.42-7.11 | 8.88 | 7.28-10.48 |
| At least moderate RTI related impairment of daily activities | 6.38 | 5.68-7.08 | 23.78 | 19.46-28.09 | 33.67 | 31.04-36.31 |
Table 3: Average monthly prevalence of respiratory tract infection symptoms, RTI related physician contacts, antibiotics use and impairment of daily activities based on 18 monthly diary reports between 2012 and 2015 (covering for each year the period between November and April)
Discussion
RTI constitute a major morbidity factor and impact tremendously on public health in our society. On the individual level, frequent and severe RTI impair the quality of life in the same order of magnitude as a disease such as depression does [12]. We applied and prospectively validated here a predefined questionnaire-based score for the identification of individuals in the general population at heightened risk for recurrent RTI. This instrument may benefit physicians seeking risk stratification and timely identification of those in need for prophylactic measures such as vaccination programs.
Association with demographic variables, smoking, and co-morbidities
As expected, a high RTI score was positively associated with more frequent contact to children of kindergarten and primary school age as main carriers of respiratory pathogens, in line with previous reports [13]. Correspondingly, older individuals tended to have a lower RTI score than younger individuals in our study population, and women tended to have higher scores than men [14,15]. Our data indicates that the age effect was more prominent in those with little contact to children compared to those with more frequent contacts (Supplementary Information 6).
We observed a trend of higher RTI scores with higher BMI score for both men and for women (Supplementary Information 6). Our findings add to increasing data that suggest an association of higher BMI and obesity with infection incidence and prevalence [16,17]. Obesity is a risk factor for a wide range of nosocomial and community-acquired infections and in particular during the 2009 H1N1 influenza pandemic, obesity was convincingly identified as an independent risk factor for a variety of markers for disease activity [18,19].
Several medical conditions that might hamper the immune response against pathogens were more often reported by individuals with a higher RTI score. Asthma and COPD were by far more prevalent among individuals with frequent and/or severe RTI. Asthmatic patients suffer an increased frequency and severity of RTI, particularly of the lower respiratory tract compared to individuals without asthma [20]. The risk for community-acquired bacterial and viral pneumonia has been estimated to be at least 2-fold among asthmatics compared to healthy individuals [21,22]. The burden of invasive pneumococcal disease attributable to asthma was 17% and 5% within population-based studies conducted in Minnesota [23] and Finland [24], respectively.
Chronic obstructive pulmonary disease (COPD) is a smoking associated lung disorder characterised by progressive irreversible airflow limitation and increased susceptibility to RTI [25]. Microorganisms have long been discussed as risk factors for exacerbations of COPD, but their aetiological role in the evolution of the disease is less clear [26]. Pulmonary colonisation with various airway viruses and bacteria was found in up to 30% of COPD patients with stable disease and in every second patient during an episode of COPD exacerbation [26]. The prominent role of RTI in the pathogenesis of these two chronic diseases, asthma and COPD, is strongly reflected in our data and likely is an indication of reverse causality. Besides these intrinsic factors, environmental factors such as smoking and vitamin D deficiency are discussed as favouring RTI, COPD and asthma alike [3,27]. Smoking (current and ex-) was strongly overrepresented among those with a higher RTI score in our study which is in line with other reports in the literature [3,27]. Evidence suggests that smoking increases the risk of community-acquired pneumonia approximately twofold [28].
Prospective validation of the RTI score
We used the data of a three-year winter/spring assessment of monthly RTI symptoms to explore how well the baseline RTI score predicts the prevalence of RTI, antibiotics use, medical consultation and impairment of daily activities. This analysis supports the ability of the RTI score to identify individuals at greater risk of RTI and RTI-related health impairment. The score we have established here sets itself apart from other instruments addressing respiratory symptoms: first, it relies solely on questionnaire items, rather than findings from physical examination and laboratory tests; second, it encompasses the entire range of respiratory infections; and third, it targets people prone to respiratory infections in the general population rather than in diseased subgroups. Other instruments, such as the Wisconsin Upper Respiratory Symptom Survey (WURSS) and the Jackson scale are limited to the evaluation of the severity of upper respiratory tract infections [29]. The Infection Probability Score (IPS) makes use of common clinical variables such as heart rate, respiratory rate and C reactive protein levels combined with the Sequential Organ Failure Assessment (SOFA) score to predict infection in critically ill patients [30]. The RTI score we propose aims to identify the range of susceptibility in adults and via its questionnaire-only based approach promises great potential for application in general practice and in chronic patient management, where early knowledge of a putative proneness to infections could have a valuable prognostic impact.
Strengths and limitations
This study carries the advantages of a large sample size, a simple, easily comprehensible questionnaire and a prospective validation. The concept of identifying infection-prone individuals in the general population through a questionnaire is novel and worthy of exploration. All components of the score and their weights have been pre-specified and were not data-driven. The value of the RTI score is strengthened by its predictive merit in the study population investigated. It highly predicted new airway infections and infection related health impairments over a timespan of three consecutive winter/spring seasons. A limitation of the RTI score is the need to rely on self-reports of infections, which may be subject to misclassification. However, a preliminary comparison of self-reported and family physician affirmed susceptibility to RTI in 743 study participants revealed a concordance rate of above 95%. A similar comparison for antibiotics use (yes/no) revealed a concordance rate of 93% by Peter H.H (unpublished data). Recall bias due to the differential recall of exposures between affected and unaffected individuals cannot be excluded. The dependence upon a paper questionnaire to calculate the score can be a limiting factor for the practicability, which can be overcome by an online tool which is in preparation on our homepage.
Conclusion
We explored the RTI burden in a large population-based study using a questionnaire-based score. The RTI score employed facilitated identification of putatively infection-prone individuals in the general population and proved predictive of the individual RTI risk. The RTI score we described might help physicians and other health professionals to quickly screen for putatively infection-prone people in the population who could eventually benefit from additional preventive or curative measures. Further external validation of the score shall be in the future focus of our work comprising longitudinal analyses as well as identification of biomarkers, lifestyle and environmental factors that influence one of the major morbidity causes in our society.
Acknowledgements
We would like to thank the study participants and primary care physicians of South Baden supporting the AWIS study. We are grateful to Irene Weber and Anika-Kerstin Biegner for their help in setting up the study and data management and to Melanie Bäurle for her support in patient recruitment.
References
- Monto AS. Studies of the community and family: acute respiratory illness and infection. Epidemiol Rev. 1994; 16: 351-73.
- Birnbaum HG, Morley M, Greenberg PE, et al. Economic burden of respiratory infections in an employed population. Chest.2002; 122: 603-11.
- Stampfli MR, Anderson GP. How cigarette smoke skews immune responses to promote infection, lung disease and cancer. Nat Rev Immunol. 2009; 9: 377-84.
- Schnoor M, Klante T, Beckmann M, et al. Risk factors for community-acquired pneumonia in German adults: the impact of children in the household. Epidemiol Infect.2007; 135: 1389-97.
- Bergman P, Lindh AU, Bjorkhem-Bergman L, et al. Vitamin D and respiratory tract infections: A systematic review and meta-analysis of randomized controlled trials. PloS one.2013; 8: e65835.
- Pedersen A, Zachariae R, Bovbjerg DH. Influence of psychological stress on upper respiratory infection--a meta-analysis of prospective studies. Psychosom Med.2010; 72: 823-32.
- Haq K, McElhaney JE. Ageing and respiratory infections: The airway of ageing. Immunol Lett.2014; 162: 323-28.
- Bossuyt X, Moens L, Van Hoeyveld E, et al. Coexistence of (partial) immune defects and risk of recurrent respiratory infections. Clinchem. 2007; 53:124-30.
- Aghamohammadi A, Moin M, Karimi A, et al. Immunologic evaluation of patients with recurrent ear, nose, and throat infections. Am J Otolaryngol.2008; 29: 385-92.
- Peter H, Goldacker S, Haraldseide J, et al. Construction and clinical validation of a questionnaire-based risk score to identify patients suffering from immunodeficiency or systemic autoimmunity. Br J Med Med Res. 2014; 4: 4751-69.
- Chunara R, Goldstein E, Patterson-Lomba O, et al. Estimating influenza attack rates in the United States using a participatory cohort. Sci Rep.2015; 5:9540.
- Linder JA, Singer DE. Health-related quality of life of adults with upper respiratory tract infections. J Gen Intern Med.2003; 18: 802-07.
- Torres A, Peetermans WE, Viegi G, et al. Risk factors for community-acquired pneumonia in adults in Europe: A literature review. Thorax 2013; 68: 1057-65.
- Falagas ME, Mourtzoukou EG, Vardakas KZ. Sex differences in the incidence and severity of respiratory tract infections. Respir Med.2007; 101: 1845-63.
- Schlagenhauf P, Weld L, Goorhuis A, et al. Travel-associated infection presenting in Europe (2008-12): An analysis of EuroTravNet longitudinal, surveillance data, and evaluation of the effect of the pre-travel consultation. Lancet Infect Dis.2015; 15:55-64.
- Campitelli MA, Rosella LC, Kwong JC. The association between obesity and outpatient visits for acute respiratory infections in Ontario, Canada. Int J Obes (Lond). 2014; 38: 113-19.
- Phung DT, Wang Z, Rutherford S, et al. Body mass index and risk of pneumonia: a systematic review and meta-analysis. Obes Rev. 2013; 14: 839-57.
- Vaillant L, La Ruche G, Tarantola A, et al. Epidemiology of fatal cases associated with pandemic H1N1 influenza 2009. Euro Surveill. 2009; 14: 33.
- Louie JK, Acosta M, Samuel MC, et al. A novel risk factor for a novel virus: obesity and 2009 pandemic influenza A (H1N1). Clin Infect Dis. 2011; 52: 301-12.
- James KM, Peebles RS Jr, Hartert TV. Response to infections in patients with asthma and atopic disease: an epiphenomenon or reflection of host susceptibility? J Allergy Clin Immunol.2012; 130: 343-51.
- Almirall J, Bolibar I, Serra-Prat M, et al.New evidence of risk factors for community-acquired pneumonia: A population-based study. Eur Respir J.2008; 31: 1274-84.
- Teepe J, Grigoryan L, Verheij TJ. Determinants of community-acquired pneumonia in children and young adults in primary care. Eur Respir J.2010; 35: 1113-17.
- Juhn YJ, Kita H, Yawn BP, et al.Increased risk of serious pneumococcal disease in patients with asthma. J Allergy Clin Immunol 2008; 122: 719-23.
- Klemets P, Lyytikainen O, Ruutu P, et al. Risk of invasive pneumococcal infections among working age adults with asthma. Thorax 2010; 65:698-702.
- Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease. N Engl J Med.2008; 359: 2355-65.
- Shaykhiev R, Crystal RG. Innate immunity and chronic obstructive pulmonary disease: a mini-review. Gerontology 2013; 59:481-49.
- Bauer CM, Morissette MC, Stampfli MR. The influence of cigarette smoking on viral infections: translating bench science to impact COPD pathogenesis and acute exacerbations of COPD clinically. Chest 2013; 143:196-206.
- Feldman C, Anderson R. Cigarette smoking and mechanisms of susceptibility to infections of the respiratory tract and other organ systems. J Infect.2013; 67: 169-84.
- Barrett B, Brown R, Voland R, et al. Relations among questionnaire and laboratory measures of rhinovirus infection. Eur Respir J.2006; 28: 358-63.
- Peres Bota D, Melot C, Lopes Ferreira F, et al. Infection Probability Score (IPS): A method to help assess the probability of infection in critically ill patients. Crit Care Med. 2003; 31: 2579-84.