Research Paper - Archives of General Internal Medicine (2018) Volume 2, Issue 1
Renal Function Outcome in Hospitalized Adolescent Girls with Anorexia Nervosa.
Mathilde Mignot-Bedetti1,2, Marie Rose Moro1,2 and Corinne Blanchet-Collet1,2*
1Maison de Solenn-Maison des Adolescents-Cochin Hospital, AP-HP, 75014, Paris, France
2CESP, INSERM 1178, Paris-Descartes University, USPC, Paris, France
- *Corresponding Author:
- Corinne Blanchet-Collet
Maison de Solenn-Maison des Adolescents-Cochin
Hospital, AP-HP, 75014, Paris, France
E-mail: corinne.blanchet@aphp.fr
Accepted on January 06, 2018
Citation: Mignot-Bedetti M, Moro MR, Blanchet-Collet C, et al. Renal function outcome in hospitalized adolescent girls with anorexia nervosa. Arch Gen Intern Med. 2018;2(1):1-7.
Abstract
Purpose: Renal complications are classically described in anorexia nervosa (AN). Our aim was to study the initial renal function and its outcome after refeeding in a population of female adolescents hospitalized for AN, and to look for predictive factors of the development of renal failure.
Methods: Renal function was assessed using the Cockcroft and Gault formula before and after the refeeding period in 106 inpatient AN girls in an adolescent medical unit. Predictive factors for renal impairment and potential for recovery were sought using patient anamnestic, anthropometric and biological characteristics.
Results: Creatinine clearance was reduced at admission, and improved significantly at discharge (63.9 mL/min vs. 84.2 mL/min, p<0.01). Only 4 patients presented an initial normal renal function, 58 a mild reduction, and 44 a moderate reduction. At discharge, these figures were respectively 40, 56 and 10. The positive predictive factors identified were BMI (r=0.22; p=0.02), weight gain (r=0.64; p<0.01) and potassium levels at admission (r=0.23, p=0.02). No differences were observed for creatinine clearance levels at admission between restrictive (ANR) and binge/ purging (ANBP) patients, but clearance rates at discharge among ANR were statistically better (87.7 mL/min vs. 75.8 mL/min, p<0.01).
Conclusion: Impairment of renal function in hospitalized anorexic adolescents may be transient or persistent after refeeding depending of initial malnutrition severity and nutritional status at the time of discharge. It seems essential not to under-diagnose the persistence of renal function deficiency following the refeeding phase, in order to limit the risk of severe renal impairment or end-stage renal disease in adulthood.
Keywords
Anorexia nervosa, Adolescents, Renal function, Hypokalemia, Malnutrition, Refeeding.
Introduction
Anorexia Nervosa (AN) is a very serious eating disorder with various somatic complications affecting somato-nutritional, psycho-social and family spheres. Patients may present a restrictive type (ANR) or a binge/purging type (ANBP) defined by DSM5 criteria [1]. The illness generally lasts two to five years with evolving symptoms and diagnostic instability. In 50% of cases, AN crosses over to another eating disorder subtype especially to bulimia or non-specific eating disorders [2]. Recovery rates range from 30 to 80% according to the criteria used [3] [4]. However comorbidities and mortality are far from negligible. In their meta-analysis, Arcelus et al., reported a death rate of 0.51% of patients per year, from suicide in one in five cases [5]. Concerning somatic comorbidities, the renal complications are not well known, under-estimated and under-investigated while they can be dramatic [6]. According to Brotman et al., 70% of the patients with AN will encounter renal complications in the course of their illness, amounting to a prevalence five times greater than in the general population [7]. The study by Zipfel et al., for its part, describes a rate of terminal renal failure necessitating hemodialysis in 5% of patients in a cohort of 84 patients followed for 21 years. This information is all the more worrying because the mean age of these patients at the end of follow-up was only 42 [8]. The main disturbances observed are electrolyte disorders of the hypokalemic type, hyponatremia or magnesium deficiency can also be cited, along with phosphorus deficiency [9-11]. Patients also present renal lithiasis for 5% [12], and nephrocalcinosis [13,14]. Episodes of acute renal failure resulting from hypovolemia or rhadomyolysis (linked to reduced phosphorus and calcium levels and intense hyperactivity) are not uncommon. The state of relative immuno-depression of these patients favors the onset of urine infections [15]. Various studies have explored the repercussions of these different anomalies on renal function. Thus Delanaye et al. reported that 33% of their patients presented a Glomerular Filtration Rate (GFR) below 60 mL/min [16]. In a case-control comparative study, the anorexic patients had a significantly lower GFR than the controls [17]. Fohlin et al. and Russel et al. obtained similar results [18,19]. These impairments do however appear to be reversible with weight gain. Thus 9 out of 10 patients in the study by Boag et al. exhibited a significant decrease in creatinemia and an improvement in clearance rates in the course of refeeding [20]. These results are coherent with those of Russel et al. [19].
There are very little data on renal function in adolescent anorexic populations. The main aim of the present study was therefore to study renal function parameters and their outcome before and after refeeding in a population of inpatients anorexic adolescents. We also sought predictive factors for the onset of renal dysfunction in our population.
Material and Methods
Population
The study was observational, retrospective and cross-sectional and was conducted in a multidisciplinary adolescent unit (Maison de Solenn), Cochin Hospital Paris.
The inclusion criteria were:
- female patient
- age 12 to 19 years
- meeting DSM5 criteria for AN
- hospitalized in the Maison de Solenn Adolescent Unit in Cochin hospital, Paris for refeeding and multidisicplinary care. The discharge weight was determined and individualized with each patient in the pre-admission period by the physician and the adolescent
- hospitalized between January 2005 and June 2013. The exclusion criteria were:
- male patients, since the aim was to obtain a homogenous study population
- female patients for whom the data was incomplete
Methods
Data were collected from patients’ medical files and included parameters relating to patient history (AN subtype that is to say ANR or ANBP, age at onset of the illness, duration of disorder prior to hospitalization, maximum pre-morbid weight) and parameters linked to the hospitalization. These data were collected at admission and at discharge. They concerned clinical elements (weight, stature, BMI, BMI Z-score) and paraclinical data including blood analysis with levels of phosphorus, urea, creatinine, and ultrasound scan of urinary tract if available. Creatinine clearance was calculated using the Cockcroft and Gault formula ((140-âge) x poids (kg) x 1.04) /creatinine (μmol/L), and expressed in mL/min. No other correction factor was used. As commonly accepted, renal function was considered normal when the creatinine clearance was ≥ 90 mL/ min, mild renal insufficiency from 60 to 89 mL/min, moderate insufficiency from 30 to 59 mL/min and severe insufficiency <30 mL/min. Finally, several parameters concerning the hospitalization as such were collected (duration, use of psychotropic medication, enteral feeding by nasogastric tube). Sub-group analyses were also performed, in order to compare patients taking psychotropic treatments with those who did not require them, and patients with solely a restrictive form of the illness with those presenting purging behaviors such as induced vomiting.
Our study was approved by the ethics panel of our institution.
Statistical Analyses
All the results were expressed in means and standard deviation. The significance threshold for p-values was set at 0.05. The Paired Student's t-test enabled the comparison of before-andafter data, Student's t-test was used for sub-group comparisons, and correlations were sought using Spearman's test.
Results
Description of the population
In all, we recovered complete data for 106 files. Among the 117 files corresponding to the selection criteria, 11 were excluded on account of lack of data on discharge, since the hospitalization came to an end as a result of transfer to another department, or the patient left the hospital against medical advice, thus not enabling adequate refeeding or biological investigations at discharge. For the other patients, the mean duration of hospitalization was 21 weeks and 3 days [±14 days].
The mean patients’ age was 16 years and 6 months [±2 years 2 months]. Their illness had lasted for 31 months on average [±25 months], corresponding to a mean age at onset of 14 years [±2 years]. The mean difference between maximum premorbid weight and minimum weight was 15.5 kg [±7 kg].
Among these patients, 82 presented restrictive AN (ANR) and 24 presented bulimic episodes and/or purging behaviors (induced vomiting) (ANBP). None presented potomania, and none reported taking laxatives or diuretics.
Comparison before and after refeeding
The mean admission BMI was 13.5 (+/- 1.27). On average the patients gained 7.2 kg [± 4.29 kg], or a BMI gain of 2.76 points [±1.73 points] corresponding to +1.08 BMI SD [± 0.69 SD] (Table 1). Weight gain was obtained with meals alone or combination of meals with night enteral refeeding using nasogastric tube or exceptionally exclusive enteral nutrition alone for patients with physiological instability. Blood test were performed at admission and discharge and controlled until biological parameters have normalized. The comparison of admission and discharge data shows a significant increase in the levels of potassium, chloride and phosphorous, and a drop in creatinemia indicating an improvement in clearance. At admission, only four patients had normal clearance values, 58 presented mild renal function impairment, 44 presented moderate renal function impairment and no severe renal dysfunction was observed. At discharge, creatinine clearance was normal for 40 patients, 56 still presented mild renal function impairment, and 10 still presented moderate renal function impairment. Thus the mean clearance value for these patients at admission was 63.9 mL/min versus 84.22 mL/min at discharge, which was significantly improved nevertheless corresponding to persistent mild renal failure.
| Admission | Discharge | p | |||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||
| Weight (kg) | 34.4 | 4.6 | 41.6 | 5.1 | < 0.01* |
| BMI (kg/m2) | 13.5 | 1.27 | 16.26 | 1.53 | < 0.01* |
| BMI Z-score (SD) | -2.71 | 0.6 | -1.63 | 0.72 | < 0.01* |
| Natremia (mmol/L) | 141.3 | 2.43 | 141.8 | 1.82 | 0.12 |
| Kalemia (mmol/L) | 3.9 | 0.52 | 4.05 | 0.31 | < 0.01* |
| Chloremia (mmol/L) | 101.4 | 4.52 | 103.8 | 2.5 | < 0.01* |
| Phosphoremia (mmol/L) | 1.26 | 0.21 | 1.4 | 0.17 | < 0.01* |
| Urea (mmol/L) | 5.5 | 1.7 | 5.54 | 1.29 | 0.59 |
| Creatinemia (µmol/L) | 71.5 | 15.24 | 65.56 | 13.06 | < 0.01* |
| Clearance (ml/min) | 63.9 | 12.97 | 84.22 | 19.18 | < 0.01* |
*Significant correlation is set at a p value ≤ 0.05.
Table 1. Anthropometric and biological measures of anorexic patients on admission to the department and on the day of discharge. SD: standard deviation.
At admission, 17 patients underwent ultrasound scan of the kidneys. For 12 of these patients, no anomaly was observed. In one patient, a bilateral aspect of renal cortical notch was observed, in another there was a slight cortical-medullar de-differentiation without dilated pyelocaliceal cavities. Two patients presented dilated pyelocaliceal cavities which subsequently returned to normal for one patient, and in the other patient it was accompanied by lithiasis in one kidney.
Correlations between clinical parameters and creatinine clearance
There was a positive correlation between admission BMI, admission BMI Z-score, and admission clearance values, and also between weight gain and admission clearance. In other words, the higher was the BMI of these patients, the better was their renal function. Likewise, discharge BMI and BMI Z-score were statistically positively correlated with discharge clearance values.
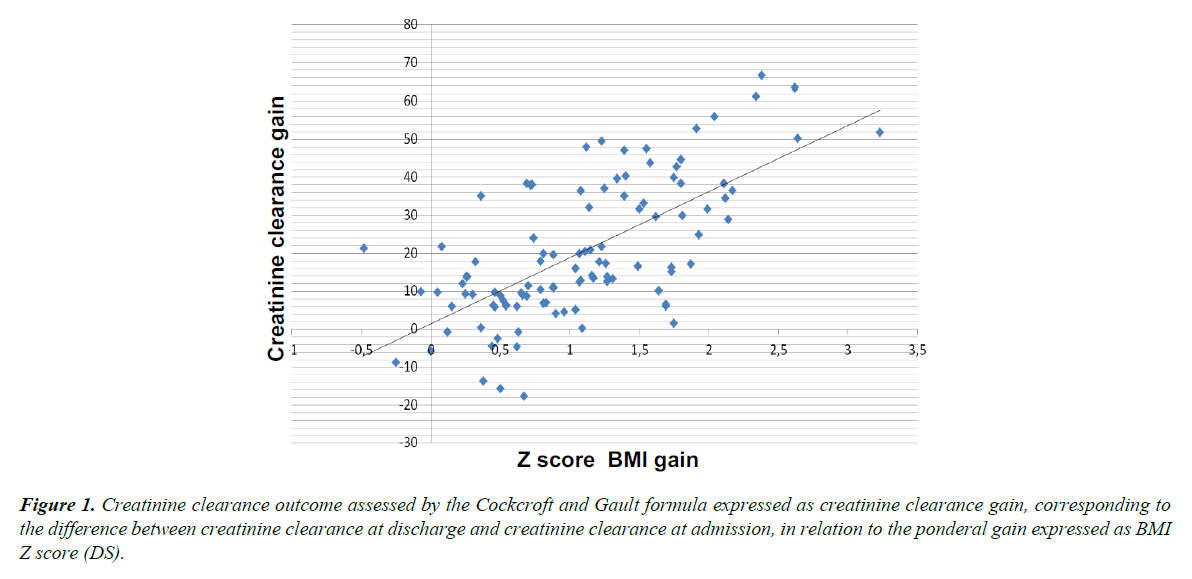
The Spearman r-coefficient linking weight gain, BMI point increase, and BMI Z-score increase to the difference between admission and discharge clearance rates was 0.64 (p<0.01). The more weight a patient gained during hospitalization, the greater the difference between her admission and discharge renal function values. Figure 1 shows the gain in clearance in relation to the increase in the BMI Z-score.
Although no correlation was observed between admission potassium and admission clearance values for these patients, there was however a correlation at discharge (r=0.23; p=0.02) (Table 2).
| Admission | Discharge | |||
|---|---|---|---|---|
| r | p | r | p | |
| Age | -0.07 | 0.5 | -0.36 | < 0.01* |
| Age at onset of AN | 0.08 | 0.44 | -0.12 | 0.21 |
| Duration of AN before admission | -0.07 | 0.5 | -0.26 | < 0.01* |
| Max weight – Min weight | 0.22 | 0.03 | 0.06 | 0.55 |
| BMI at admission | 0.22 | 0.02 | -0.09 | 0.36 |
| Z-score BMI at admission | 0.25 | 0.01 | 0.13 | 0.19 |
| Kalemia at admission | 0.12 | 0.23 | 0.23 | 0.02* |
| BMI at discharge | - | - | 0.51 | < 0.01* |
| Z-score BMI at discharge | - | - | 0.55 | < 0.01* |
| Kalemia at discharge | - | - | -0.06 | 0.54 |
| Duration of hospitalization | - | - | 0.25 | < 0.01* |
| Weight gain | - | - | 0.53 | < 0.01* |
| BMI gain | - | - | 0.51 | < 0.01* |
| Z-score BMI gain | - | - | 0.49 | < 0.01* |
*Significant correlation is set at a p value ≤ 0.05.
Table 2. Data on correlations between various clinical parameters and creatinine clearance values on admission and on discharge. r: Spearman’s correlation coefficient.
Comparison of restrictive and binge/purging anorexic subtypes
Table 3 compares the patients' anthropometric and biological data. The ANR patients had significantly lower maximum and minimum weight at admission, and a lower admission BMI and BMI Z-score. The duration of hospitalization was the same as for the ANBP subtype, but weight gain, mean BMI and BMI Z-score increases were lower than for the ANBP group. Discharge weight and BMI were not statistically different between the two populations.
| ANR (n=82) | ANBP (n=24) | p | |||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||
| Maximum weight (kg) | 46.5 | 8.5 | 52.3 | 10.3 | 0.02* |
| Minimum weight (kg) | 31.8 | 4.2 | 34.3 | 4.3 | 0.02* |
| Weight at admission (kg) | 33.7 | 4.5 | 36.9 | 3.6 | < 0.01* |
| BMI at admission (kg/m2) | 13.3 | 1.3 | 14.2 | 1.1 | < 0.01* |
| Z-score BMI at admission (SD) | -2.8 | 0.6 | -2.5 | 0.6 | 0.04* |
| Kalemia at admission (mmol/L) | 4 | 0.4 | 3.5 | 0.62 | < 0.01* |
| Creatinemia at admission (µmol/L) | 68.8 | 14.3 | 80.5 | 14.8 | < 0.01* |
| Clearance at admission (mL/min) | 64.8 | 13 | 60.7 | 12.5 | 0.17 |
| Weight at discharge (kg) | 41.6 | 5.2 | 41.6 | 4.4 | 0.98 |
| BMI at discharge (kg/m2) | 16.3 | 1.5 | 16 | 1.7 | 0.42 |
| Z-score BMI at discharge (SD) | -1.6 | 0.7 | -1.8 | 0.7 | 0.26 |
| Kalemia at discharge (mmol/L) | 4.1 | 0.3 | 4 | 0.3 | 0.08 |
| Creatinemia at discharge (µmol/L) | 63.6 | 12 | 72.3 | 14.4 | 0.01* |
| Clearance at discharge (mL/min) | 86.7 | 19.7 | 75.8 | 14.5 | <0.01* |
| Weight gain (kg) | 7.9 | 4.1 | 4.7 | 3.8 | < 0.01* |
| BMI gain (kg/m2) | 3 | 1.7 | 1.8 | 1.6 | < 0.01* |
| Z-score BMI gain (SD) | 1.2 | 0.7 | 0.7 | 0.7 | < 0.01* |
*Significant correlation is set at a p value ≤ 0.05.
Table 3. Comparison of the patients' anthropometric and biological data between restrictive type and binge/purging type anorexic patients at admission and at discharge. SD: standard deviation.
Admission potassium values for the patients with ANBP were statistically lower than for ANR patients, although within the normal laboratory range (3.5 mmol/l versus 4 mmol/l, p<0.01). Admission creatinemia was higher, but was not reflected in creatinine clearance, since there was no significant difference between the measures. However, at discharge, patients with the ANBP subtype had statistically higher creatinine levels and lower clearance rates (75.8 mL/ min versus 86.7 mL/min, p<0.01). Potassium and the other parameters of the blood ionogram (with the exception of natremia, significantly higher among the ANBP subtype patients) exhibited no differences.
Comparison of patients with and without psychotropic treatment
The comparisons of medical and biological data are shown in Table 4. The duration of the illness was significantly longer among patients receiving psychotropic medication (35 versus 22 weeks, p=0.01). Patients not receiving psychotropic medication gained significantly more on BMI and BMI Z-score than patients receiving psychotropic medication, despite significantly shorter hospitalizations (17.6 weeks versus 23.5 weeks, p=0.02).
| Pta (n=73) | NPTb (n=33) | p | |||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||
| Age (years) | 16.7 | 2.1 | 16.1 | 2.1 | 0.15 |
| Duration of AN before admission (months) | 35 | 25 | 22 | 21 | 0.01* |
| Duration of hospitalization (weeks) | 23.5 | 15.1 | 17.6 | 9.4 | 0.02* |
| Weight at admission (kg) | 34.6 | 4.8 | 34.1 | 3.9 | 0.6 |
| BMI at admission (kg/m2) | 13.6 | 1.3 | 13.3 | 1.1 | 0.15 |
| Z-score BMI at admission (SD) | -2.7 | 0.6 | -2.8 | 0.6 | 0.53 |
| Kalemia at admission (mmol/L) | 3.9 | 0.51 | 3.9 | 0.54 | 0.51 |
| Creatinemia at admission (µmol/L) | 69.6 | 15.3 | 74.6 | 14.6 | 0.14 |
| Clearance at admission (mL/min) | 65.4 | 13.3 | 60.7 | 11.6 | 0.07 |
| Weight at discharge (kg) | 41.1 | 5.3 | 42.5 | 4.5 | 0.19 |
| BMI at discharge (kg/m2) | 16.1 | 1.5 | 16.5 | 1.6 | 0.24 |
| Z-score BMI at discharge (SD) | -1.7 | 0.64 | -1.5 | 0.82 | 0.12 |
| Kalemia at discharge (mmol/L) | 4 | 0.3 | 4.1 | 0.3 | 0.09 |
| Creatinemia at discharge (µmol/L) | 66.1 | 13.4 | 64.5 | 12.1 | 0.55 |
| Clearance at discharge (mL/min) | 82.4 | 18.5 | 88 | 20 | 0.18 |
| Weight gain (kg) | 6.6 | 4 | 8.4 | 4.5 | 0.06 |
| BMI gain (kg/m2) | 2.5 | 1.6 | 3.2 | 1.9 | 0.05* |
| Z-score BMI gain (SD) | 1 | 0.6 | 1.3 | 0.7 | 0.03* |
*Significant correlation is set at a p value ≤ 0.05.
Table 4. Comparison of the patients' anthropometric and biological data between patients with psychotropic treatment (PT) and with no psychotropic treatment (NPT) at admission and at discharge. SD: standard deviation.
Discussion
This study explored the outcome of renal function before and after partial refeeding among inpatients anorexic adolescent girls. At admission, 96% of patients (102/106) presented impaired renal function whom 43% (44/102) a moderate renal insufficiency. This data is in line with that found in the literature [19,20-24].
In our study, refeeding and nutritional support enabled electrolytic disturbances correction, a significant improvement in nutritional status, and in all anthropometric measures, but not a systematic normalization of corpulence. The patients' renal function improved significantly, with a mean discharge clearance of 84.2 mL/min versus 63.9 mL/min at admission. These results are similar with previous researches. Herzog et al., on a cohort of 84 young anorexic women over a period of 12 years showed an improvement in creatinemia with weight gain [25]. Among the female patients studied by Boag et al., creatinine clearance also increased with nutritional management, the exception being patients who gained no weight [22]. Similar results were reported by Fohlin et al., who measured inulin clearance in 31 female patients [19]. Finally, measures of urinary excretion of creatinine among the patients studied by Russel et al., showed values close to those of the control group after refeeding, and inulin clearance also improved [21].
Thus, some of our patients had transitory acute renal impairment. This condition is most often of functional origin, secondary to hypovolemia [26]. Indeed ten patients have hyperuremia at admission but none was clinically dehydrated. Kidney lithiasis, favored by dehydration, ionic disorders, the consumption of certain toxic substances (one patient in our study), urinary infections (none of our patients at the time of the study, but some had previously had acute pyelonephritis), or inappropriate refeeding syndrome (no patient in our sample), can also be the cause. Finally, hypophosphatemia, hypokalemia, and physical hyperactivity could favor the development of acute transitory renal failure [27,28]. In contrast, other patients appear to evolve towards chronic renal insufficiency which is probably multifactorial [29]. It is estimated that 70% of AN patients are likely to sustain renal function impairment in the course of their illness and 5% terminal kidney failure [9,10]. It appears that calorie deficiency decrease renal function. According to Fohlin, this could be an adaptive mechanism, whereby the fall in GFR could enable an economy in energy expenditure [20]. It is therefore possible to assume that the persistence of a state of thinness could in part explain the maintenance of abnormal renal function for some of our patients. Others aspects could be mentioned such as micronutrients depletion, especially zinc deficiency. In fact zinc contributes to reducing oxydative stress and inflammation, and to inhibiting apoptosis, and plays a part in the preservation of renal function [30]. Infortunately, we had no data about zinc rates because it is not a routine test in our clinical practice; because of the severe malnutrition of our patients we could hypothesize that zinc depletion is an important factor to be evaluated in further studies. Some patients develop nephrocalcinosis as a result of electrolyte disturbances, kidney stones, chronic diarrhea causing tubular obstruction, nephron atrophy or chronic inflammation [15,26]. Finally, according to Li Cavoli et al., calorie deficiency, chronic dehydration, and metabolic disorders could lead to the release of substances deriving from tissue catabolism (myoglobin, LDH, CPK) which could affect the kidneys [29]. The hypothesis of a direct link between malnutrition and creatinine clearance is supported by our correlation results. Indeed, the higher the weight, BMI, and BMI Z-score of our patients the better was their creatinine clearance, at admission and at discharge alike. Thus Fohlin et al., found a correlation between weight loss and GFR [20]. Likewise, there is a positive correlation between the patient's weight and creatinine clearance (measured by 24-hour urine collection) among the patients studied by Russel et al. [21]. Discharge creatinemia in our study was positively correlated with weight and BMI. There was a statistically significant correlation between weight gain, BMI and BMI Z-score, and higher clearance. Boag et al., obtained similar results among their patients hospitalized for refeeding. Weight gain was positively correlated with creatinine clearance [22].
In contrast with the results from other studies, we did not evidence any correlations between patient age, illness duration and creatinine clearance. Takakura et al., measured creatinine clearance from 24-hour urine collection among 45 hospitalized patients, and found a significant negative correlation (r=0.58, p<0.001) between patient age, duration of the illness, and clearance values [30-32]. However, Takakura's patients were older (18 years for restricting patients and 24 years for the ANBP AN subtype), with longer illness duration, in particular for those exhibiting purging behaviors, and the malnutrition was more pronounced. In addition, a large proportion presented chronic hypokalemia which is one of the causes of nephropathies. It can therefore be thought that the longer the illness lasts, the greater the risk of renal function impairment. Hypokalemia causes chronic tubulo-interstitial nephropathy, an inflammatory infiltration of the interstitium and hyperplasia of the juxtaglomerular system [33]. These effects cause irreversible damage to the kidney among young patients. Unlike Takadura et al., we did not evidence any correlation between the potassium levels at admission and admission clearance values. In the study by Takadura, the higher the potassium (upper normal range) the better the creatinine clearance is [32]. However here again the populations are difficult to compare. In addition, the comparison between ANBP and ANR patients corroborates this hypothesis. ANR patients had lower weight, BMI and BMI Z-score than the ANBP group, and they also had more difficulty gaining weight. This could lead to hypothesize that these more undernourished patients sustain greater impairment of renal function. Yet there was no statistically significant difference in admission clearance rates between the two populations. Potassium level among the ANBP subtype patients, although still within the normal laboratory range, was on average lower than that of the restricting patients (3.5 versus 4 mmol/L, p<0.01). At discharge, creatinine clearance was lower among the ANBP patients (75.8 mL/min versus 86.7 mL/min, p<0.01) Thus the recovery potential of the kidneys is worse among ANBP patients, possibly on account of the onset of hypokalemic nephropathies.
The comparison of anthropometric or biological data between patients taking one or several psychotropic drugs and those not taking any evidenced no significant differences between the two groups. The majority of psychotropic drugs is non-toxic for the kidneys, and is even used among patients with terminal chronic renal failure [34]. However, side effects such as electrolyte disorders, inappropriate ADH secretion, acute urine retention and potomania secondary to dryness of the mouth can have an impact on the kidneys. We did not observe any difference in creatinine clearance values between these two treatment groups. This result appears reassuring, but needs to be interpreted cautiously because these patients often require this type of treatment over a number of years, and we do not have data for long enough periods. To our knowledge, no other study has compared these two population subtypes, so that there is no data to back up ours.
Our study presents certain limitations. Because it was retrospective, we chose to use the reference formula proposed by Cockcroft and Gault to estimate renal function in our patients. This formula has not been validated among underweight subjects since creatinemia among these patients is considerably reduced on account of their muscle loss and their insufficient intake of dietary proteins [35]. It is thus possible that GFR values were overestimated [17,36]. Estimating the renal function of these patients is a delicate matter. To our knowledge, a very few studies have used direct evaluation of creatinine clearance. Furthermore, most of them are outdated and concern a very limited number of patients [19,20,32,18]. The reference equations, whether that developed by Cockcroft and Gault, the MDRD equation (Modification of Diet in Renal Disease) or the MAYO are not well suited [37]. Measurement of creatinine clearance via 24-hour urine collection is difficult on account of the risk of poor compliance among these patients with hyper-hydration or hydric restrictions behaviors. The use of inulin values or iodothalamnate method for its part appears too invasive in clinical practice. Finally, the use of Cystatin C (plasma marker of renal function) although promising, has proved disappointing in this population, and is still very costly [18].
Our patients are probably not fully representative of the anorexic population because of the specific vulnerabilities associated with adolescent period. Furthermore, the high degree of malnutrition may induce a potential selection bias. Hospitalization enabled weight gain, but not a return systematic to normal weight. Thus the refeeding was partial, and conclusions cannot be drawn on the recovery of renal function. For this reason, evaluation of renal function in the medium and long term would be valuable. Finally, the ANR and ANBP subgroups included notably different numbers of patients.
On the basis of our review of the literature, this is the largest cohort ever published concerning the evaluation of renal function in adolescent patients with eating disorders. Unlike previous studies, our population was homogenous and concerned female adolescent subjects hospitalized for severe forms of anorexia. This study has enabled the description of renal function in situations of malnutrition. We also evidenced the harmful role of potassium in the lower normal range. The persistence of moderate renal function impairment in some cases despite appropriate refeeding points to the need for subsequent surveillance, particularly among patients presenting regular relapses of their anorexic disorder, and having difficulty reaching normal weight.
Conclusion
According to our results and the literature, anorexic adolescent patients seem to present renal failure that can persist despite a normalization of caloric intake and a diversified food in a context of partial but effective renutrition. The origin of renal function failure is multifactorial, and the exact physiopathology is not well understood. The severity of initial undernourishment and the level of correction of nutritional status could be determining factors in the recovery of renal function. In contrast, in our adolescent population, we found no links between age, illness duration and the severity of renal insufficiency. It seems essential not to under-diagnose the persistence of renal function deficiency following the refeeding phase, in order to limit the risk of severe renal impairment or end-stage renal disease in adulthood. A cohort follow-up study among patients who developed anorexia nervosa in the pre- or peri-pubertal period could provide more support for our results.
Conflict of Interest
The Author declare that there is no conflict of interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorder, Fifth Edition. Feeding and Eating disorder. 2013; pp: 329-53.
- Milos G, Spindler A, Schnyder U, et al. Instabiliity of eating disorder diagnoses: prospective study. Br J Psychiatry. 2005;187:573-8.
- Steinhausen H. The outcome of anorexia nervosa in the 20th century. Am J Psychiatry. 2002;159(8):1284-93.
- Keski-Rahkonen A, Hoek HW, Susser E, et al. Epidemiology and Course of Anorexia Nervosa in the Community. Am J Psychiatry. 2007;164:1259-65.
- Arcelus J, Mitchell AJ, Wales J, et al. Mortality rates in patients with anorexia nervosa and others eating disorders. A meta-analysis of 36 studies. Arch Gen Psychiatry. 2011;68(7):724-31.
- Stheneur C, Bergeron S, Lapeyraque AL. Renal complications in anorexia nervosa. Eat Weight Disord. 2014;19(4):455-60.
- Brotman AW, Stern TA, Brotman DL. Renal disease and dysfunction in two patients with anorexia nervosa. J Clin Psychiatry. 1986;47:433-4.
- Zipfel S, Lowe B, Reas DL, et al. Long-term prognosis in anorexia nervosa: lessons from a 21-years follow-up study. Lancet. 2000;355:721-2.
- Miller KK, Grinspoon SK, Ciampa J, et al. Medical findings in outpatients with anorexia nervosa. Arch Intern Med. 2005;165(5):561-6.
- Kanbur N, Katzman D. Impaired Osmoregulation in Anorexia Nervosa: Review of the literature. Pediatric Endocrinology Review. 2011;8(3):218-21.
- Bouquegneau A, Dubois B, Krzesinski JM, et al. Anorexia Nervosa and the Kidney. Am J Kidney Dis. 2012;60(2):299-307.
- Roux H, Blanchet C, Stheneur C, et al. Somatic outcome among patients hospitalised for anorexia nervosa in adolescence: disorders reported and links with global outcome. Eat Weight Disord. 2013;18(2):175-82.
- Lim A, Hooke D, Kerr P. Anorexia nervosa and senna misuse: nephrocalcinosis digital clubbing and hypertrophic osteoarthropathy. MJA. 2008;188(2):121-2.
- Roberts M, Thorpe C, Mac Gregor D, et al. Severe renal failure and nephrocalcinosis in anorexia nervosa. MJA. 2005;182(12):635-6.
- Winston A. The clinical biochemistry of anorexia nervosa. Ann Clin Biochem. 2012;49:132-43.
- Delanaye P, Cavalier E, Radermecker R, et al. Estimation of GFR by different creatinine and cystatin-C based equations in anorexia nervosa. Clinical Nephrology. 2009;71(5):482-91.
- Aperia A, Broberger O, Fohlin L. Renal function in anorexia nervosa. Acta Paediatr Scand. 1978;67:219-24.
- Fohlin L. Body composition, cardiovascular and renal function in adolescent patients with anorexia nervosa. Acta Paediatr Scand. 1977;268:1-20.
- Russel G, Bruce J. Impaired Water diuresis in Patients with Anorexia Nervosa. American Journal of Medicine. 1966;40:38-48.
- Boag F, Weerakoon J, Ginsburg J, et al. Diminished creatinine clearance in anorexia nervosa: reversal with weight gain. J Clin Pathol. 1985;38:60-3.
- Brion LP, Boeck MA, Gauthier B, et al. Estimation of glomerular filtration rate in anorectic adolescents. Pediatr Nephrol. 1989;3:16-21.
- Palla B, Litt IF. Medical complications of Eating Disorders in Adolescents. Pediatrics. 1988;81:613-23.
- Herzog W, Deter HC, Fiehn W, et al. Medical findings ans predictors of long-term physical outcome in anorexia nervosa: a prospective, 12-year follow-up study. Psychological Medicine. 1997;27(2):269-79.
- Alvin P. Anorexies et boulimies à l’adolescence, 3 ème edition, Douin éditeurs, 2007.
- Walder A, Baumann P. Increased Creatinine Kinane and Rhabdomyolysis in Anorexia Nervosa. Int J Eat Disord. 2008;41:766-7.
- Wada S, Nagase T, Koike Y, et al. A Case of Anorexia Nervosa with Acute Renal Failure Induceed by Rhabdomyolysis; Possible Involvement of Hypophosphatemia or Phosphate Depletion. Internal Medicine. 1992;31:478-82.
- Li Cavoli G, Mule G, Rotolo U. Renal Involvement in Psychological Eating Disorders. Nephron Clin Pract. 2011;119:338-41.
- Tomat AL, Costa Mde, Arranz CT. Zinc restriction during different periods of life : Influence in renal and cardiovascular diseases. Nutrition. 2011;27:392-8.
- Takakura S, Nozaki T, Nomura Y, et al. Factors related to renal dysfunction in patients with anorexia nervosa. Eating Weight Disord. 2006;11:73-7.
- Manzato E, Mazzullo M, Gualandi M, et al. Anorexia nervosa: from purgative behavior to nephropathy. A case report. Cases J. 2009;2:46.
- Cohen LM, Tessier EG, Germain MJ, et al. Update on Psychotic Medication Use in Renal Disease. Psychosomatics. 2004;45:34-48.
- Schneiter S, Berwert L, Bonny O, et al. Les conséquences rénales de l’anorexie. Rev Med Suisse. 2009;5:440-4.
- Hari P, Bagga A, Mahajan P, et al. Effects of malnutrition on serum creatinine and Cystatin C levels. Pediatr Nephrol. 2007;22:1757-61.
- Fabbian F, Pala M, Scanelli G, et al. Estimation of Renal Function in Patients with Eating Disorders. Int J Eat Disord. 2011;44:233-7.