Research Article - Journal of Public Health and Nutrition (2022) Volume 5, Issue 4
Nutritional aspects of autism spectrum disorder-Case study of the role of folate.
Lam DMK1, Lee MKK2, Luo Y, Kiu G1,Ayoub G1,3*
1World Eye Organization, 1209 Shui on Centre, 6 Harbour Road, Hong Kong
2School of Professional and Continuing Education, University of Hong Kong, Hong Kong
3Department of Psychology, University of California, Santa Barbara, California 93106 USA
- Corresponding Author:
- Ayoub G
Department of Psychology
University of California
Santa Barbara California 93106 USA
E-mail: ayoub@psych.ucsb.edu
Received: 30-Mar-2022, Manuscript No. AAJPHN-22-59086; Editor assigned: 31-Mar-2022, Pre QC No. AAJPHN-22-59086(PQ); Reviewed: 21-Apr-2022, QC No. AAJPHN-22-59086; Published: 28-Apr-2022, DOI: 10.35841/aajphn- 5.4.120
Citation: Lam DMK, Lee MKK, Luo Y, et al. Nutritional aspects of autism spectrum disorder– Case study of the role of folate. J Pub Health Nutri. 2022;5(4):120
Abstract
Autism Spectrum Disorder (ASD) is a developmental disability that can create significant behavioral and communication challenges. The prevalence of ASD among children at 8 years of age is approximately 2%, and the prevalence is similar across ethnic groups and countries. Studies have shown that the majority of ASD children develop autoantibodies to the high-affinity folate receptor in response to a dietary component. This Folate Receptor Antibody (FRA) blocks transport of folate across the Blood-Brain Barrier (BBB), resulting in a Cerebral Folate Deficiency (CFD). In clinical trials, these ASD children showed improvement in communication when placed on a daily supplement of folate in its reduced form, which can enter the brain via low-affinity transport. Here, we report that nutritional modification combined with psychotherapy in ASD children can partially overcome this CFD, reducing ASD symptoms. Our study indicates that nutritional treatment of CFD with reduced folate in children at the earliest stages may be most productive in limiting long-term ASD symptoms.
Keywords
ASD, Folate, Autism, Clinical.
Introduction
Vitamin B-9, folate, is necessary for normal nervous system development [1,2]. This vitamin is present in bio-available form in many foods, including legumes, leafy greens, and fruits. It is necessary for neural tube formation and closure in the human embryo and plays an essential role throughout fetal brain development [3-5]. Insufficient folate can result in spina bifida in the newborn, and is implicated in other developmental conditions [6,7]. To reduce these complications, prenatal vitamins contain Vitamin B-9, typically in its stable, oxidized form of folic acid. In contrast, natural foods contain the reduced form of folate. Most individuals can convert sufficient folic acid to folate. However, about 10% of the population has a limitation in absorbing folate into the brain due to the presence of an autoantibody to the folate receptor [8].
This Folate Receptor Antibody (FRA) blocks the high affinity FR, preventing folate from crossing the blood brain barrier [8,9]. Clinical studies have shown that blood levels of the FRA can be reduced by changes in diet, particularly the elimination of dairy products [10]. If levels of FRA are reduced or eliminated, this would thus allow sufficient folate to enter the brain.
While FRA is present in a small minority of the population, clinical tests find that about 70% of Autism Spectrum Disorder (ASD) children have FRA [11]. Meta-analysis of multiple studies has shown that ASD children were nearly 20 times more likely to have FRA, [12] pointing to a potential genetic component that restricts brain absorption of Vitamin B-9. While FRA can be reduced with significant change in diet, [10] a symptom of ASD is resistance to dietary change. Thus, the dietary factors contributing to FRA are self-perpetuating due to this change resistance, as FRA causes a reduction of folate entering the brain. Those with the FRA have a reduced amount of folate reaching their brain, creating Cerebral Folate Deficiency (CFD). This CFD cannot be countered by supplementation with folic acid, the synthetic form of the B-9 vitamin, but trials have revealed that CFD may be alleviated by supplementation with elevated levels of the natural version of folate, in the form of methyl-folate or folinic acid [8,9,13].
In multiple clinical trials with ASD children, of the children who have FRA, their ASD related communication symptoms are alleviated when given daily folate supplements (in the form of folinic acid) for three months [14,15]. Additional trials are underway to assess the use of L-folinic acid (L-Leucovorin) or methyl-folate as the folate sources and the optimal daily dosage. But it is now clear that supplementation with reduced folate can help overcome CFD.
Once CFD is reduced or eliminated, it would be possible to work to change the diet of the ASD children, in order to reduce production of FRA. This may provide an optimal method to reduce ASD symptoms. Key in the dietary treatment is decreasing the agents that can stimulate FRA production and the provision of food sources of folate. These two things, elimination of autoantibody-stimulating foods and consumption of the natural form of folate (the reduced form), provide the key conditions for reducing severity of ASD symptoms.
An important aspect of FRA contributing to CFD is the finding that this may start very early in development. This was confirmed by Ramaekers, [9,16] who documented that when either or both parents have FRA, the chance of their child being diagnosed ASD several years later is higher. It thus seems that elevated risk of ASD in a child is related to elevate prenatal FRA, particularly when the FRA is present in the mother, but also when it is present in the father. One study has found FRA presence in autistic children was 75.6%, with the FRA prevalence 34% in their mothers and 29% in their fathers, as compared to 3% FRA positivity in healthy controls [17]. Another report found FRA prevalence of 76% in autistic children, 75% in unaffected siblings, 69% in fathers and 59% in, while the prevalence of this FRA in unrelated normal controls was 29% [18].
These findings are consistent with the presence of critical periods in development [19]. The presence of critical periods, first shown for the visual system, is the developmental time when sensory stimulation is necessary for visual perception to become established. If an animal is deprived of the sensory input during the critical period, the functional perception of visual images is impaired throughout its life [19,20]. Similarly, depletion of cerebral folate during an infant’s fetal development may be similar to a critical period that could lead to increased severity of autism. We postulate that ASD may result from a modified critical period of some type, where CFD impairs neurological development of certain contacts, increasing the probability of a later development of ASD if such conditions prevail.
In pregnancy, the presence of FRA in the mother blocks folate delivery to the developing fetus. These FRAs are common in pregnancies that have births with spina bifida and ASD [6]. Identifying pregnancies where FRA is present and treating with folinic acid or methyl-folate [21] should permit sufficient folate to reach the fetus, in turn lowering the risk of developing ASD and other developmental disorders [22].
There may be an additional advantage to diagnose FRA in prospective parents. A recent report documented that Vitamins B9 and B12 supplementation increase pregnancy and live birth in women who have experienced difficulty in conceiving, [21] indicating an essential role of these vitamins in pregnancy and healthy fetal development.
The clinical studies to date have shown that when FRA is present, supplementation with folate and B12 can overcome the FRA and allow sufficient folate for a child’s brain development to continue its normal course [11, 15]. We here report our ongoing work using nutritional changes and psychotherapy to alleviate ASD symptoms in children. Our studies began prior to the documented use of methyl-folate for FRA and we assessed ASD with standard psychological tests rather than folate receptor autoantibody tests. Our treatment used Cognitive-Behavioral Therapy (CBT) and vitamin supplementation. Using such treatment was productive of reducing ASD in over 70% of the children, ages 3-11, as measured with standard clinical assessments.
Our finding is that ASD children of ages 3-11 had improvement in their verbal communication skills over the course of 6 months with use of CBT and vitamin supplementation. We based the vitamin supplementation on blood analysis of circulating vitamins, adding Vitamins A, folate, B12,C,B,E,D, along with DHA and Calcium to bring each child’s level of these vitamins to appropriate levels.
Methods
Families with ASD children were recruited at the Innate Potential Decode Center (Hong Kong). Assessment of the child’s communication skill was made using a parent/caregiver assessment tool, the CARS2-QPC. Responses to sections 1 (how the subject communicates with others) and 2 (how the subject expresses them) were tallied to make the assessment of the child’s communication skill as reported by the parent/ caregiver. The data presented have three categories of severity in communication: No Problem, Mild/Moderate, and Severe. All graphs use these three levels, and they are depicted on the vertical axes as equidistant. This is to depict qualitative data in a manner that can be visually reviewed. The CARS2-QPC was administered prior to the child entering CBT and nutritional supplementation and re-administered after 3 months and again after 6 months. The data presented from these tallied scores are in each figure.
Treatment of children followed a consistent process for each child. In our evaluation, phenotypic testing for innate brain structure and blood tests (Vitas, Oslo) were used to learn of cognitive issues and nutritional deficiencies. Autistic children were put on a daily regimen of nutritional multivitamins to make up for deficiencies with Vitamins A-E. Each child received regular monthly psychotherapy using Cognitive-Behavioral Therapy (CBT). CBT is the most common psychotherapy used with ASD children, and our supplementation with nutritional multivitamins proved advantageous in finding improvement in communication. Given that the clinical trials have used folinic acid (Leucovorin) to ameliorate the impact of FRA, our findings are consistent with these trials and extend that utility by incorporating CBT with a nutritional supplement.
The data derived from the parent/guardian measure of communication are depicted in the figures. The numbers of children of each age are shown in the (Table 1).
| Age | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 |
|---|---|---|---|---|---|---|---|---|---|
| Boys | 4 | 9 | 13 | 13 | 12 | 17 | 9 | 6 | 8 |
| Girls | 2 | 4 | 1 | 2 |
Table 1. Age and Gender of children in study.
Results
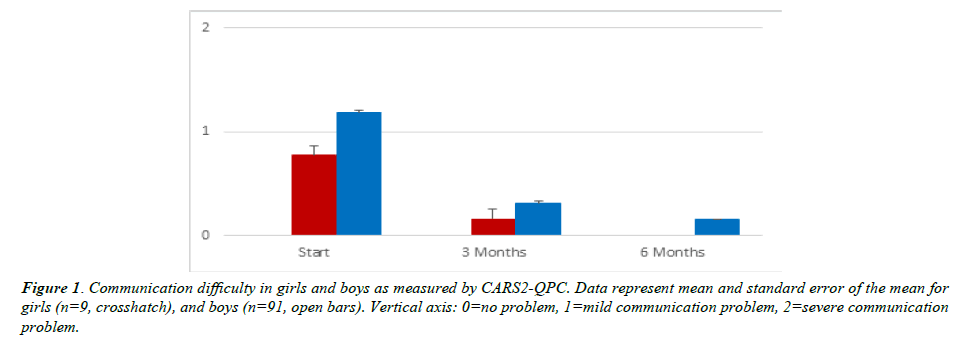
(Figure 1) is a depiction of the average rating of communication difficulty in girls and boys. Due to the small number of girls in the study, we have insufficient data to make a combined comparison by sex and age. The CARS2-QPC showed the average assessment of the girls was less than Mild/Moderate, while for boys it was greater than Mild/Moderate. Treatment for 3 months and for 6 months reduced the severity for all children, with 6 months showing a reduction to No Problem in communication for all the girls. Data depict the mean and standard error of the mean for girls (n=9) and boys (n=91).
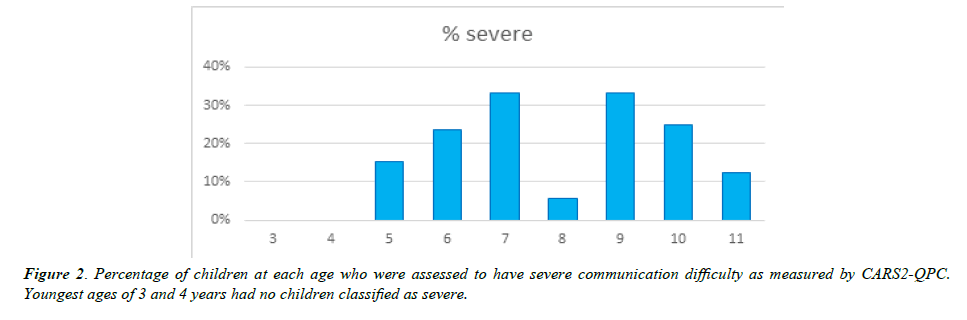
Due to the few girls in this study, the other figures combine results from boys and girls. (Figure 2) shows the percent of children at each age who were classified as having severe communication problems at the start of the study. As can be seen, no children under 5 were assessed as Severe, but by age 7, one third of the children had severe communication difficulty. This decreased for the older children, which may be related to a self-selection among parents in seeking treatment at that age. Most families will have sought treatment for an ASD child prior to age ten.
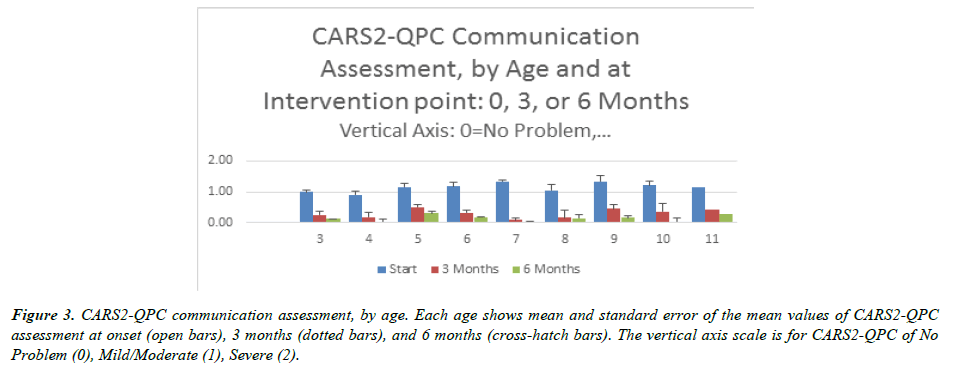
(Figure 3) displays the findings by age for all the children, with each bar cluster showing the assessment at the Start (blue), 3 Months (orange) and 6 Months (gray), and the clusters depicted by age of child at the start. All values are means for the age group, with error bars representing standard error of the mean.
Figure 3: CARS2-QPC communication assessment, by age. Each age shows mean and standard error of the mean values of CARS2-QPC assessment at onset (open bars), 3 months (dotted bars), and 6 months (cross-hatch bars). The vertical axis scale is for CARS2-QPC of No Problem (0), Mild/Moderate (1), Severe (2).
It is evident that at the youngest ages of 3 and 4 years, the children started with Mild/Moderate level of communication difficulty and the vast majority achieved No Problem in communication in the 6-month period of study. For ages 5 through 7, the average level started at greater than Mild/ Moderate communication difficulty, and was steadily reduced toward No Problem for a large fraction of the children. Ages 9 through 11 also started at greater than Mild/Moderate, and had a less significant improvement than the younger ages.
Our findings, while limited in number of children involved, suggest that age at the start of treatment is a key factor. Children over 5 start with greater communication difficulties, and have a smaller level of improvement, with children of 9 and above having the smallest benefit from the treatment. We believe this may be related to the ASD becoming more advanced and perhaps passing some critical periods in its progression that limit reversal of some symptoms.
(Table 2) shows the percentage of children in each age group that improved to the No Problem assessment in 3 and 6 months. Again, the youngest ages of 3 and 4 responded best to therapy overall, with all ages showing improvements in communication with our nutrition and CBT therapy routine.
| Age | 3 Months | 6 Months |
|---|---|---|
| 3 | 75% | 75% |
| 4 | 73% | 100% |
| 5 | 46% | 62% |
| 6 | 59% | 71% |
| 7 | 83% | 100% |
| 8 | 72% | 72% |
| 9 | 67% | 78% |
| 10 | 50% | 88% |
| 11 | 75% | 75% |
Table 2. Percentage of children at each age that showed improvement in the CARS2-QPC assessment of communication difficulty.
Discussion
Our findings show that nutritional supplementation of ASD children, coupled with CBT psychotherapy, and are effective in increasing communication skills in 2/3 to 3/4 of the children. This provides confirmation that the clinical trials treating ASD of children presenting with the folate receptor antibody [15,17] with pharmacological doses of folate can be replicated with nutritional modification and that inclusion of CBT therapy with nutritional supplementation increases the communication assessment of ASD children with Mild or Severe communication difficulties.
The youngest ages in our study, those under age 5, showed the least communication difficulty and were most readily reversed. This suggests that nutritional treatment of children at young ages (four and under) is most likely to reduce ASD symptoms. This finding leads us to propose that there is a type of critical period [19] during fetal development and early childhood that sets the stage for conditions such as ASD. We propose such a type of critical period, likely occurring in utero, does not necessarily produce ASD, but is a precondition for development of ASD during childhood.
A key factor in establishing a dysfunction-bias critical period appears to be folate. This vitamin is a methyl donor for DNA gene activation and regulation. Because transport across the blood brain barrier requires the folate receptor alpha to move folate efficiently, the presence of folate receptor autoantibodies during fetal and childhood development could be the agent that predisposes for neurological dysfunction, by creating a cerebral folate deficiency that leads to a bias for conditions such as ASD.
Our work shows this critical period dysfunction bias is reversible in some cases with use of appropriate nutritional treatment in the form of vitamins and the use of CBT psychotherapy at early ages. Hence, this is not a traditional critical period, but rather a critical period where preconditions are set that could permit dysfunctional development in early childhood, depending on availability of appropriate nutrition.
Conclusion
We suggest these modified critical periods in the development of ASD, where the first period is in utero and a later one is in the first five years of life. These periods can predispose a child to be more likely to develop ASD (in utero), and begin to set the conditions for ASD development (ages 3-5). Since nutritional supplementation along with CBT was effective for the children in this study, it suggests such therapy and nutrition should be provided in the first years of life to maximize positive outcomes. Additionally, other reports indicating that the presence of FRA can predispose a child toward ASD leads us to recommend that women presenting with FRA who may become pregnant are advised to take a prenatal supplement that includes a reduced form of folate (such as methyl-folate or folinic acid), and that children born to parents with FRA or children who have FRA would be advised to have nutritional supplementation to ensure sufficient levels of these vitamins for normal brain development.
References
- Alam C, Kondo M, O’Connor DL, et al. Clinical implications of folate transport in the central nervous system. Trends Pharmacol Sci. 2020;41(5):349-61.
- Reynolds E. Vitamin b12, folic acid, and the nervous system. The Lancet Neurology. 2006;5(11):949-60.
- Pitkin RM. Folate and neural tube defects. Am J Clin Nutr. 2007;85(1):285S-88S.
- Blom HJ, Shaw GM, den Heijer M, et al. Neural tube defects and folate: case far from closed. Nat Rev Neurosci. 2006;7(9):724-31.
- Bobrowski-Khoury N, Ramaekers VT, Sequeira JM, et al. Folate receptor alpha autoantibodies in Autism spectrum disorders: diagnosis, treatment and prevention. JPM. 2021;11(8):710.
- Imbard A, Benoist JF, Blom HJ. Neural tube defects, folic acid and methylation. Int J Environ Res Public Health. 2013;10(9):4352-89.
- Shapira I, Sequeira JM, Quadros EV. Folate receptor autoantibodies in pregnancy related complications. Birth defects research. Part A Clin Mol Teratol. 2015;103(12):1028-30.
- Frye RE, Rossignol DA, Scahill L, et al. Treatment of Folate Metabolism Abnormalities in Autism Spectrum Disorder. Semin Pediatr Neurol. 2020;35:100835.
- Ramaekers VT, Rothenberg SP, Sequeira JM, et al. Autoantibodies to folate receptors in the cerebral folate deficiency syndrome. N Engl J Med. 2005;352(19):1985-91.
- Ramaekers VT, Sequeira JM, Blau N, et al. A milk-free diet downregulates folate receptor autoimmunity in cerebral folate deficiency syndrome. Dev Med Child Neurol. 2008;50(5):346-52.
- Frye RE, Sequeira JM, Quadros EV, et al. Cerebral folate receptor autoantibodies in autism spectrum disorder. Mol Psychiatry. 2013;18(3):369-81.
- Rossignol DA, Frye RE. Cerebral Folate Deficiency, Folate Receptor Alpha Autoantibodies and Leucovorin (Folinic Acid) Treatment in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis. J Pers Med. 2021;11(11).
- Ramaekers VT, Blau N. Cerebral folate deficiency. Dev Med Child Neurol. 2004;46(12):843-51.
- Renard E, Leheup B, Guéant-Rodriguez RM, et al. Folinic acid improves the score of Autism in the EFFET placebo-controlled randomized trial. Biochimie. 2020;173:57-61.
- Frye RE, Slattery J, Delhey L, et al. Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial. Mol Psychiatry. 2018;23(2):247-56.
- Ramaekers VT, Blau N, Sequeira JM, et al. Folate receptor autoimmunity and cerebral folate deficiency in low-functioning autism with neurological deficits. Neuropediatrics. 2007;38(6):276-81.
- Ramaekers VT, Sequeira JM, DiDuca M, et al. Improving Outcome in Infantile Autism with Folate Receptor Autoimmunity and Nutritional Derangements: A Self-Controlled Trial. Autism Research and Treatment. 2019; 2019:7486431.
- Quadros EV, Sequeira JM, Brown WT, et al. Folate receptor autoantibodies are prevalent in children diagnosed with autism spectrum disorder, their normal siblings and parents. Autism Research. 2018;11(5):707-12.
- Wiesel TN. Nobel Lecture. NobelPrize.org
- Wiesel TN, Hubel DH, Lam DMK. Autoradiographic demonstration of ocular-dominance columns in the monkey striate cortex by means of transneuronal transport. Brain Res. 1974;79(2):273-79.
- Cirillo M, Fucci R, Rubini S, et al. 5-Methyltetrahydrofolate and Vitamin B12 Supplementation Is Associated with Clinical Pregnancy and Live Birth in Women Undergoing Assisted Reproductive Technology. Int J Environ Res Public Health. 2021;18(23):12280.
- Levine SZ, Kodesh A, Viktorin A, et al. Association of Maternal Use of Folic Acid and Multivitamin Supplements in the Periods Before and During Pregnancy With the Risk of Autism Spectrum Disorder in Offspring. JAMA psychiatry. 2018;75(2):176-84.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref