Research Article - Biomedical Research (2017) Volume 28, Issue 16
Research on the influence of miRNA145 on growth and transfer of osteosarcoma cells
Fei Hu1, FaMing Lv2, PingBo Chen2 and Gang Lv2*
1Traditional Chinese Medicine Hospital of Ili Kazakh Autonomous Prefecture, Xinjiang, PR China
2Traditional Chinese Medicine Hospital of the XinJiang Uygyr Autonomous Region, Urumqi, PR China
- *Corresponding Author:
- Gang Lv
Traditional Chinese Medicine Hospital of the XinJiang Uygyr Autonomous Region
PR China
Accepted date: July 27, 2017
Abstract
Purpose: To observe the influence of miRNA145 on the reproduction and transfer capacities of human osteosarcoma cell line MG-63.
Method: The Lipofectamine 2000 lipofection method is adopted to transfect miRNA145 mimic into human osteosarcoma cell line MG-63; at the same time, small RNA fragments with fluorescence are transfected as the contrast for transfection efficiency detection, so as to detect the transfection efficiency with the flow cytometer. The real-time quantitative RT-PCR detection is conducted 24 h after the transfection to detect the miRNA145 expression level in MG-63 cells. The MTT test is conducted to detect the influence of miRNA145 on the reproduction capacity of MG-63 cells. The wound healing test is adopted to detect the influence of miRNA145 on the transfer capacity of MG63 cells.
Results: The Lipofectamine 2000 lipofection method is adopted for transfection of MG-63 cells, with transfection efficiency of more than 90%. The miRNA145 expression level in MG-63 cells is significantly increased 24 h after the transfection of miRNA145 mimic (P<0.05). The transfection of miRNA145 can significantly restrain the reproduction and transfer capabilities of MG-63 cells (P<0.05).
Conclusion: miRNA145 plays a regulating role in the occurrence and development of osteosarcoma by affecting the reproduction and transfer capabilities of osteosarcoma cells.
Keywords
MiRNA145, Osteosarcoma cells, Reproduction, Transfer
Introduction
MicroRNA (miRNA) after transcription plays a role of negative regulating in expression level of genes [1]. miRNA plays important roles in a lot of biological processes, including development, differentiation, apoptosis, metabolism, immunity and tumor progression. More and more evidences indicate that miRNA plays a regulating role in the occurrence and development of tumors, with important roles during transfer and invasion of tumor cells [2-5]. miRNA145 has been verified to have abnormal expressions in multiple tumor tissues, with close relationship to the occurrence and development of multiple tumors [6-8]. However, no definite conclusion on the influence of miRNA145 on growth and transfer capacities of osteosarcoma cells has been drawn so far. Therefore, the Lipofectamine2000 lipofection method is adopted in this research to transfect miRNA145 mimic to human osteosarcoma cell line MG-63 and up-regulate the miRNA145 expression level in human osteosarcoma cell line MG-63, so as to observe the influence of miRNA145 on growth and transfer capacities of human osteosarcoma cell line MG-63. It further discusses on the role of miRNA145 in osteosarcoma progression. The results are reported as follows.
Materials and Methods
The cells and the main reagent of human osteosarcoma cell line MG-63 were purchased from ATCC, and the cells were cultivated in RPMI-1640 culture medium including 10% fetal calf serum. Lipofectamine2000, the transfection reagent, small RNA fragments with fluorescence, DMSO and TRIzol were purchased from U.S. Invitrogen Company; fetal calf serum and RPMI-1640 culture medium were purchased from Gibco Company; SYBR Green PCR reagent kit, miRNA145 and negative control (NC) were purchased from Ambion Company.
Cell transfection
The cells of 70% integration were applied for the transfection, which was operated strictly according to the Specifications for Transfection of Lipofectamine2000.
The fluorogenic quantitative PCR Trizol was adopted to extract the total RNA of cells, which was then conducted with inverse transcription to cDNA. The PCR reaction conditions: 95°C for 5 min, 95°C for 10 sec and 60°C for 20 sec, with totally 40 cycles. The method of 2-ΔΔCt was adopted to count the relative expression quantity of miRNA145. miRNA145 forward primer sequence: 5’-GTCCAGTTTTCCCAGGAAT-3’, reverse primer sequence: 5’-TGGTGTCGTGGAGTCG-3’; internal reference U6 forward primer sequence: 5’- CTCGCTTCGGCAGCACA-3’ and reverse primer sequence: 5’-AACGCTTCACGAATTTGCGT-3’.
MTT cell reproductive capacity detection
After the transfection, the cells were inoculated in a 96-hole culture plate, with further cultivation of 0 d, 1 d, 2 d, 3 d and 4 d. The MTT solution of 20 μL was added in the holes, and the supernatant was extracted after the action of 4 h, and then the DMSO solution of 150 μL was added. After that, the OD value was measured at the wave length of 490 nm after low velocity vibration of 10 min.
Detection on cell transfer capacity
The wound healing test method was adopted to detect the cell transfer capacity, with detailed operating procedures reported in literatures [9].
Statistical method
The SPSS 16.0 statistical software was adopted for data analysis. The t-test was adopted for comparisons between 2 groups of data, and the difference had statistical significance if P<0.05.
Results
Detection on transfection efficiency
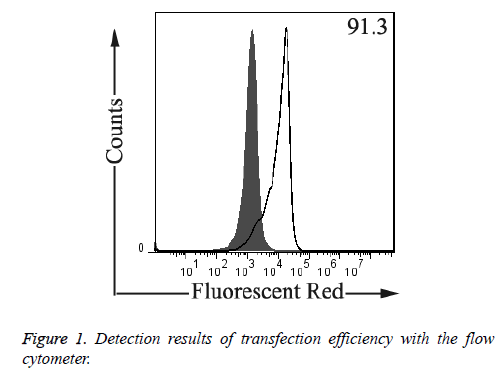
As shown in Figure 1, according to the detection result of the flow cytometer, the transaction efficiency 48 h after the transfection was higher than 90%, which can be applied to future tests.
Detection on the miRNA145 expression in MG-63 after the transfection of miRNA145 mimic
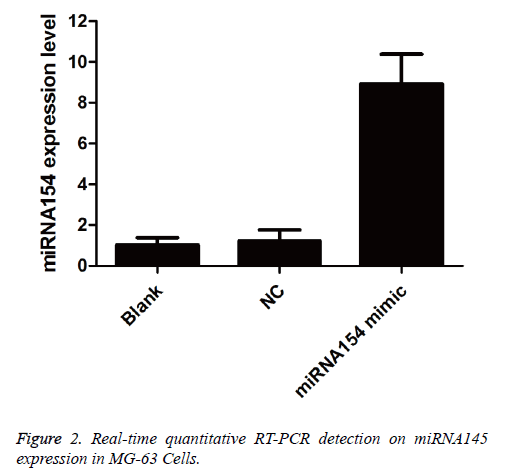
As shown in Figure 2, compared with that of the blank control group (1.04 ± 0.33) and the NC group (1.21 ± 0.56), the miRNA145 expression (8.93 ± 1.45) in MG-63 cells of the transfected miRNA145 mimic group was significantly increased (P<0.05) 48h after the transfection of miRNA145 mimic.
The influence of miRNA145 on reproduction capacity of MG-63 cells
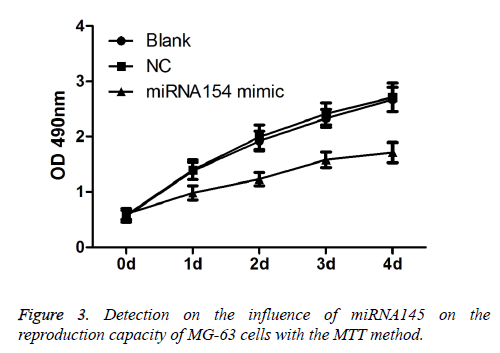
As shown in Figure 3, compared with the blank control group and the NC group, the transfected miRNA145 mimic significantly restrained the reproduction capacity of MG-63 cells (P<0.05).
The influence of miRNA145 on the transfer capacity of MG-63 cells
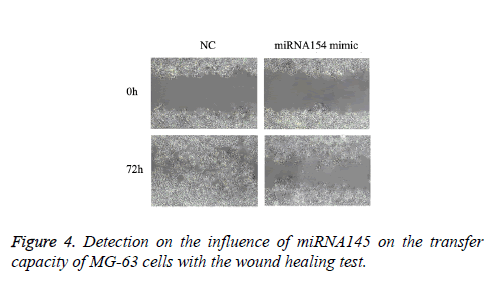
As shown in the results of the wound healing test in Figure 4, compared with the NC group, the transfected miRNA145 mimic significantly restrained the transfer capacity of MG-63 cells (P<0.05).
Discussion
As a kind of small non-coding RNAs, microRNA (miRNA) has the length of about 19-25 nucleotides. According to large numbers of researches in recent years, miRNA plays a very important role in progression of human tumors [10-16]. Therefore, miRN145 is regarded as a new tumor restraining miRNA. Besides, it has been verified by related research findings that the miRN145 expression level is significantly reduced in multiple tumor cells [6-8]. At present, most researches related to the influence of miRN145 on the occurrence and development of osteosarcoma lay emphasis on its influence on the reproduction and apoptosis of osteosarcoma cells. However, little literature is reported on the influence of miRN145 on the transfer of osteosarcoma cells. According to the findings of this research, compared with the blank control group and the NC group, the transfected miRNA145 mimic can significantly restrain the reproduction capacity of MG-63 cells (P<0.05). The result is consistent with the influence of miRNA145 on the reproduction capacity of osteosarcoma cells reported in literature [17]. Based on further research results, besides the influence on the reproduction capacity of osteosarcoma cells, miRNA145 also significantly restrains the transfer capacity of osteosarcoma cells (P<0.05). In conclusion, the role played by miRN145 on the progression of osteosarcoma cells is observed in this research. According to research findings, miRN145 can restrain the growth and transfer capacities of osteosarcoma cells. The research findings in this article provide a new opinion on the progression of osteosarcoma. In addition, the research findings indicate that miRN145 can be utilized as a potential therapeutic target in the treatment of osteosarcoma. However, considering that the biological effect based on miRNAs downstream target gene determines the effect of miRNAs, further researches shall be conducted to figure out the specific target gene for miRNA145 to play the restraining role in the growth and transfer of osteosarcoma cells.
References
- Yanming L, Duo Y, Ning W. Observation on the influence of miRNA-200c on the invasive activity to ovarian carcinoma cells [J]. Chinese J Cancer Prevent Treat 2014; 21: 732-735.
- Li L, Danrong L, Yanping Z. Primary screening on difference on secretory proteins between ovarian carcinoma cells and directional lymphatic metastatic cells of ovarian cancer cells. Chinese J Cancer Prevent Treat 2013; 20: 1053-1056.
- Lin Y, WuJ, Chen H. Cyclin-dependent kinase 4 is a novel target in micoRNA-195-mediated cell cycle arrest in bladder cancer cells. FEBS Lett 2012; 586: 442-447.
- Wang J, Gu Z, Ni P. NF-kappaB P50/P65 heterodimer mediates differential regulation of CD166/ALCAM expression via interaction with micoRNA-9 after serum deprivation, providing evidence for a novel negative auto-regulatory loop. Necleic Acids Res 2011; 39: 6440-6455.
- Suzuki H, Tokino T, Shinomura Y. DNA methylation and cancer pathways in gastrointestinal tumors. Pharmacogenomics 2008; 9: 1917-1928.
- Akao Y, Nakagawa Y, Naoe T. MicroRNAs 143 and 145 are possible common onco-microRNAs in huan cancers. Oncol Rep 2006; 16: 845-850.
- Sachdeva M, Zhu S, Wu F. p53 represses c-Myc through induction of the tumor suppressor miR-145. Proc Natl Acad Sci USA 2009; 106: 3207-3212.
- Chen X, Gong J, Zeng H. MicroRNA145 targets BNIP3 and suppresses prostate cancer progression. Cancer Res 2010; 70: 2728-2738.
- Fu X, Tan D, Hou Z. miR-338-3p is down-regulated by hepatitis b virus x and inhibits cell proliferation by targeting the 3'-UTR region of cyclind1. Int J Mol Sci 2012; 13: 8514-8539.
- Hong L, Han Y, Yang J. MicroRNAs in gastrointestinal cancer: prognostic significance and potential role in chemoresistance. Expert Opin Biol Ther 2014; 14: 1103-1111.
- Joshi HP, Subramanian IV, Schnettler EK. Dynamin 2 along with microRNA-199a reciprocally regulate hypoxia-inducible factors and ovarian cancer metastasis. Proc Natl Acad Sci USA 2014; 111: 5331-5336.
- Zhang Y, Zheng L, Huang J. MiR-124 Radiosensitizes human colorectal cancer cells by targeting PRRX1. PLoS One 2014; 9: e93917.
- Ogata-Kawata H, Izumiya M, Kurioka D. Circulating exosomal microRNAs as biomarkers of colon cancer. PLoS One 2014; 9: e92921.
- Zhang Y, Zheng D, Xiong Y. miR-202 suppresses cell proliferation in human hepatocellular carcinoma by downregulating LRP6 post-transcriptionally. FEBS Lett 2014; 588: 1913-1920.
- Xie K, Liu J, Chen J. Methylation-associated silencing of microRNA-34b in hepatocellular carcinoma cancer. Gene 2014; 543: 101-107.
- Bing L, Hong C, Li-Xin S. MicroRNA-543 suppresses endometrial cancer oncogenicity via targeting FAK and TWIST1 expression. Arch Gynecol Obstet 2014; 290: 533-541.
- Lei F, Zengwu S, Qiang W. The influence of microrna-145 on the reproduction and apoptosis of osteosarcoma cells. China Oncol 2012; 22: 247-251.