- Biomedical Research (2011) Volume 22, Issue 1
Putative pathogenic effect of oxidative stress in sickle cell disorder
Kalyan Goswami1* and Debes Ray 2
1Kalyan Goswami, Department of Biochemistry, Mahatma Gandhi Institute of Medical Sciences, Sevagram. Wardha, India
2Department of Biochemistry, Kolkata Medical College, Kolkata, India.
- *Corresponding Author:
- Kalyan Goswami
Department of Biochemistry
Mahatma Gandhi Institute of Medical Sciences
Sevagram, Wardha-442102
India
Accepted date: August17 2010
Abstract
Sickle cell disorder is a major hemoglobinopathy associated with hemolytic anemia. It mani-fests either as less severe heterozygous or severe homozygous form. Evidences suggest in-volvement of oxidative stress in this disorder. Hence we made an attempt to find out any pa-thogenic impact of oxidative stress in this disease. From the rural area of central India 80 heterozygous and 20 homozygous cases were registered as two separate groups. Along with this 100 and 66 age and sex matched healthy individuals were included as controls against these two groups of cases respectively. Malondialdehyde and superoxide dismutase activity in serum and catalase activity in erythrocytes were assayed as markers of oxidative stress. Hemoglobin level and reticulocyte count were measured to assess the severity of hemolysis. Statistical comparisons were made between cases and corresponding control levels for all these parameters. Also study was done to find any association between oxidative and hema-tological parameters.The result showed overall oxidative and hemolytic stress in both group of cases. However, except for malondialdehyde which showed significant increase in both the forms of the disease, all other parameters studied showed significant difference only in homozygous but not in heterozygous cases. Also significant association between oxidative parameters and hematological response was recorded only in homozygous cases. Finally li-pid peroxidation was found to be the strongest predictor variable for hemolytic stress re-sponse. Results indicate association of oxidative pathology with the severe form of this dis-order and advocate oxidative damage as the putative pathogenic determinant in this disor-der.
Key words
Sickle cell disorder, oxidative stress, hemolysis, lipid peroxidation.
Introduction
Sickle cell disorder is the most common heritable hemo-globin associated disease [1]. In India, it is more common in the central and southern regions [2]. Due to autosomal recessive inheritance, it may present itself either in milder heterozygous or in severe homozygous form. The pre-dominant pathology of sickling of red blood cells (RBC) occurs due to aberrant polymerization of the mutant de-oxygenated hemoglobin [3]. This process eventually dis-torts the red cells leading to formation of irreversible sick-led cells (ISC). Impaired hemoglobin stabilizing capacity in sickle cell disease and other such mutational hemoglo-binopathies makes RBC vulnerable towards severe oxida-tive stress which overcomes the antioxidant defense [4].
Therefore, this disorder is emerging as a new model of oxidative stress related pathology, which has supposed contribution towards ISC-mediated membrane change leading to hemolytic preponderance [5]. Hemolytic stress response in turn, decreases hemoglobin level along with compensatory reticulocyte proliferation response. Thus it may be surmised that there might be an underlying nexus between oxidative stress and the hematological response in this disorder.
Thus, in the present study parameters of oxidative stress like lipid peroxidation (MDA), super oxide dismutase (SOD) and catalase enzymes and hematological response in terms of hemoglobin level and reticulocyte count were measured to detect any significant difference in both het-ero and homozygous conditions against respective control values. Further analysis was performed for any possible association between such oxidative and hematological parameters to detect evidence of pathogenic significance of oxidative stress in this disorder.
Materials and Methods
This study was carried out in the rural area of Maharash-tra, located in central India. The study populace was se-lected after confirmation of the diagnosis as either het-erozygous (HbAS) or homozygous (HbSS) by hemoglo-bin electrophoresis [6] and all these cases were also ruled out for co-existence of any other hemoglobin disorder. Thus a total of 80 heterozygous [HbAS; median age 23 (range 16-35; 61% male)] and 20 homozygous [HbSS; median age 9.5 (range 7-12); 60% male)] cases were in-cluded in the study. These persons did not receive any medical therapy earlier. Considering the fact that the two case groups differ largely in their observed age range, two corresponding age and sex matched control groups were formed with 100 and 66 individuals for the heterozygous and homozygous cases respectively. People of these con-trol groups were selected from apparently healthy indi-viduals (without any history of major illness and history of blood transfusion) with the same socioeconomic back-grounds residing in the same villages. Any obvious dif-ference in average nutritional status between cases and controls were ruled out by matching for anthropometric values and also by observation of comparable mean level of albumin and total protein level in sera by standard method [data not shown].
About 5ml of blood was collected from antecubital vein of each individual from all these case and control groups after taking informed consent. From each of these blood samples, one half was put in EDTA vial and divided fur-ther into two parts; of which one part was used to perform reticulocyte count and the rest for preparation of the he-molysate for hemoglobin electrophoresis and RBC cata-lase activity assay. Electrophoresis was done on cellulose acetate membrane at alkaline pH. The remaining half of each of the collected blood samples was allowed to co-agulate so as to separate serum for estimation of malon-dialdehyde and SOD enzyme activity.
Estimation of serum malondialdehyde (MDA)
Lipid peroxidation was measured using Thiobarbituric acid (TBA) method [7]. To 0.5 ml of culture supernatant, 2.5ml of 10% TCA and 1 ml of 0.67% TBA were added and boiled in water bath for 30 min. After cooling, resul-tant chromogen was extracted in 4 ml n-butyl alcohol by separation of organic phase with centrifugation at 3000 rpm for 10 min. Absorbance was measured at 530 nm against distilled water. The concentrations of MDA in samples were calculated using standard curve and ex-pressed as nM/ml.
Estimation of serum superoxide dismutase (SOD: EC1.15.1.1.) activity
This method utilizes the inhibitory effect of SOD enzyme on auto-oxidation of pyrogallol [8]. The reaction mixtures were prepared for various concentrations of standards, tests and controls. Constituent of this 3 ml assay mixture was 100 μl each of 1 mM EDTA and 1 mM DTPA in air equilibrated Tris HCl Buffer (50 mM; pH 8.2). To this, 100 μl of standards (with varying concentrations of SOD enzyme) or test sera were added. In control, neither test sample nor standard was added to assay mixture to obtain uninhibited auto oxidation of pyrogallol. Finally, 100 μl volumes of 0.2 mM pyrogallol were added in all vials to start reaction.
Initial 10 seconds period was considered as induction pe-riod of enzyme, after which at every 10 seconds interval for a period of 4 minutes, changes in absorbance at 420 nm was recorded. Average change in the absorbance per minute was calculated and the percentage of inhibition in standards and test samples was calculated using the for-mula.
Percent Inhibition = 100 - (Δ A standard or test per min. × 100)/ Δ A control per min.
The amount of enzyme required to inhibit auto-oxidation of pyrogallol by 50% was defined as one unit of SOD enzyme activity. Accordingly the activity of enzyme in different standard concentrations was expressed in units. Standard graph was constructed with percentage of inhibi-tion versus SOD enzyme activity in described units. The activity of SOD enzyme in each test serum was calculated by using this standard graph.
Estimation of activity of catalase (E. C. I.II. I. 6) in erythrocytes
Catalase was assayed as described earlier [9]. The method utilizes H2O2 splitting activity of catalase in the sample hemolysate. Briefly, 0.1 ml hemolysate was added to a mixture of 2 ml dichromate reagent (5% solution of K2Cr2O7 in glacial acetic acid in the ratio of 1:3) and 0.1 ml of 0.2 M H2O2 with 0.01 M Phosphate buffer (pH 7). After boiling the reaction mixtures for 10 min followed by cooling, the resultant chromogenic product, chromic acetate was measured spectrophotometrically at 570 nm against reagent blank. Activity of catalase was expressed as units/g hemoglobin. Unit of catalase activity was repre-sented as the amount of enzyme that degrades 1 μM H2O2 / min. For plotting standard graph, different concentra-tions of H2O2 were used. The activity of enzyme in each test sample was calculated by using this standard graph.
Estimation of hemoglobin
The concentrations of hemoglobin in all the hemolysate samples were measured by Drabkin’s method in which hemoglobin was oxidized to methemoglobin with alkaline ferric cyanide (Drabkin) reagent to give a brown coloured compound [10]. To 2.5 ml of Drabkin’s reagent, 10μl of whole blood was added. After 10 minutes, absorbance was measured against reagent blank at 540 nm. It was compared with the commercial Cyanomethemoglobin standard (15.5g/dL) and values were expressed as g %.
Calculation was done as, Concentration of Hemoglobin in percentage = (Reading of the test/Reading of the stan-dard) x Concentration of standard.
Estimation of reticulocyte count
To 1 ml of anti-coagulated (EDTA) blood 2-3 drops of the dye solution (1g Methylene blue in 100 ml of isotonic phosphate buffer pH-7.4) were added. The mixture was incubated at 37° C for 15-20 min. The cells were resus-pended by gentle mixing and films were made on glass slides which were then allowed to dry. A suitably visible area of film was chosen for the count where the cells were undistorted and where the staining was good. The reticulocyte count was made as percentage of the total number of cells counted in particular fields [11].
Statistical analysis
Comparative analysis for significant difference in means of various parameters between case and respective control groups was performed by student’s t test. Pearson correla-tion analysis was carried out to detect any association between individual oxidative stress and hematological parameters. The 5% significance level was considered. Further, regression analysis with sequential addition of potential predictor variables was done to explore their effect on reticulocyte count in homozygous cases. Statis-tical analysis of the data was carried out in SPSS 10.0 version.
Results
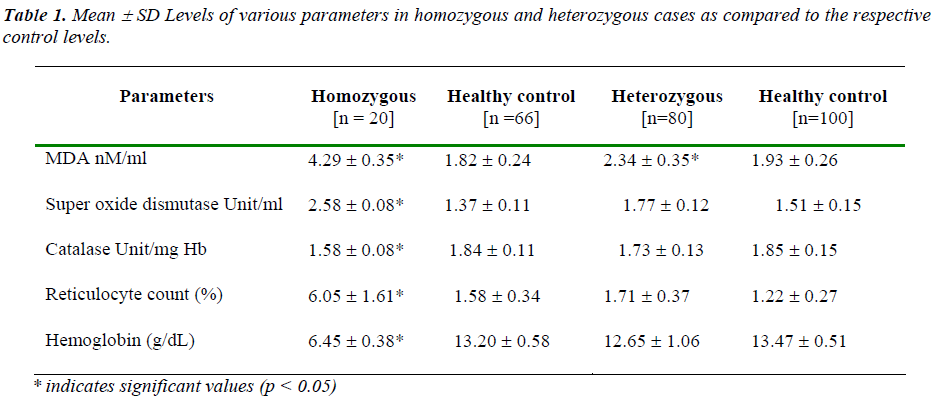
All the oxidative parameters except for RBC catalase acti-vity had shown increased level in cases as opposed to res-pective control levels. Catalase activity in RBC hemolys- ate actually showed a decrease in cases against control. However, apart from serum malondialdehyde which showed significant change in both the case groups as op-posed to respective control levels, all the other oxidative parameters had shown statistically significant difference only in homozygous case group, but not in heterozygous group. As a marker of hematological response hemoglo-bin level was recorded to be lower and reticulocyte count was higher in both the cases; however, these parameters were also found to be significantly different from respec-tive controls only in homozygous cases [Table1].
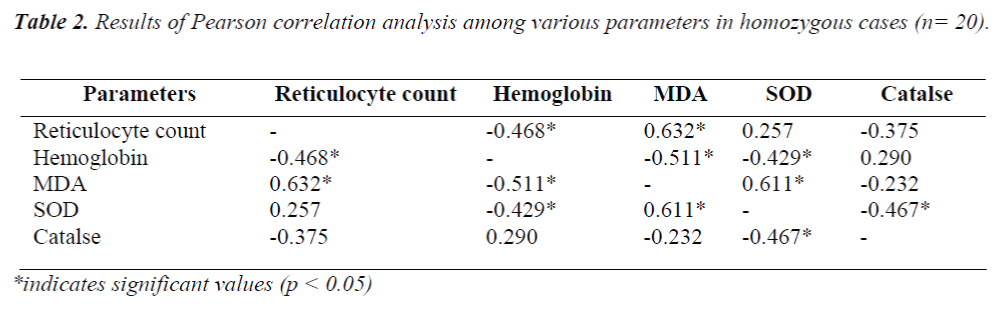
In heterozygous cases no parameter had shown any sig-nificant association among themselves. However, in ho-mozygous cases lipid peroxidation showed significant positive association with reticulocyte count and superox-ide dismutase activity; whereas a significant negative as-sociation with hemoglobin level was found. Significant inverse correlation was recorded between hemoglobin and reticulocyte count. Superoxide dismutase activity was found to bear significant negative correlation with hemo-globin level and also with catalase activity [Table 2].
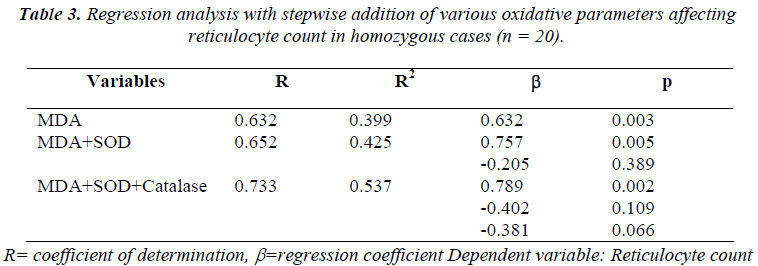
As deduced by linear regression analysis, lipid peroxida-tion was found to be the only oxidative parameter which was significantly associated with the reticulocyte count (R2 = 0.399, β = 0.632, p = 0.003).
Further regression study with stepwise addition of oxida-tive variables was carried out to assess the impact of vari-ous oxidative parameters on reticulocyte count, which showed lipid peroxidation marker, MDA as the signifi-cant predictor of the reticulocyte count even when the other oxidative parameters were added in the model [Table 3].
Discussion
Sickle cell disorder is associated with obvious hemolytic anemia due to the presence of abnormal hemoglobin vari-ant and such hemolytic response is predictably associated more with the homozygous than the heterozygous state. The severity of such response as mirrored by the degree of decrease in hemoglobin level in this study supported this fact. Such hemoglobin deficiency in turn may bring forth compensatory erythropoietic up-surge [12]. Hence reticulocyte may be considered as a direct marker of this response [13].
Hemolysis associated hematological stress in terms of erythropoietic response is documented in the form of hy-droxyurea induced oxidative stress leading to erythroid differentiation [14]. There is also evidence of oxidative stress mediated hemolysis [15] and such stress is impli-cated in erythropoietic response as well [16]. In the pre-sent study significant increase in hematological stress (recorded in terms of decrease in hemoglobin and con-comitant increase in reticulocyte count) in homozygous cases but not in heterozygous cases indicates the severity of the former condition. Moreover, significant increase in the observed levels of all the oxidative parameters in this severe form but not in the relatively milder heterozygous form of the disorder suggests a putative nexus between oxidative impact and the severity. In this context it is worth mentioning that significant increase in lipid peroxi-dation level in both the conditions probably indicates that this marker might be considered as a common final out-come of the oxidative assault. Even for this parameter, the observed increase against the corresponding control level is noticeably higher in homozygous cases. This further emphasizes a proportionate relationship of such oxidative stress with more severe form.
Only in homozygous cases significant association was found to exist between reticulocyte count and lipid per-oxidation and a concomitant negative association between hemoglobin level and lipid peroxidation. This suggests lipid peroxidative assault as an underlying factor for he-molysis and the consequent reticulocytosis. Superoxide dismutase, the first line of defense against free radical attack, also showed an analogous relationship with lipid-oxidative end product, MDA. However, catalase and SOD which acts in tandem was paradoxically found to bear significant negative association between themselves. In fact, a significant low catalase level in homozygous cases and also a non-significant yet definitive lower value of this enzyme in heterozygous cases appeared to be quite puzzling. In face of augmented demand for heme synthe-sis to meet erythropoietic upsurge in this pathology, pos-sibility of consequent plummet in production of catalse (also a porphyrin derivative) should be explored.
From these results it can be envisaged that there is a de-finitive association between the oxidative stress and the hematological impact in terms of hemolysis and reticulo-cytosis. Hence we performed a regression analysis with stepwise addition of oxidative stress parameters to find out most significant variable among these parameters for prediction of the reticulocytosis as a marker of hemopoi-etic response. As observed from correlation analysis, among all oxidative parameters studied, lipid peroxidation appeared to have most significant relationship with reticu-locyte count. Hence this parameter was considered as the first factor followed by sequential addition of other two enzymatic parameters of oxidative stress namely SOD and catalase to perform a regression analysis. The result clearly showed that the level of MDA may be considered as an independent factor for predicting the hematological stress response in sickle cell anaemia patients. It is inter-esting to note, that only this particular parameter was found to be significantly raised in both the clinical condi-tions as opposed to the respective control levels. This fur-ther endorses its significance in the diseases pathology.
Lipid peroxidation is the outcome of oxidative damage of the membrane lipid. Quite interestingly, earlier work in this disease pathogenesis confirmed that such oxidative membrane damage might be a crucial determinant of irre-versible sickling phenomenon in this disorder (1). More-over, increase in such ISC formation is reported to be di-rectly associated with severity of hemolysis in sickle cell anemia [17]. In this context it is worth mentioning that our earlier work in this disorder pointed out significant deficiency of vitamin E and vitamin C [18]. Strikingly these vitamins are major protectors of membrane oxida-tive assault in tissues with high oxygen tension like RBC [19].
Hence it can be surmised that probable impact of oxida-tive onslaught on erythrocyte membrane lipids might be considered as a pathogenic determinant in this disorder. However, to validate such crucial association of oxidative membrane damage in this hemoglobinopathy, a study dedicated exclusively in erythrocyte model is highly war-ranted. This study further advocates the potential of lipid peroxiadative parameter, as a candidate marker for assess-ing the severity of this clinical condition.
References
- Wang WC. Sickle cell anemia and other sicklingsyn-dromes: In: Wintrobe’s Clinical Hematology. 11th ed. Lipincott Williams& Wilkins, Philadelphia 2004; pp 1234-1311.
- Agarwal MB. The burden of haemoglobinopathies inIndia - time to wake up? J Acad Phys Ind 2005; 53: 1017-1018.
- Chiu D, Vichinsky E, Yee M, Kleman K, et al.Peroxi-dation, vitamin E, and sickle cell anemia. Ann N Y Acad Sc 1982; 393:323-335.
- Winterbourn CC. Oxidative destruction in congenitalhaemolytic anemias: The unstable hemoglobins. Semin Hematol 1990; 27: 41-50.
- Stock J, Offerman EL, Modell CB, et al. Thesuscepti-bility to autoxidation of human red cell lipids in health and disease.Brit J Haematol 1972; 23: 713-724.
- Wild BJ, Bain BJ. Investigations of abnormalhaemo-globins and thalassemia. In: Dacie and Lewis Practical Haematology. 9thed. Churchill Livingstone, London 2001; pp231-268.
- Satoh K. Serum lipid peroxide in cerebrovasculardis-orders determined by a new colorimetric method. Clin Chim Acta 1978; 90:37-43.
- Marklund S, Marklund G. Involvement of superoxideanion radical in the autooxidation of pyrogallol and a convenient assay forsuperoxide dismutase. Eur J Bio-chem 1974; 47: 469-474.
- Sinha AK. Colorimetric assay of catalase. AnalBio-chem. 1972; 47: 389-394.
- Drabkin D, Austin H. Spectrophotometric studies:Preparations from washed blood cells. J Biol Chem 1935; 112: 51-55.
- Dacie JV, Lewis SM. Basic HaematologicalTech-niques: In: Practical Haematology. 9th ed. Churchill Livingstone, London2001; pp19-46.
- Guarnone R, Centenara E, Zappa M, et al.Erythropoi-etin production and erythropoiesis in compensated and anaemic statesof hereditary spherocytosis. Brit J Haematol 2003; 92(1): 150-154.
- Golzan EF. Red Blood cell disorder: In: Pathology.Elsevier Mosby Philadelphia 2004; pp 121-141.
- Nagai T, Tarumoto T, Miyoshi T, et al. Oxidativestress is involved in hydroxyurea-induced erythroid differen-tiation. Brit JHaematol 2003; 121(4): 657-661.
- McMillan DC, Sarvate SD, Oatis JE Jr, et al. Roleof oxidant stress in lawsone-induced hemolytic anemia. Toxicol Sc 2004; 82(2):647-655.
- Duca L, Ponte AD, Cozzi M, et al. Changes inerythro-poiesis, iron metabolism and oxidative stress after half-marathon.Inter Emerg Med 2006; 1 (1): 30-34.
- Serjeant GR, Serjeant BE, Milner PF. Theirreversibly sickled cell: A determinant of haemolysis in sickle cell anaemia.Brit J Haematol 1969; 17: 527-529.
- Ray D, Deshmukh P, Goswami K, et al. Antioxidantvitamin levels in sickle cell disorders. Nat Med J Ind 2007; 1: 11-13.
- Bender DA, Mayes PA. Vitamins & minerals: In:Har-per‘s Illustrated Biochemistry. 26th ed. McGraw Hill (Asia). India2003; pp481-497.