Research Article - Environmental Risk Assessment and Remediation (2017) Novel approaches to Chemical Risk Assessment
Polybrominated diphenyl ether, polychlorinated biphenyl, and dechlorane plus concentrations in human serum, and their relationships with thyroid hormones.
Huixia Wang1, Meng Xu2,3, Junqi Li2,3, Ying Wang2,3, Songjie He2,3, Yiming Sun2,3, Mingyuan Li2,3, Jun Jin2,3*
1Beijing BCEG Golden Sources Environmental Protection Development Co., Ltd., 100101, PR China
2College of Life and Environment Sciences, Minzu University of China, Beijing 100081, PR China
3Engineering Research Center of Food Environment and Public Health of Beijing 100081, PR China
- *Corresponding Author:
- Jun Jin
Minzu University of China
(Central University for Nationalities)
PR China
Tel: +86 18910087635
E-mail: junjin3799@126.com
Accepted date: September 20, 2017
Citation: Wang H, Xu M, Li J, et al. Polybrominated diphenyl ether, polychlorinated biphenyl, and dechlorane plus concentrations in human serum, and their relationships with thyroid hormones. Environ Risk Assess Remediat. 2017;1(3):68-79
Abstract
Persistent halogenated organic compounds, including polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs), and dechlorane plus (DP), were determined in human serum collected in Weifang and Nanning, China, and the relationships between their concentrations and thyroid hormone concentrations were determined. The total PBDE (Σ10PBDEs), total PCB (Σ14PCBs), and total DP (ΣDP) concentrations were higher in Weifang (medians 220, 4.3, and 2.2 ng/g lipid, respectively) than in Nanning (medians 180, 1.9, and 1.5 ng/g lipid, respectively). The donors’ ages correlated with Σ14PCBs but not with Σ10PBDEs or ΣDP in Weifang. Age positively correlated with Σ10PBDEs but not Σ14PCBs and ΣDP in Nanning. The correlations between the individual PBDE, PCB, and DP congener and thyroid hormone concentrations were analyzed by stepwise multiple linear regression after log2-transforming the variables. Triiodothyronine (T3) concentrations were associated with BDE-99 concentrations and ΣDP in Weifang but with BDE-153 concentrations and Σ10PBDEs in Nanning. A positive correlation was found, for the first time, between anti-DP and free-T3 concentrations in Weifang, but free-T3 concentrations correlated with PCB-118 and PCB-170 concentrations in Nanning. Thyroxine (T4) concentrations correlated with total dioxin-like PCB concentrations in Weifang but with PCB-156 concentrations in Nanning. No significant correlations were found between the analyte and free-T4 concentrations in Weifang, but negative correlations were found between free-T4 and total dioxin-like PCB and BDE-190 concentrations in Nanning. No significant correlations were found between the analyte and thyroid-stimulating hormone (TSH) concentrations in Weifang but negative correlations were found between Σ10PBDEs and the TSH concentrations in Nanning. The correlations between the analyte and thyroid hormone concentrations followed different trends in Weifang and Nanning but were similar to those that have been found in animal and epidemiological studies.
Keywords
persistent halogenated organic compounds; human serum; thyroid hormones; correlation
Introduction
Persistent halogenated organic compounds have been used in a variety of commercial products around the world. They are of global public health concern because of both their persistence and the potentially adverse health effects they can cause. Disturbances in concentrations of thyroid hormones (THs) have been found in animals and humans that have been exposed to brominated flame retardants and polychlorinated biphenyls (PCBs) [1-3]. Polybrominated diphenyl ethers (PBDEs) are a class of brominated flame retardants and they have been widely used in many commercial products, including furniture and textiles [4,5]. PBDEs have been found to bioaccumulate in environment because of their high degrees of lipophilicity and resistances to degradation [5,6]. The production of the pentaBDE and octaBDE commercial PBDE products has been phased out because of the potential toxicities of their components. The use of the decaBDE commercial product has also been banned in the EU and in some states in the USA [7,8]. PBDEs have been recognized as being ubiquitous contaminants, and they have been found in environmental media [9,10], wildlife [11,12], and humans [13-17]. PBDE concentrations in human serum have increased exponentially in the last three decades [18,19]. PBDEs are structurally similar to thyroxine (T4), and it has been shown in in vivo animal studies that exposure to PBDEs can potentially lead to disruptions to thyroid homeostasis and TH functions [20-24]. It is also suspected that PBDEs can disrupt thyroid functions in humans, but reports of impacts of PBDE exposure on human thyroid functions have been controversial [25-27].
Until they were banned in most countries in the late 1970s, PCBs were used extensively in industrial and commercial applications, including as dielectric fluids in capacitors, heat exchange fluids, and transformers. Although they have been banned for decades, PCBs are still ubiquitous environmental pollutants around the world, and they have been found in environmental media (including air, water, sediment, and soils) and in human serum and milk [28,29]. PCB concentrations in human blood have been decreasing since the 1970s, mainly because of the ban on their use [19]. It has been observed in rats and seals that PCBs can disturb thyroid homeostasis and TH functions [30]. PCBs can also disturb TH homeostasis in humans, but the reports regarding this are few and their results have not been consistent [31-34].
Dechlorane plus (DP) is a highly chlorinated flame retardant with major applications in polymers [35], and is currently classified as a high production volume chemical in the USA [36,37]. The only DP manufacturing plant in China is in Huai’an, Jiangsu Province, and it produces 300-3000 t of DP per annum [38]. DP has been detected in air, sediment, soil, fish, herring gull eggs, and human hair, breast milk and serum [39-44]. Data on human body burdens of DP, especially data suitable for the toxicological assessment of the potential effects of DP in the general population, remain scare [45].
Thyroid hormones of thyroxine (T4), triiodothyronine (T3), thyroid-stimulating hormone (TSH), free T4 (fT4), and free T3 (fT3) play very important roles in neurodevelopment. It has been suggested in a number of publications (a number that continues to grow) that exposure to persistent organic pollutants, including PBDEs and PCBs, may disrupt TH homeostasis in humans, but the clinical implications of this are questionable [46-48]. Few specific comparative studies in two different populations that are exposed to these chemicals have been performed. The first aim of the study presented here was to determine the PBDE, PCB, and DP concentrations in serum collected from people living in two areas of China. The two areas were Weifang, where brominated flame retardants are produced, and Nanning, where none of the analytes are produced. The second aim was to evaluate the relationships between the PBDE, PCB, DP and TH concentrations in the serum samples from each region.
Materials and Methods
Study participants and sample collection
Weifang is a major halogenated flame retardant production region, and the main product is decaBDE (which is predominantly composed of the congener BDE-209). The people living in Weifang are exposed to an environment that is heavily contaminated with PBDEs. Samples from 75 native residents of Weifang were chosen for use in this study. Samples from 73 people who live in Nanning, where there is less industrial activity using halogenated flame retardants, were selected for use as a control group, for comparison with the Weifang group. The serum samples in our study were collected with the help of nurses from Weifang Binhai People’s Hospital and Nanning Yudong’s Hospital between June 2011 and June 2012. The serum samples were collected by nurses, which were used for routine wellness tests. The nurses asked the serum provider if they were willing to be involved in our study. When the adult volunteers or the caretakers and guardians on behalf of the children volunteers agreed to participate in our study and they said YES for the sample collection after the nurses in hospital had clearly explained the project, the surplus stored serum were numbered and were used in our study. Then the nurses transferred these numbered serum samples to us with the information of age, sex of the sample provider, the TH concentrations in the serum, and the collection date. All the serum samples we obtained were anonymized. Then we selected about one sixth of the total number of serum samples available on each sampling date. The ethics committees approved this consent procedure for adults and children. About 3.0 mL of serum was collected from each sample and a total of 148 serum samples were collected (75 from Weifang and 73 from Nanning). Each sample was taken from the cubital vein and collected in a polypropylene vacuum tube by a nurse. Each sample was centrifuged at 3 000 rpm for 15 min, then the serum was transferred into a new tube. The samples were frozen, shipped to the laboratory, and stored at -18°C until they were analyzed. The mean age of the donors was 49 years (range 26-83 years) in Weifang and 39 years (range 17-85 years) in Nanning. Our study was reviewed and approved by the ethic committee in the College of Life and Environment Science in Minzu University of China.
Chemicals and materials
The chemicals that were analyzed were specific BDE congeners (BDE-28, -47, -85, -99, -100, -153, -154, -183, -190, and -209), specific PCB congeners (PCB-77, -81, -105, -114, -118, -123, -126, -156, -157, -167, -169, -170, -180, and -189), and syn- and anti-DP. Unlabeled BDE-28, -47, -99, -100, -153, -154, -183, and -209, 13C12-labeled BDE-139 and -209, and 13C10-labeled syn-DP were purchased from Cambridge Isotope Laboratories (Andover, MA, USA). Unlabeled anti-DP and syn-DP were purchased from Wellington Laboratories (Guelph, Canada). A 13C12-labeled PCB internal standard mixture (PCB-77, -81, -105, -114, -118, -123, -126, -156, -157, -167, -169, -170, -180, and -189) was purchased from Cambridge Isotope Laboratories and an unlabeled PCB standard mixture (PCB-28, -52, -77, -101, -105, -114, -118, -123, -126, -138, -153, -156, -167, -169, -180, and -189) was purchased from Dr. Ehrenstorfer (Augsburg, Germany).
Dichloromethane, methyl tert-butyl ether (MTBE), hexane, and acetone were of LC grade or pesticide grade, and were purchased from J.T. Baker (Phillipsburg, NJ, USA). Other chemicals were of analytical grade, and were purchased from Beihua (Beijing, China). Water was purified using a Milli-Q system (Millipore, Billerica, MA, USA). Helium (99.999%), methane (99.999%), and nitrogen (99.999%) were obtained from Chengweixin (Beijing, China). Bio-beads SX-3 (0.098-0.046 mm) were purchased from Bio-Rad Laboratories (Shanghai, China).
Sample preparation
The serum samples were extracted and cleaned using a method that has been described previously [49]. Briefly, each sample was spiked with a known amount of internal standards (13C12-labeled BDE-139 and -209, 13C12-labeled PCBs, and 13C10-labeled syn- DP), and hydrochloric acid and isopropanol were added. The sample was then extracted twice with a hexane/MTBE mixture (1:1 v:v), washed with aqueous potassium chloride (1% w/w), and then evaporated to dryness. Next, 4.0 mL hexane and 2.0 mL 0.50 mol/L KOH were added to each sample, the mixture was shaken, and the hexane was transferred to another vessel. The sample was extracted twice more with 3.0 mL hexane each time, and the three hexane fractions were mixed together and then concentrated to approximately 1.0 mL. The extract was then cleaned up using gel permeation chromatography and a multilayer silica gel column, then concentrated to 100 μL for instrumental analysis.
Instrumental analyses
The extracts were analyzed using an Agilent 6890N gas chromatograph and an Agilent 5975N mass spectrometer (Agilent Technologies Inc., Santa Clara, CA, USA). The PBDEs (except for BDE-209), PCBs, and DP were separated using a DB-5MS gas chromatography column (30 m long, 0.25 mm i.d., 0.1 μm film thickness; J&W Scientific., Folsom, CA, USA), and BDE-209 was separated using a DB-5 column (15 m long, 0.25 mm i.d., 0.1 μm film thickness; J&W Scientific).
For the PBDE (except for BDE-209) and DP analyses, the oven temperature program was started at 100°C (held for 3 min), then increased by 4°C/min to 300°C, which was held for 12 min. 13C12-labeled BDE-139 was used as a surrogate internal standard for the other BDE congeners and 13C10-labeled syn-DP was used as a surrogate internal standard for anti-DP. The m/z ratios that were monitored were 79.0, 81.0, 573.6, and 575.6 for 13C12-labeled BDE-139, 79.0 and 81.0 for the unlabeled BDE congeners, 652.0 and 654.0 for syn- and anti-DP, and 666.0 and 668.0 for 13C10-labeled syn-DP.
For BDE-209, the oven temperature program was started at 100°C (held for 3 min), then increased by 10°C/min to 290°C, which was held for 8 min. 13C12-labeled BDE-209 was used as a surrogate internal standard for BDE-209. The m/z ratios 407.0 and 486.0 were monitored for the unlabeled BDE-209 and the m/z ratios 415.0 and 494.0 were monitored for 13C12-labeled BDE-209.
For the PCBs, the oven temperature program was started at 100°C, which was held for 3 min, then increased by 5°C/min to 250°C. The m/z ratios that were monitored were 290.0 and 292.0 for PCB-81 and -77, 326.0 and 328.0 for PCB-123, -118, -114, -105, and -126, 360.0 and 362.0 for PCB-167, -156, -157, and -169, 394.0 and 396.0 for PCB-180, 396.0 for PCB-170, and 394.0 and 396.0 for PCB-189. The 13C12-PCBs were monitored using the unlabeled analog m/z ratio plus 12.
The carrier gas was helium, and it was used at a flow rate of 1.0 mL/min. Sample aliquots (1 μL) were injected in splitless mode. The mass spectrometer was used in negative chemical ionization mode and the ion source and quadrupole temperatures were both 150°C. The chemical ionization moderating gas was methane.
Quality assurance/quality control
A method blank sample was included with every batch of serum samples that was analyzed. The analyte concentrations in the blank samples were very low, so the concentrations in the samples were not blank corrected.
The instrumental limit of detection (LOD) was defined as a peak giving a signal to noise ratio of 5. The instrumental LODs for the dioxin-like (DL-) PCBs, PCB-170, and PCB-180 were 0.050-0.20, 0.020, and 0.010 pg, respectively. The instrumental LOD for BDE-209 was 10pg, and the instrumental LODs ranged from 0.10 to 0.20 pg for the other BDE congeners. The instrumental LOD for DP was 0.20 pg.
The mean recoveries of the 13C-labeled syn-DP, BDE-139, BDE-209, and PCBs in the serum samples were 84-100%, 92- 110%, 72-92%, and 55-61%, respectively.
Statistical analysis
Analytes that were detected in at least 50% of the serum samples were included in the statistical analyses, and a value of LOD/2 was used for concentrations below the LOD. The statistical analyses were carried out using SPSS 16.0 software (SPSS Inc., New York, NY, USA). The nonparametric Kolmogorov- Smirnov Z test was used to test the normality of the distribution. The analyte and TH concentrations were not normally distributed (Kolmogorov–Smirnov Z test, p<0.05). Differences in the chemical concentrations in the samples from the two sampling areas were tested using the Mann–Whitney U test. Spearman correlation tests were used to identify correlations between the analyte concentrations and age and between the analyte and TH concentrations. The analyte and TH concentrations were log2 transformed to give approximately normal distributions, then correlations between the analyte and TH concentrations were identified using a stepwise multiple linear regression method.
Results and Discussion
A statistical summary of the analyte and TH concentrations in the serum samples from the two study regions is presented in Table 1. The PBDEs were the most abundant analytes in both regions, the total PBDE concentration contributing 97% and 98% of the total analyte concentrations in Weifang and Nanning, respectively. The PCBs were the next most abundant analyte group and the DPs were the least abundant group.
| Weifang (n=75) | Nanning (n=73) | |||||||
|---|---|---|---|---|---|---|---|---|
| All donors | All donors | |||||||
| detection | Meana ± SDb | Range | Median | detection | Mean ± SD | Range | Median | |
| frequencty | (ng/g lipid) | (ng/g lipid) | (ng/g lipid) | frequencty | (ng/g lipid) | (ng/g lipid) | (ng/g lipid) | |
| Age (years) | 49 ± 17 | 26–83 | 43 | 39 ± 18 | 17–85 | 33 | ||
| BDE-28 | 93% | 2.6 ± 5.7 | <0.30–48 | 1.3 | 22% | 0.14 ± 0.29 | <0.30–1.0 | <0.30 |
| BDE-47 | 78% | 1.3 ± 3.5 | <0.30–28 | 0.6 | 29% | 0.46 ± 1.5 | <0.30–11 | <0.30 |
| BDE-100 | 100% | 1.3 ± 0.64 | <0.20-4.2 | 1.2 | 93% | 1.3 ± 0.79 | <0.20–6.1 | 1.2 |
| BDE-99 | 100% | 3.4 ± 3.0 | <0.20–17 | 2.6 | 100% | 2.7 ± 1.1 | 0.82–5.0 | 2.6 |
| BDE-154 | 21% | 0.10 ± 0.22 | <0.30–0.95 | <0.30 | 16% | 0.054 ± 0.18 | <0.30–0.79 | <0.30 |
| BDE-85 | 90% | 1.3 ± 0.88 | <0.30–4.7 | 1.3 | 79% | 1.0 ± 0.82 | <0.30–3.1 | 0.85 |
| BDE-153 | 100% | 4.3 ± 4.7 | <0.40–32 | 2.9 | 81% | 8.5 ± 61 | <0.40–520 | 1.2 |
| BDE-183 | 60% | 1.7 ± 3.6 | <0.40–27 | 0.95 | 45% | 0.84 ± 2.5 | <0.40–19 | <0.40 |
| BDE-190 | 90% | 3.9 ± 3.1 | <1.0–14 | 2.8 | 86% | 2.4 ± 1.6 | <1.0–6.3 | 2.3 |
| BDE-209 | 96% | 2100 ± 12000 | <15–9700 | 210 | 92% | 550 ± 1500 | <15–9700 | 160 |
| ∑PBDEsc | 100% | 2100 ±12000 | 3.9–9700 | 220 | 100/00% | 570 ± 1500 | 4.2–9700 | 180 |
| PCB-81 | 0.00% | n.d. | <0.20 | <0.20 | 11% | 0.067 ± 0.21 | <0.20–1.1 | <0.20 |
| PCB-77 | 1.40% | 0.049±0.41 | <0.20–3.5 | <0.20 | 6.90% | 0.13± 0.63 | <0.20–4.1 | <0.20 |
| PCB-123 | 9.70% | 0.068 ± 0.22 | <0.10–1.6 | <0.10 | 8.20% | 0.020 ± 0.069 | <0.10–0.31 | <0.10 |
| PCB-118 | 67% | 0.77 ± 0.47 | <0.10–2.0 | 0.66 | 47% | 0.29 ± 0.39 | <0.10–1.9 | <0.10 |
| PCB-114 | 17% | 0.041± 0.075 | <0.04–0.25 | <0.040 | 8.20% | 0.011 ± 0.037 | <0.04–0.18 | <0.040 |
| PCB-105 | 49% | 0.26 ± 0.14 | <0.10–0.60 | 0.26 | 42% | 0.11 ± 0.15 | <0.10–0.58 | <0.10 |
| PCB-126 | 0.00% | n.d. | <0.04 | <0.04 | 0.00% | 0.0088 ± 0.070 | <0.04–0.59 | <0.040 |
| PCB-167 | 68% | 0.13 ± 0.074 | <0.04-0.41 | 0.11 | 52% | 0.059 ± 0.080 | <0.04-0.41 | 0.046 |
| PCB-156 | 83% | 0.40 ± 0.38 | <0.04–2.6 | 0.35 | 63% | 0.18 ± 0.18 | <0.04–0.75 | 0.19 |
| PCB-189 | 44% | 0.033 ± 0.049 | <0.02–0.29 | <0.02 | 32% | 0.041 ± 0.16 | <0.02–1.3 | <0.020 |
| PCB-157 | 47% | 0.044 ± 0.054 | <0.04–0.18 | <0.04 | 40% | 0.025 ± 0.033 | <0.04–0.13 | <0.040 |
| PCB-169 | 0.00% | n.d. | <0.04 | <0.04 | 0.00% | n.d. | <0.04 | <0.040 |
| PCB-180 | 96% | 2.4 ± 1.9 | <0.02–10 | 1.9 | 96% | 0.95 ± 0.63 | <0.02–3.7 | 0.78 |
| PCB-170 | 90% | 0.67 ± 0.58 | <0.04–3.6 | 0.49 | 73% | 0.24 ± 0.24 | <0.04–1.2 | 0.19 |
| ∑DL-PCBsd | 86% | 1.8 ± 1.1 | 0.43–7.9 | 1.5 | 68% | 0.94 ± 1.1 | <0.02–5.1 | 0.75 |
| ∑PCBse | 96% | 4.8 ± 3.1 | 0.92–17 | 4.3 | 96% | 2.1 ± 1.4 | <0.02–6.1 | 1.9 |
| s-DP | 69% | 1.2 ± 1.9 | <0.30–11 | 0.9 | 37% | 0.55 ± 1.3 | <0.30–7.5 | <0.30 |
| a-DP | 93% | 2.6 ± 3.9 | <0.30–26 | 1.3 | 82% | 1.9 ± 2.3 | <0.30–13 | 1.3 |
| ∑DP | 93% | 3.8 ± 5.4 | <0.30–36 | 2.2 | 8219% | 2.4 ± 3.4 | <0.30–20 | 1.5 |
| fant f | 0.71 ± 0.20 | 0.27–1.0 | 0.68 | 0.87 ± 0.17 | 0.35–1.0 | 1 | ||
| unit | Mean ± SD | Range | Median | unit | Mean ± SD | Range | Median | |
| T3g | ng/ml | 1.6 ± 0.92 | 0.42–7.7 | 1.4 | nmol/L | 1.7 ± 0.44 | 0.69–2.8 | 1.7 |
| T4h | μg/dl | 9.4 ± 2.6 | 4.8–24 | 9.1 | nmol/L | 110 ± 26 | 54–170 | 120 |
| fT3 | pmol/L | 4.5 ± 2.4 | 1.7–23 | 4.2 | pmol/L | 4.4 ± 0.90 | 1.9–6.7 | 4.5 |
| fT4 | pmol/L | 16 ± 3.0 | 8.1–24 | 16 | pmol/L | 15 ± 3.5 | 7.5–28 | 16 |
| TSHi | μIU/ml | 2.0± 1.9 | 0.02–14 | 1.5 | mIU/L | 1.4 ± 1.1 | 0.020–8.3 | 1.1 |
Table 1: Concentrations of polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs), dechlorane plus(DP),and thyroid hormonesin serum samples from Weifang and Nanning, China.
PBDEs in serum from the two study regions
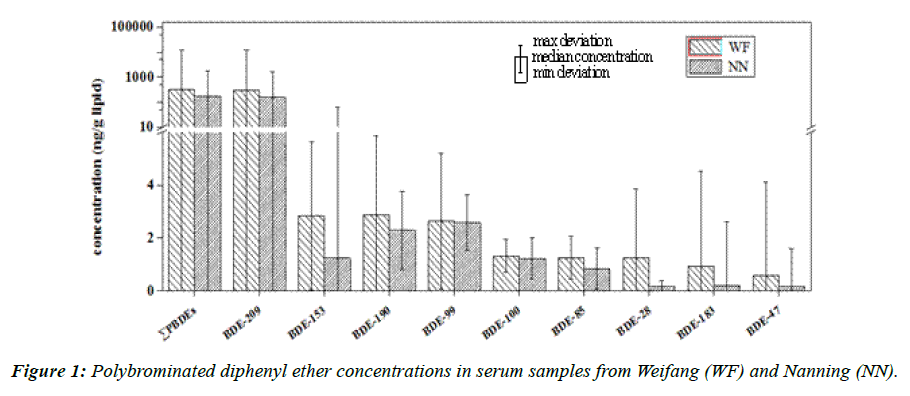
Ten BDE congeners (BDE-28, -47, -85, 99, -100, -153, -154, -183, -190, and -209) were found in the serum samples. The sum of the concentrations of these congeners (Σ10PBDEs) and the concentrations of the dominant BDE congeners in the samples from the two study regions are shown in Figure 1. The Σ10PBDEs concentrations ranged from 3.9 to 9 700 ng/g (median 220 ng/g lipid) and 4.2 to 9 700 ng/g lipid (median 180 ng/g lipid) in the samples from Weifang and Nanning, respectively. The Σ10PBDEs concentrations were significantly higher in the Weifang samples than that in the Nanning samples (Mann–Whitney U test, p<0.05) (Figure 1), which is consistent with elevated exposures expected in the Weifang population because of the local production of PBDEs. The concentration of each BDE congener was also higher in Weifang (Figure 1) than in Nanning, which implies that the emission of each BDE congener is higher in Weifang than in Nanning.
The highest PBDE concentration in a human that has been reported up to now was 9600 ng/g lipid, and it was found in an adipose tissue sample collected in New York [50]. Eleven BDE congeners (dibrominated to hexabrominated diphenyl ethers) were identified in that sample. A Σ10PBDEs concentration of 9700 ng/g lipid was found in a serum sample taken from a 27 year old male in Weifang in our study, and this is the highest PBDE concentration that has ever been reported in the literature. It is possible that this high PBDE concentration was caused by the individual working in a factory in which PBDEs are produced. The median Σ10PBDEs concentrations in the samples from Weifang (220 ng/g lipid) and Nanning (180 ng/g lipid) were lower than the concentrations that were found in electronic dismantling plant workers in Guiyu, China (median sum of the concentrations of 16 BDE congeners 600 ng/g lipid) [51], but higher than have been found in the general population in northern China (median sum of the concentrations of 41 BDE congeners 7.1ng/g lipid) [52], UK (median sum of the concentrations of 19 BDE congeners 5.6 ng/g lipid) [53], and Korea (median sum of the concentrations of 27 congeners 6.0 ng/g lipid) [54].
The most abundant BDE congener in the serum samples from both regions was BDE-209, which contributed 93% and 92% of the Σ10PBDEs in Weifang and Nanning, respectively. This implies that large amounts of the decaBDE commercial mixture is currently being produced and used in China. It is not surprising, therefore, that serum from both the region in which PBDEs are produced (Weifang) and the region in which PBDEs are not produced (Nanning) contained high proportions of BDE- 209 relative to the other PBDEs.
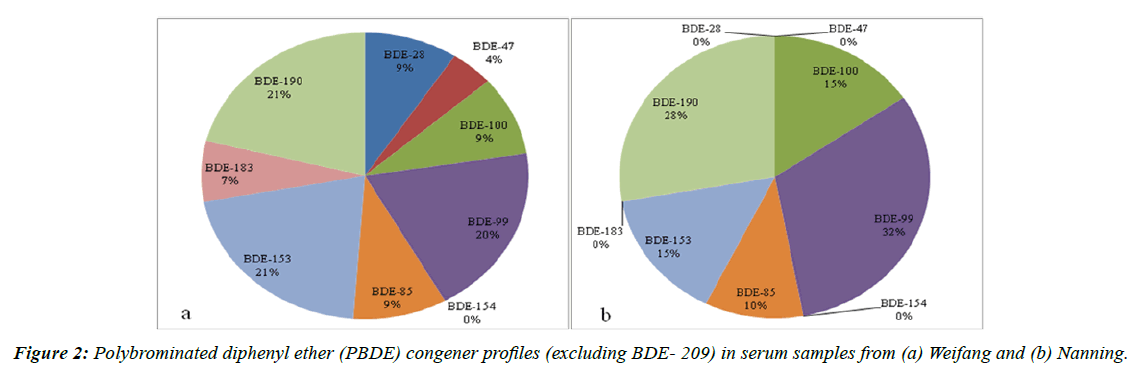
After excluding the contribution of BDE-209 to the samples, BDE-190 and -153 contributed the most (21%) to the Σ9PBDEs (tribrominated to heptabrominated diphenyl ether congeners) concentrations in Weifang, whereas BDE-99 was the largest contributor (32%) to the Σ9PBDEs concentrations in Nanning (Figure 2). This is unusual because BDE-47 is normally the largest contributor to the total PBDE concentration (excluding BDE-209) [55-57]. The PBDE patterns we found were also different from the patterns found in personnel at an electronic waste dismantling plant, in which BDE-183 was the major congener after BDE-209 [51].
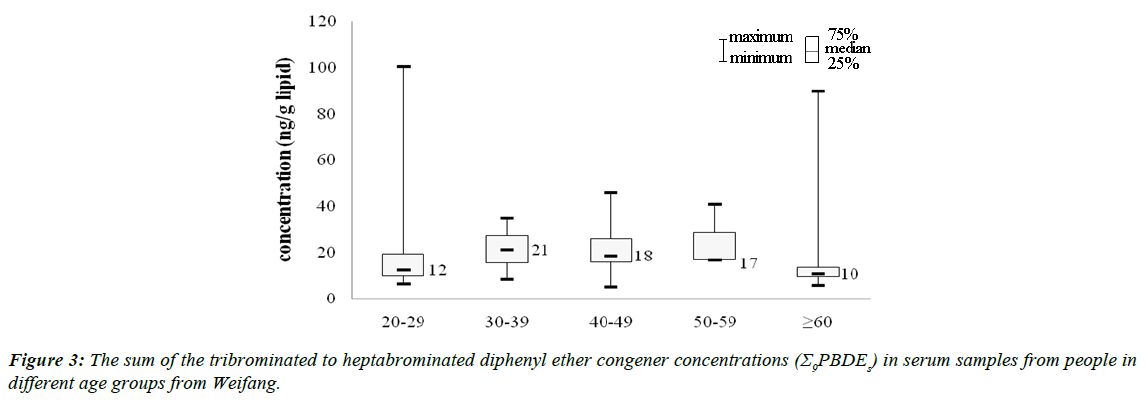
We performed a Spearman’s rank-order correlation analysis between the donors’ ages and the PBDE concentrations. The BDE congener concentrations in the serum did not correlate with the age of the donor except for BDE-85 (rho = −0.319, p=0.007) in Weifang (Table 1 in the SI). No correlations were found between the donors’ ages and the Σ10PBDEs or Σ9PBDEs concentrations (p>0.05), and no correlations were found between PBDE concentrations and donors’ ages in previous studies [50,58,59]. The group aged 30-39 years had the highest Σ9PBDEs concentrations (Figure 3), which is consistent with the PBDE production history in Weifang (PBDE production began in the 1980s, around the time when people aged 30-39 years were born). The people aged 30-39 years were, therefore, exposed to an environment that was contaminated with PBDEs from the time they were born, leading to them having the highest PBDE concentrations of all of the age groups. This result is consistent with the results of our previous study [60].
In contrast, the BDE-209 and Σ10PBDEs concentrations positively correlated with age in Nanning (p<0.05) (Table 1 in the SI), which is not surprising because Nanning is not an area in which PBDEs are produced. Continued exposure to PBDEs might have caused the PBDE concentrations in the serum samples to increase with the age of the participants of our study. However, the BDE-85, -99, -100, -190, and Σ9PBDEs concentrations did not follow this trend, and actually had negative correlations with age (rho<0, p<0.05) (Table 1 in the SI), which may be explained by younger people, in general, having more contact than older people with consumer electronic products that may contain the pentaBDE commercial product.
PCB concentrations in serum samples from the two study regions
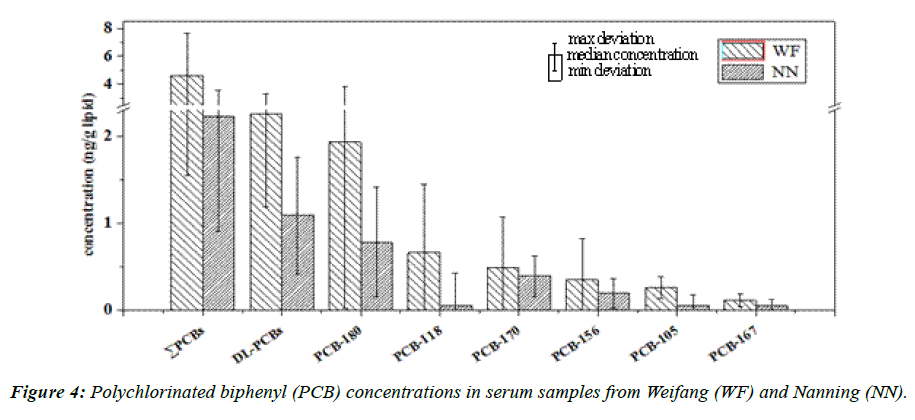
The DL-PCBs (PCB-77, -81, -105, -114, -118, -123, -126, -156, -157, -167, -189, and -169), PCB-170, and PCB-180 were found in the serum samples. PCB-180 was the most abundant of these congeners in both regions, accounting for 47% and 41% of the sum of the 14 congener (Σ14PCBs) concentrations in Weifang and Nanning, respectively. The Σ14PCBs concentrations in the serum ranged from 0.92 to 17 ng/g lipid (median 4.3 ng/g lipid) in Weifang and from <0.20 to 6.1 ng/g lipid (median 1.9 ng/g lipid) in Nanning. The Σ14PCBs concentrations were significantly higher (Mann–Whitney U test, p<0.05) in the serum samples from Weifang than from Nanning (Figure 4). The concentrations of the main PCB congeners (shown in Figure 4) were also significantly higher in the samples from Weifang than from Nanning (Mann–Whitney U test, p<0.05). The mean Σ14PCBs concentrations in the samples from Weifang (4.3 ng/g lipid) and Nanning (1.9 ng/g lipid) were both lower than the concentrations that have been found in serum samples from Guangdong, China (sum of the concentrations of 23 congeners 65 ng/g lipid) [51] and Shanghai, China (sum of the concentrations of 20 congeners 70 ng/g lipid) [61], and much lower than the concentrations that have been found in serum samples from UK (sum of the concentrations of 23 congeners 130 ng/g lipid) [53] and Canada (sum of the concentrations 14 congeners 180 ng/g lipid) [62].
The Σ14PCBs concentrations in the serum samples correlated with age in Weifang (rho=0.276, p=0.019), and each of the main congeners included in the analysis correlated with age (Table 1 in the SI, p<0.05). PCB concentrations have generally been found to increase with age in other studies [50,59]. However, in this study the Σ14PCBs concentrations did not correlate with age in Nanning, although the concentrations of a few individual congeners did correlate with age (Table 1 in the SI, p<0.05). This was probably because the PCB concentrations in the environment in Nanning are relatively low, and differences in exposure related to age may not be very large.
DP concentrations in the serum samples from the two study regions
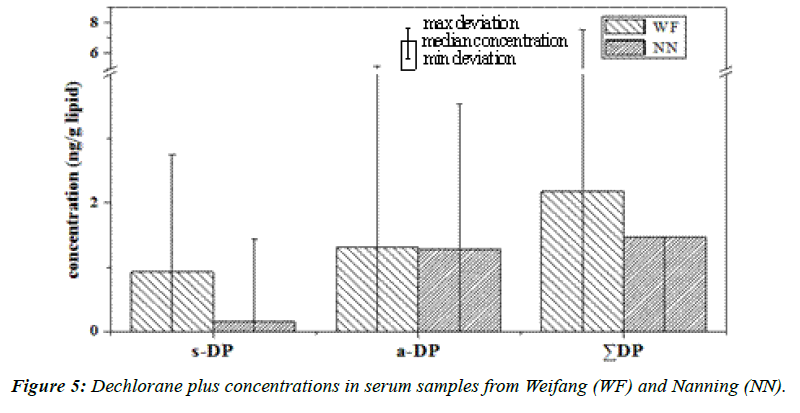
The serum samples were analyzed for syn-DP and anti-DP. The total DP (ΣDP) concentrations ranged from <0.30 to 36 ng/g lipid (median 2.2 ng/g lipid) and from <0.30 to 20 ng/g lipid (median 1.5 ng/g lipid) in the serum samples from Weifang and Nanning, respectively. The ΣDP, syn-DP, and anti-DP concentrations were higher in the samples from Weifang than from Nanning (Figure 5). The ΣDP concentrations in the samples from both regions were lower than were found in serum samples from the general population in Guangzhou, China (median 4.6 ng/g lipid) [63] and much lower than were found in serum samples from residents of the electronic waste dismantling regions of Guiyu (median 43 ng/g lipid) [64] and Qingyuan (median 150 ng/g lipid) [63], which is consistent with the dismantling of electronic waste products being a major source of DP [64].
In the serum samples from both of the study regions, anti-DP was the dominant congener, accounting for 58% and 88% of the ΣDP concentrations in Weifang and Nanning, respectively. The DP isomer ratios (fanti) found in the serum samples are shown in Table 1. The median fanti values were 0.68 and 1.0 in the samples from Weifang and Nanning, respectively, and these values were significantly different (Mann–Whitney U test, p<0.05). The median fanti value for the samples from Weifang was comparable to the fanti values that have been found in commercial products (0.60–0.80) [8,42,65-67] while the median fanti for the samples from Nanning was higher than the fanti that has been found in commercial products. The fanti values that have been found in biota samples have ranged from 0.0030 to 1.0 [8,42,64,68,69], and Yan et al. (2012) found fanti values of 0.56 to 0.71 (median 0.60) in samples taken from the general in Guangzhou. Ren et al. (2009) determined DP concentrations in serum taken from residents of the Guiyu electronic waste dismantling region and residents of Haojiang, and found fanti values of 0.40 to 0.77 (median 0.53) in Guiyu and 0.57 to 0.78 (median 0.64) in Haojiang, which are lower than the values that we found in serum samples from Weifang (median 0.68) and Nanning (median 1.0) [61]. Different fanti values have been found in serum samples in other studies [63,70].
The ΣDP, syn-DP, and anti-DP concentrations in the serum samples from Weifang correlated with the donors’ ages (p<0.05) (Table 1 in the SI, p<0.05). However, no such correlations were found for the samples from Nanning. No correlation between the fanti values and the volunteers’ ages was found for either region.
Relationships between TH concentrations and PBDE, PCB, and DP concentrations
The T3, T4, fT3, fT4, and TSH concentrations were determined in all of the serum samples that were analyzed in this study. Interestingly, the T3, T4, fT3, fT4, and TSH concentrations in the serum samples that contained extremely high Σ10PBDE concentrations (9700 ng/g lipid in serum from a male in Weifang and 9700 ng/g lipid in serum from a female in Nanning) were within the normal ranges. Correlations between the analyte and the TH concentrations were examined using Spearman’s rankorder correlation test (Table 2 in the SI).
| WF | NN | |||||
|---|---|---|---|---|---|---|
| Independent | Dependent | Dependent | ||||
| Predictor | B | p-value | Predictor | B | p-value | |
| T3 | log2∑DP | 0.177 | 0 | log2∑PBDEs | −0.078 | 0.001 |
| log2BDE-99 | −0.192 | 0.05 | log2BDE-153 | 0.064 | 0.017 | |
| T4 | log2∑DL-PCBs | −0.094 | 0.022 | log2PCB-156 | 0.056 | 0.008 |
| fT3 | log2PCB-118 | 0.113 | 0.000 | |||
| log2a-∑DP | 0.111 | 0.001 | ||||
| log2PCB-170 | −0.06 | 0.003 | ||||
| fT4 | log2∑DL-PCBs | 0.135 | 0.001 | |||
| log2BDE-190 | 0.101 | 0.006 | ||||
| TSH | log2∑PBDEs | −0.127 | 0.049 | |||
Table 2: Significant correlations between thyroid hormones (triiodothyronine (T3), thyroxine (T4), free T3 (fT3), free T4 (fT4), and thyroidstimulating hormone (TSH)) and polybrominated diphenyl ether (PBDE), polychlorinated biphenyl (PCB), anddechlorane plus (DP) concentrations in serum samples from Weifang (WF) and Nanning(NN), determined using a stepwise multiple linear regression method.
The T3 concentrations in the serum samples from Weifang significantly correlated with the syn-DP and ΣDP concentrations. The BDE-47, syn-DP, anti-DP, and ΣDP concentrations significantly positively correlated with the fT3 concentrations in the samples from Weifang. The T4 concentrations significantly negatively correlated with the PCB-180, PCB-170, Σ14PCBs, and ΣDL-PCBs concentrations in the samples from Weifang. The Σ10PBDEs concentrations significantly correlated with the fT4 concentrations and the BDE-85 concentrations significantly correlated with the TSH concentrations in the samples from Weifang. However, the T3 concentrations significantly negatively correlated with the BDE-209 and Σ10PBDEs concentrations in the samples from Nanning, and the fT3 concentrations correlated with the BDE-85, -99, -100, and -190, Σ9PBDEs, PCB-118, and ΣDL-PCBs concentrations and negatively correlated with the BDE-209 and Σ10PBDEs concentrations. The PCB-156 concentrations significantly positively correlated with the T4 concentrations in the samples from Nanning. The fT4 concentrations significantly correlated with the BDE-28, -85, -99, -100, and -190, Σ9PBDEs, PCB-118 and -156, Σ14PCBs, and ΣDL-PCBs concentrations and negatively correlated with the BDE-209 and Σ10PBDEs concentrations in the samples from Nanning. The TSH concentrations correlated with the PCB-118 concentrations and negatively correlated with the BDE-209 and Σ10PBDEs concentrations in the samples from Nanning. In summary, we did not find any consistent correlations between the TH and analyte concentrations in the two study regions.
The analyte and TH concentration data were log2 transformed and then stepwise multiple linear regressions were performed. It can be seen from Table 2 that the ΣPBDEs concentrations did not correlate well with the TH concentrations in the samples from Weifang despite the high Σ10PBDEs concentrations that were found in the serum samples. Correlations between TH and PBDEs concentrations in blood have also not been found in several previous studies [71-73]. Specifically, Zhang et al. (2010) did find non-significant correlations between PBDE concentrations and total T3, T4, and TSH concentrations in blood samples from Chinese infants living in an electronic waste recycling area in southeast China [69].
The log2ΣDP concentrations significantly correlated and the log2BDE-99 concentrations significantly negatively correlated with the T3 concentrations in the samples from Weifang, and the log2BDE-153 concentrations significantly correlated and the log2Σ10PBDEs concentrations significantly negatively correlated with the T3 concentrations in the samples from Nanning. The negative correlation between the log2Σ10PBDEs and T3 concentrations in the samples from Nanning were consistent with the results of a previously published study [74], in which T3 concentrations were found to be negatively correlated with PBDE concentrations (the sum the concentrations of BDE-47, -99, -100, and -153) in 308 adult male sport-fish consumers. The positive correlation between the log2BDE-153 and T3 concentrations in the samples from Nanning were consistent with the results of another study [25]. However, the negative correlation we found between the log2BDE-99 and T3 concentrations were inconsistent with the positive correlation that has previously been found [75]. It should be noted that positive correlations between the log2ΣDP and T3 concentrations and the log2anti-DP and fT3 concentrations were found in the samples from Weifang. No studies of the relationships between DP TH concentrations have been performed before, so this is the first time a relationship between DP and TH concentrations has been reported.
As mentioned above, a positive correlation was found between the log2anti-DP and fT3 concentrations in the samples from Weifang for the first time. However, the log2PCB-118 concentrations positively correlated and the log2PCB-170 concentrations negatively correlated with the fT3 concentrations in the samples from Nanning. The positive correlation between the log2PCB-118 and fT3 concentrations in the samples from Nanning is inconsistent with the results of previous studies [76,77]. A negative correlation between the T4 and log2ΣDL-PCBs concentrations was found in the samples from Weifang while a positive correlation between the T4 with log2PCB-156 concentrations was found in the samples from Nanning. Negative correlations between T4 and total PCB concentrations have been reported previously [76]. No significant correlations were found between the analyte and fT4 concentrations in the samples from Weifang, but negative correlations were found between the fT4 and log2ΣDL-PCBs and log2BDE-190 concentrations in the samples from Nanning. A negative correlation between total PCB and fT4 concentration in samples from 334 pregnant women in California, USA has been reported previously [78]. Our results for the samples from Nanning showed the same negative correlation between the total PCB and TH concentrations. No significant correlations were found between the analyte and the TSH concentrations in the samples from Weifang. However, negative correlations between the log2Σ10PBDEs and TSH concentrations were found in the samples from Nanning. Non-significant relationships between PBDE and TSH concentrations have been found in most previous studies of human blood, and only two have shown a negative correlation between the TSH and PBDE concentrations in serum. These negative correlations were found in samples from 110 men who consumed fish from the Baltic Sea [79] and 405 men who consumed fish from the North American Great Lakes [74].
In summary, the correlations between the analyte and TH concentrations in the samples from Weifang and Nanning showed different trends. It has been reported in other publications that TH concentrations could correlate positively or negatively with PBDE or PCB concentrations, and the actual relationship may be dependent on the exposure level, population characteristics, the specific congeners that are measured, and other study characteristics [79-83].
Conclusion
In this study, we acquired data on PBDE, PCB, and DP concentrations in serum samples from the residents of Weifang and Nanning. This study is unique in that we have measured 30 individual PBDE, PCB, and DP congeners and all five types of TH in serum collected at the same time from residents of an area in which PBDEs are produced (Weifang) and an area in which they are not produced (Nanning). The PBDE, PCB, and DP concentrations were significantly higher in the samples from Weifang than in the samples from Nanning. We found that even low PBDE, PCB, and DP concentrations could influence TH concentrations in the samples from Nanning. The correlations between the analyte and TH concentrations followed different trends in the samples from Weifang and Nanning. High PBDE concentrations found in serum samples from Weifang did not significantly correlate with TH concentrations, but the PBDE concentrations found in serum samples from Nanning correlated with the TH concentrations. It is difficult to conclusively determine any causal relationships between exposure to PBDEs and adverse effects on the thyroid gland. However, the results of this study clearly show that the concentrations of several of the pollutants that were studied consistently correlated with the thyroid function, and these relationships were similar to relationships that have been found in several epidemiological studies and studies on animals. The body burdens of DP, PBDEs, and PCBs in humans might influence circulating TH concentrations in the human body, and this should be studied carefully in the near future.
Acknowledgements
This research project was supported by the Program for New Century Excellent Talents in University (NCET-10-0106), a 985 Engineering Project (MUC985-9), and the Institution of Higher Education Innovation talent recruitment program (111 Program, B08044), Weifang Binhai People’s hospital and Yudong hospital in Nanning.
References
- Brouwer A, Blaner WS, Kukler A, et al. Study on the mechanism of interference of 3,4,3′,4′-tetrachlorobiphenyl with the plasma retinol-binding proteins in rodents. Chemico-Biological Interactions. 1988;68:203-17.
- Ott MG, Zober A, GermannC. Laboratory results for selected target organs in 138 individuals occupationally exposed to TCDD. Chemosphere. 1994;29:2423-37.
- Triebig G, Werle E, Papke O, et al. Effects of dioxins and furans on liver enzymes, lipid parameters, and thyroid hormones in former thermal metal recycling workers. Environmental Health Perspectives. 1998;106:697-700.
- Wei GL, Li DQ, Zhuo MN, et al. Organophosphorus flame retardants and plasticizers: sources, occurrence, toxicity and human exposure. Environ Pollut. 2015;196:29-46.
- Lin SM. Negative associations between PBDE levels and thyroid hormones in cord blood. International Journal of Hygiene & Environmental Health. 2011;214:115-20.
- Darnerud PO, Eriksen GS, Jóhannesson T, et al. Polybrominateddiphenyl ethers: occurrence, dietary exposure, and toxicology. Environmental Health Perspectives. 2001;109:49-68.
- de Wit CA, Herzke D, Vorkamp K. Brominated flame retardants in the Arctic environment- trends and new candidates. Science of the Total Environment. 2010;408:2885-2918.
- Hoh E, Zhu, Hites RA. Dechlorane Plus, a Chlorinated Flame Retardant, in the Great Lakes. Environmental Science & Technology. 2006;40:1184-89.
- Birgul A, Katsoyiannis A, Gioia R, et al. Atmospheric polybrominateddiphenyl ethers (PBDEs) in the United Kingdom. Environmental Pollution. 2012;169:105-11.
- Wu F, Guo J, Chang H, et al. Polybrominateddiphenyl ethers and decabromodiphenylethane in sediments from twelve lakes in China. Environmental Pollution. 2012;162:262-68.
- Shaw SD, Berger ML, Weijs L, et al. Tissue-specific accumulation of polybrominateddiphenyl ethers (PBDEs) including Deca-BDE and hexabromocyclododecanes (HBCDs) in harbor seals from the northwest Atlantic. Environment International. 2012;44:1-6.
- Desforges J-PW, Ross PS, Loseto LL. Transplacental transfer of polychlorinated biphenyls and polybrominateddiphenyl ethers in arctic beluga whales (Delphinapterusleucas). Environmental Toxicology and Chemistry. 2012;31:296-300.
- Fujii Y, Ito Y, Harada KH, et al. Regional variation and possible sources of brominated contaminants in breast milk from Japan. Environmental Pollution. 2012;162:269-74.
- Hedley AJ, Hui LL, Kypke K, et al. Residues of persistent organic pollutants (POPs) in human milk in Hong Kong. Chemosphere. 2010;79:259-65.
- Kim J, Kang J-H, Park H, et al. Assessment of polybrominateddiphenyl ethers (PBDEs) in serum from the Korean general population. Environmental Pollution. 2012;164:46-52.
- Miller MF, Chernyak SM, Domino SE, et al. Concentrations and speciation of polybrominateddiphenyl ethers in human amniotic fluid. Science of the Total Environment. 2012;417-418: 294-98.
- Schecter A, Colacino J, Sjödin A, et al. Partitioning of polybrominateddiphenyl ethers (PBDEs) in serum and milk from the same mothers. Chemosphere. 2010;78:1279-84.
- Schecter A, Päpke O, Tung KC, et al. Polybrominateddiphenyl ether flame retardants in the U.S. population: current levels, temporal trends, and comparison with dioxins, dibenzofurans, and polychlori-nated biphenyls. Journal of Occupational and Environmental Medicine. 2005;47:199-211.
- Sjödin A, Jones RS, Focant JF, et al. Retrospective time-trend study of polybrominateddiphenyl ether and polybrominated and polychlorinated biphenyl levels in human serum from the United States. Environmental Health Perspectives. 2004;112:654-58.
- Costa LG, Giordano G. Developmental neurotoxicity of polybrominateddiphenyl ether (PBDE) flame retardants. NeuroToxicology. 2007;28:1047-67.
- Darnerud PO, Aune M, Larsson L, et al. Plasma PBDE and thyroxine levels in rats exposed to Bromkal or BDE-47. Chemosphere. 2007;67:386-92.
- Talsness CE. Overview of toxicological aspects of polybrominateddiphenyl ethers: A flame-retardant additive in several consumer products. Environmental Research. 2008;108: 158-67.
- Talsness CE, Andrade AJM, Kuriyama SN, et al. Components of plastic: experimental studies in animals and relevance for human health. Philosophical Transactions of the Royal Society B: