Case Report - Archives of Digestive Disorders (2017) Archives of Digestive Disorders (Special Issue 2-2017)
Neuroendocrine cell hyperplasia in ulcerative colitis and lymphocytic colitis.
Arevalo Fernando1,2*, Zurita Fiorella1, Vergara Greys1, Ramirez Rocio1, Castillo Joseph1,2, Monge Eduardo1,2,31Hospital Nacional Daniel A Carrion, Guardia Chalaca 2176, Bellavista 07016, Peru
2Universidad Nacional Mayor de San Marcos, Lima, Peru
3Universidad Peruana de ciencias aplicadas, Distrito de Lima 15023, Peru
- *Corresponding Author:
- Arevalo Fernando
Hospital Nacional Daniel A Carrion
Guardia Chalaca 2176
Bellavista 07016
Peru
Tel: 980624225
E-mail: histodiagnostico1303@hotmail.com
Accepted Date: May 03, 2017
Citation: Fernando A, Fiorella Z,Greys V, et al. Neuroendocrine cell hyperplasia in ulcerative colitis and lymphocytic colitis. Arch Dig Disord. 2017;1(2):1-3.
Abstract
Some authors have found increase of neuroendocrine cells in microscopic colitis and ulcerative colitis. The aim of this study is to evaluate the presence of neuroendocrine cells in ulcerative colitis, lymphocytic colitis and collagenous colitis. Immunohistochemistry was performed to identify neuroendocrine cells through marker chromogranin A (CgA). The study included 10 cases of Lymphocytic colitis, 10 cases of collagenous colitis, 10 cases of ulcerative colitis and 10 controls. There was statistically significant difference in the count of neuroendocrine cells, between lymphocytic colitis and control (p=0.019104), and between ulcerative colitis and controls (p=0.0077). In Collagenous colitis there was an increase in neuroendocrine cells but we failed to find statistical differences. In conclusion similar to reported by other authors, we could observe neuroendocrine cell hyperplasia in lymphocytic colitis and ulcerative colitis compared with controls.
Keywords
Colitis, Chromogranin A, Neuroendocrine cells, Serotonin
Introduction
Neuroendocrine cells are located throughout the gastrointestinal mucosa and play different roles according to the molecules they produce. In the colon, these cells are called enterochromaffin cells and are capable of releasing serotonin. Serotonin exerts a wide range of effects in the gut, largely due to the presence of multiple receptor subtypes that are present on smooth muscle, enteric neurons, enterocytes and immune cells [1].
There is general agreement that serotonin release could stimulate motility and secretion. An increase of serotonin could lead to increased motility and secretion, and therefore diarrhea and abdominal pain. Many authors have studied the presence of these cells in patients with diarrhea. For example, Kim et al. showed an increase of serotonin and PYY containing neuroendocrine cells in a small group of patients with post infectious irritable bowel syndrome compared to controls [2]. We previously studied the presence of enterochromaffin cells in irritable bowel syndrome, but we could not find significant differences compared to controls. It should be noted that our study was limited by the use of histochemical (silver stain) in the study of enterochromaffin cells instead of immunohistochemistry markers [3].
Neuroendocrine cells and Serotonin release seems to be closely related to inflammation. It has been demonstrated that colitis induction in mice can cause enterochromaffin cells hyperplasia [4].
Microscopic colitis is a common cause of chronic diarrhea, characterized by normal endoscopic findings. Histologically, it comprises 2 subtypes: lymphocytic colitis, characterized by an increase of intraepithelial lymphocytes in colon mucosa and collagenous Colitis characterized by a thickened collagen band underneath the surface epithelium.
The etiology and pathophysiology of microscopic colitis is not well understood but is likely to be multifactorial, involving mucosal immune responses to luminal factors in a genetically predisposed individual [5].
In lymphocytic colitis, El Salhy et al. have reported neuroendocrine cell hyperplasia. The authors propose that this could explain, at least partially the chronic diarrhea in these patients [6]. Ulcerative colitis, in turn, is a chronic, relapsing inflammatory disease of the colon, characterized endoscopically by diffuse continuous involvement of the mucosa and histologically by crypt distortion and basal lymphoplasmocytosis In ulcerative colitis, some authors have also found an increase in enterochromaffin cells [7], which could be related to a later development of neuroendocrine neoplasia.
The aim of this study was to evaluate the presence of neuroendocrine cells in 3 types of colitis: ulcerative colitis and lymphocytic colitis and collagenous colitis.
Case Presentation
The study included 10 cases of Lymphocytic colitis, 10 cases of collagenous colitis and 10 cases of ulcerative colitis. Histological slides were reviewed by 3 different pathologists. All selected cases fulfilled diagnostic histologic criteria. 10 Controls were selected among patients with minimal inflammation, which did not fulfilled criteria for specific colitis.
The following diagnostic criteria were defined:
Lymphocytic colitis
Colonic mucosa with increased number of intraepithelial lymphocytes (more than 20 lymphocytes per 100 epithelial cells) on well-oriented biopsy specimens, accompanied by epithelial damage without crypt distortion [5].
Collagenous colitis
Colonic mucosa with thickness of the collagen band that exceed 10 mm (normal, <3 mm) on well-oriented biopsy specimens, accompanied by epithelial damage without crypt distortion [5].
Ulcerative colitis
Colonic mucosa with crypt distortion and basal lymphoplasmocytosis [8]. Immunohistochemistry was performed to identify neuroendocrine cells through a marker chromogranin A (CgA). The avidin-biotin complex / immunoperoxidase method was used. The primary antibody used was a polyclonal rabbit for chromogranin A (Dako).
Count of neuroendocrine cells was obtained from the average of the number of positive chromogranin A cell in 3 high power fields (40x) with higher density of these cells. The analysis of the data was done using the student T test for comparison of means. We used the epi info cdc software for our analysis, we considered a p value of less than 0.05 as significant.
Results
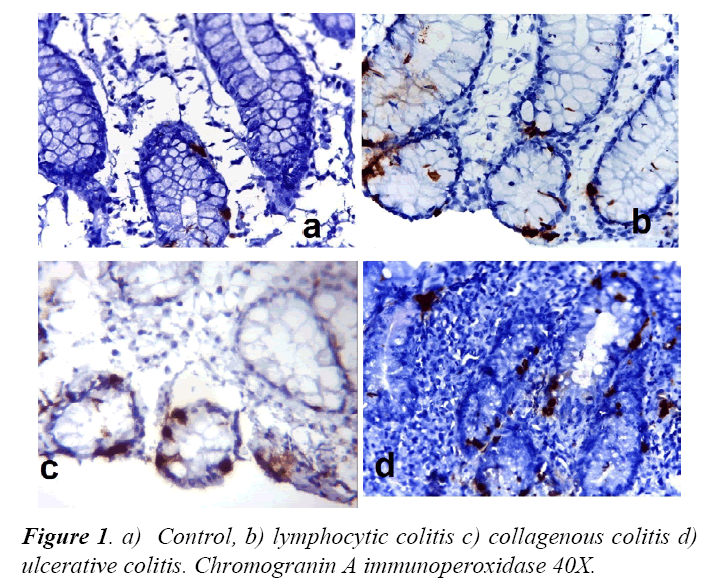
We included 10 patients of lymphocytic colitis, 10 with collagenous colitis, 10 with ulcerative colitis and controls. All patients fulfilled the diagnosis criteria required for pathological diagnosis. Chromogranin-A imunohistochemistry show positivity for neuroendocrine cells in control and patients, with differences in cell density. Figure 1, displays differences in the density of neuroendocrine cells in control, lymphocytic colitis and ulcerative colitis.
There was statistically significant difference in the count of neuroendocrine cells, in both lymphocytic colitis and ulcerative colitis with respect to controls. Table 1 shows cell count and the differences found.
Table 1. Mean of chromogranin cells count in lymphocytic colitis, collagenous colitis and ulcerative colitis.
| Types of colitis | Chromogranin positivie cells mean |
Chromogranin positivie cells Extreme values |
p* |
|---|---|---|---|
| Control | 5.66 | 3-11 | |
| Lymphocytic colitis | 8.5 | 4-14 | 0.019 |
| Collagenous colitis | 6.8 | 4-9 | 0.2 |
| Ulcerative colitis | 12.18 | 2-21.3 | 0.007 |
*with respect to controls.
Discussion
Our results show an increase of neuroendocrine cells (hyperplasia) in lymphocytic colitis and ulcerative colitis, similar to what has been reported by other authors.
We found a significant increase of chromogranine A positive cells in our patients with lymphocytic colitis. El Salhy et al. showed a significant increase of serotonin neuroendocrine cells and PYY neuroendocrine cells in 57 patients with lymphocytic colitis [6]. Unlike the El Salhy study, we only use chromogranin A to study neuroendocrine cells. Chromogranin A is an acidic glycoprotein expressed in the secretory granules of most normal and neoplastic neuroendocrine cell types.
In our study, the difference of neuroendocrine cells in ulcerative colitis compared with control was signicant. Chojnaki et al. also could demonstrate in 30 patients with ulcerative colitis a significant increase of neuroendocrine cells. According to those authors, the increase of neuroendocrine cells in ulcerative colitis is not only related to increase of serotonin release. Melatonin, an hormone synthetized from serotonin, could exerts a beneficial anti-inflammatory effect in ulcerative colitis [7].
Some authors believe carcinoid tumors and neuroendocrine neoplasias other than carcinoids may also be associated with ulcerative colitis, suggesting that long-standing inflammation is directly responsible for the development of these kind of neoplasiaS [9].
In Collagenous colitis there was an increase in neuroendocrine cells but we failed to find statistical differences. One study showed in 12 patients with collagenous colitis higher values of cromogranin A and secretoneurin in feces compared to controls [10]. We did not find any histological or immunohistochemical study about the relationship between neuroendocrine cells and collagenous colitis.
Association between inflammation and neuroendocrine cells has been studied in the colon. Experimentally it has been demonstrated that neuroendocrine hyperplasia could be caused in trinitrobenzene sulfonic acid -induced colitis mice [11]. Other experimental studies have shown that neuroendocrine cell hyperplasia could be related to Il13 release. Using the nematode Trichuris muris model of colitis in Il 13 deficient mice, Manocha et al. demostrated that enterocromaffin cells count were lower in Trichuris muris–infected IL-13 deficient mice compared with the wild-type mice [12].
On the other hand, some studies have shown that serotonin may contribute to the initiation of intestinal inflammation. Experimental data suggest that colitis induced in tryptophan hydroxylase1-deficient mice, which have significantly reduced amounts of serotonin in the gut, is less severe compared to wildtype mice [13].
Further studies have demostrated that Serotonin increases the in vitro phagocytic activity of macrophages in a dose-dependent manner [14].
Conclusion
We could observe neuroendocrine cell hyperplasia in lymphocytic colitis and ulcerative colitis compared with controls. These findings highlight the close relationship between enterochromaffin cells and inflammation, and supports the idea of the existence of a neuroendocrine-immuno axis in gastrointestinal pathology. More data are required to provide better insight on this relationship.
References
- Manocha M, Khan WI. Serotonin and GI disorders: An update on clinical and experimental studies. Clin Transl Gastroenterol. 2012;3(4):e13.
- Kim HS, Lim JH, Park H, et al. Yonsei Increased immunoendocrine cells in intestinal mucosa of postinfectious irritable bowel syndrome patients 3 years after acute Shigella infection--an observation in a small case control study. Med J. 2010;51(1):45-51.
- Arévalo F, Aragon V, Montes P, et al. Monge increase of intraepithelial lymphocytes in patients with irritable bowel syndrome. E Rev Gastroenterol Peru. 2011;31(4):315-8.
- Wang H, Steeds J, Motomura Y, et al. CD4+ T cell-mediated immunological control of enterochromaffin cell hyperplasia and 5-hydroxytryptamine production in enteric infection. Gut. 2007;56(7):949-57.
- Munch A, Langner C. Microscopic colitis: Clinical and pathologic perspectives. Clin Gastroenterol Hepatol. 2015;13:228-236.
- El-Salhy M, Gundersen D, Hatlebakk JG, et al. High densities of serotonin and peptide YY cells in the colon of patients with lymphocytic colitis. World J Gastroenterol. 2012;18:6070-6075.
- Chojnacki C, Wiśniewska-Jarosińska M, Kulig G. Evaluation of enterochromaffin cells and melatonin secretion exponents in ulcerative colitis. World J Gastroenterol. 2013;21:3602-7.
- DeRoche TC, Xiao SY, Xiuli L. Histological evaluation in ulcerative colitis. Gastroenterology Report. 2014;2(3):178-192.
- Grassia R, Bodini P, Dizioli P, et al. Neuroendocrine carcinomas arising in ulcerative colitis: Coincidences or possible correlations? World J Gastroenterol. 2009;15(33):4193-4195.
- Wagner M, Stridsberg M, Peterson CG, et al. Increased fecal levels of chromogranin A, chromogranin B, and secretoneurin in collagenous colitis. Inflammation. 2013;36(4):855-61.
- Zang KH, Rao Z, Zhang GQ, et al. Anticolitis activity of chinese herbal formula Yupingfeng powder via regulating colonic enterochromaffin cells and serotonin. Indian J Pharmacol. 2015;47(6):632-7.
- Manocha M, Shajib MS, Rahman MM, et al. IL-13-mediated immunological control of enterochromaffin cell hyperplasia and serotonin production in the gut. Mucosal Immunol. 2013;6(1):146-55.
- Khan WI, Ghia JE. Gut hormones: emerging role in immune activation and inflammation. Clinical and Experimental Immunology. 2010;161(1):19-27.
- Freire-Garabal M, Núñez MJ, Balboa J, et al. Serotonin upregulates the activity of phagocytosis through 5-HT1A receptors. Br J Pharmacol. 2003;139(2):457-63.