Research Article - Journal of Fisheries Research (2021) Volume 5, Issue 5
Development and characterization of a gill cell line from striped catfish, Pangasianodon hypophthalmus, Sauvage (1878)
Mukunda Goswami1*, A. Sathiyanarayanan1, Nagpure N S 1 , Gireesh Babu. P1, Dhanjit Kumar Das2
1Department of Biotechnology, ICAR-Central Institute of Fisheries Education, Maharashtra, India.
2Department of Genetics, National Institute for Research in Reproductive Health, Maharashtra, India.
- Corresponding Author:
- Mukunda Goswami Department of Biotechnology, ICAR-Central Institute of Fisheries Education, Maharashtra, India E-mail: mukugoswami@gmail.com
Accepted: September 04, 2021
Citation:Goswami M, Sathiyanarayanan A, Nagpure NS, et al.. Development and characterization of a gill cell line from striped catfish,
Pangasianodon hypophthalmus, Sauvage (1878) J Fish Res 2021;5(5):11-15.
Abstract
susceptibility, cytotoxicity, gene expression studies etc. The striped catfish, Pangasianodon hypophthalmus is one of the main species in Asian aquaculture, especially in countries like Thailand, Indonesia, China, India, Bangladesh and Vietnam. The present study reports the development of a permanent cell line from gill of P. hypophthalmus designated as PHG and its application in toxicological research. Leibovitz’s-15 (L-15) cell culture medium supplemented with 15% fetal bovine serum (FBS) was used for the maintenance of cell line PHG. The morphology of the PHG cell line was fibroblastic in nature. PHG cells grew well at varied temperatures ranging from 24 to 300C with an optimum temperature of 280C. The PHG cell line was characterized using sequence of mitochondrial cytochrome C oxidase subunit I, which authenticated the species of origin of the cell line. The cell line was transfected with pEGFP-C1 plasmid and transfection reporter gene was successfully expressed 48 hr post-transfection with 9% transfection efficiency. The toxicity assessment of two organophosphate pesticides, chlorpyrifos and malathion using PHG cell line revealed that the two organophosphate pesticides were cytotoxic to the cell line at varied concentrations.
Keywords
Onuphidae, Shrimp aquaculture, Reproduction, Gonadal development, Western Pacific.
Introduction
Polychaetes are Annelids, which intervene in the structure and functionality of benthic communities, participate in the recycling and removal of sediment, and in the burial of organic matter are ecosystem engineers by regulating the community structure and the functioning of shallow water areas in addition to being considered a food source for benthic organisms and migratory shorebirds in tidal plains and of great ecological importance, since they dominate numerically and in biomass [1-5].
The diet of farmed shrimp and especially the reproductive ones are improved with the incorporation of polychaetes, to accelerate gonadal development and the intake of protein and high-energy lipids. At present, the use of polychaetes is not restricted to reproductive aspects since their use has been diversified in other types of aquaculture activities [6].
More specific studies on the Americonuphis reesei species highlight the ecological reproductive genetic contamination fishing aspects in particular energy morphometric and due to the nutritional value, that is linked to prostaglandins that induce the rapid maturation of penaeid shrimp [7-16].
Geographical Distribution
In Panama, the Onuphidae Family represented by eight species that highlights the Americonuphis genus, included within the Diopatra group [17]. It is restricted to the Northern Hemisphere of which has been found the species A. reesei in the Eastern Tropical Pacific, in the Gulf of Panama, in the intertidal zone of Chomes Beach, Costa Rica and outside the Bay of Jiquilisco, El Salvador while A. magna has been identified in the Caribbean [18-23].
Most of the studies of this organism have been carried out in shallow waters and the species come from the tidal zone [20].
In the case of A. reesei, investigations have been carried out on the systematics of these organisms in Panama and indicate that they inhabit rocky environments, reefs, and soft sandy bottoms and have been little studied [19].
Collection Areas
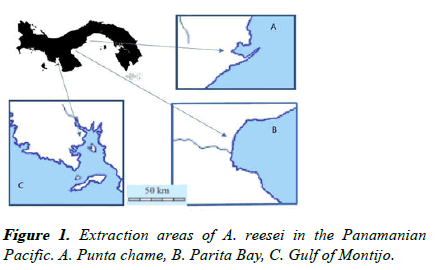
In the case of Panama, the collection and processing of polychaete A. reesei are regulated by Executive Decree 4 of 1997 where the extraction sites in the Gulf of Panama are declared, which have been expanded later to the Gulf of Montijo. Likewise, export processing plants are regulated by Resolution 3 of 2007, issued by the Panama Aquatic Resources Authority (ARAP) as shown in Figure 1 [24,25].
Description and Habitat
The polychaete A. reesei, of the Onuphidae family, a softbottomed tubeworm reaches sizes between 1.5 and 2.5 m with a defined anterior section and reduced antennae, with long jaws strongly developed as shown in Figure 2. It inhabits intertidal areas of sandy muddy beaches of the Central American Pacific, in U-shaped tubes, covered with sand and inorganic material with a length of up to 2 m, with both ends protruding to the outside up to 7 cm above the sediment as shown in Figure 3 [25,26].
Methods of Extraction
Collections are made in the intertidal zone on exposed banks at low tide to depths of 0.5 m.
The traditional collection method of the extractor crews consists of the introduction of a plastic tube connected to an air pump for bicycle tires, inserted in the front part of the polychaete cabin where air pressure is driven that expels the organism at the rear end (Figure 4) [27].
The annual extraction of these organisms is regulated through Resolutions of the Panama Aquatic Resources Authority (ARAP) issued every year, in which the monthly collection days are established, which coincide with the broadest syzygy tides of the country, month, of both moons, full and new, according to the time of year, and the annual closed periods of three months are declared, which vary between May to August, about to the amplitude of the tide. The closed periods established by law agree with the amplitude of the tide that does not favor the discovery of the polychaete banks indicate that points out that as the banks with the ebb of the tide, larger polychaetes with dark reddish coloration are extracted [27].
The extraction chronology begins in 1994 with two crews in the Aguadulce area in 1995. It increases to seven crews, including the area of Punta Chame and Darien. From October 1994 to May 1996 an extraction of 196 998 lb has been recorded at a rate of 10 lbs. man-day in 117 annual allowable days of collection, in periods of spring tides, whose market value was set between U.S. $ 1.00 and the U.S. $ 1.35 per pound [27].
Between 2007 and 2011, the annual catch ranged from 15 to 29 tons by the end of that period there were three polychaete processing plants in Panama, whose slaughter represented 7% of the total of products of marine origin processed in the country. However, currently only two plants operate [28,29].
Variable annual withdrawals are recorded with 10 588.00 kg in 2013, above the average of 5 411 kg. The catch per unit of effort was kept between 4 and 5 kg/trip /collector [12].
Even though the export statistics of the National Institute of Statistics and Census do not appear the polychaete item as an exportable product on the website Veritrade, of Information on foreign trade in Latin America and the world [30]. The company “Exportación Inversiones F.A.L. de Panamá S.A.” and Sea Technology, registered in the directory of exporters of fishery products of Panama [31]. It indicates the export of this product to Honduras [32,33].
According to the National Agriculture Research Institute, the capture of polychaetes from the natural environment is a negative aspect, not only for the reproduction of these organisms but also affects the biodiversity of species that live with them [34]. In El Salado beach the community associated with polychaete tubes is dominated by Mollusks, 94%, followed by polychaetes, 2%, unlike El Agallito beach, mollusks dominate in a 64%, followed by polychaetes, 27% and crustaceans 4.5% [35,36]. The Pacora area, in the Gulf of Montijo, shows the dominance of polychaetes, 79%, followed by Mollusks 20%, while in Tres Islas, mollusks represented 79% and polychaetes 13%, which indicates that the groups associated with polychaetes A. reesei vary according to the physical and oceanographic characteristics of each locality [37].
Importance of Polychaetes in Fisheries and Aquaculture
Polychaetes have been one of the benthic groups most used as indicators of contamination by organic matter, hydrocarbons, wastewater, and contamination in general [38,39]. The importance of the use of these organisms as bait for fishing, distributed in 18 families, however it was reported 27 families [40,41].
Other studies have highlighted the importance of these polychaetes in aquaculture, in this way cryopreservation techniques for larvae, photoperiod manipulation of the rearing time, and optimization of the growth process have been generated [42]. This author has even patented the artificial feeding methodology for detritivore polychaetes [43].
The aquaculture importance of Australonuphis sp., From the Ecuadorian coast, a group originally included in the genus Onuphis in Australia, which differs from the genus Americonuphis, due to morphological characteristics [44]. The Lumbrineris and Perineries genera have also been tested in experimental crops in Ecuador [45].
Perinereis shows promising characteristics to follow the investigations of the cultivation of these polychaetes, because it accepts better artificial food, shows less stress when handling, and is not very brittle, compared to Australonuphis, which is easily manipulated in addition to serving as a source of food for farmed shrimp Litopenaeus vannamei [46]. On the other hand, this polychaete, typical of the Ecuadorian coast, despite being abundant is the least suitable aquaculture species for cultivation because it is more fragile to handle. Draw attention to the fact that Perinereis nuntia from the natural environment has a greater quantity of fatty acids and eicosapentaenoic acid than those from the culture [47]. Another aquaculture species, such as Diopatra aciuculata, shows a dense-dependent relationship with growth, mortality percentage, and a higher number of fragmented posterior segments similar to the Australonuphis characteristic [48].
On the other hand, the use of culture waste from the flatfish Hypoglosus hypoglosus, in the feeding of Nereis virens, which shows high efficiency, is reflected in the number of fatty acids produced [49]. The growth and nutritional composition of polychaetes fattened with waste from fish farms were tested however Hediste diversicolor fed a diet for farmed fish, grows less [50].
The technical data sheet for polychaete A. reesei, made in Honduras by SUMACUA indicates that 100 g of this organism contains: 77.0% moisture, 7.2% ash, 2.2% lipids, 8.9% protein, 0.2% fiber, and 4.3% carbohydrate; in addition to micronutrients in concentrations of 455 ppm of calcium, 215 ppm of phosphorus, 97 ppm of magnesium, 355 ppm of potassium, 17 ppm of copper, 7.3 ppm of zinc and 116 ppm of iron. in Nereis virens, Perenereis cultrifera and Glycera sp. [51]. Species used in the feeding of culture organisms, present more humidity, carbohydrates and less ash and protein than A. reesei [52].
Relevant Studies on A. reesei in Panama
Energy substrates
Studies carried out show the presence of carboxylic fatty acids, prostaglandins: PGA+PGB, PGD, PGE, PGF, in the intestine, blood and the last two in the musculature of A. reesei, which are related to the development of gametes, also Arachidonic Acid (AA) and 12-HETE acid, precursors of prostaglandins, which become an element that accelerates the maturation of penaeid shrimp [16].
Regarding energy substrates, the presence of lipids between 1.5 to 3.4 g/100 g long-chain polyunsaturated fatty acids of type 20: 5w3 from total fat which does not vary throughout the year which it favors the synthesis of eicosanoids indicate, which could accelerate the rate of gonadal development of the shrimp that feed on them. The polychaetes have two cycles of higher protein concentration, the main peaks are observed between October and December, a time that coincides with the greatest gonadal development, which suggests the participation of proteins in the reproduction process [14].
Reproduction
Gonadal maturation is intraovarian, due to the scarce coelom of this species, with female gonads located in the wall of the first 20 metamers and male gonads in the next 30. Histological analysis shows gonadal development throughout the year; however, it is greater between October and December, which indicates the period of greatest reproduction which coincide with levels of oxygen consumption that vary from 220 to 320 g/ μL and it accelerates in the said period [53,54].
The larval stages in zooplankton samples analyzed to indicate the scarcity of these in samples from the distribution zones of A. reesei, which is indicative of the short period that these larvae remain in the water column, which suggests lecithotrophic organisms with the rapid larval settlement, or that they reproduce in the lunar period that does not coincide with that studied [55].
Genetic studies of A. reesei, carried out in the Gulf of Panama, present sequencing of 5.8s rDNA, with a clear close relationship between the populations that inhabit two areas in the Bay of Parita, whose extraction areas are separated by 25 km [9].
Physiological aspects
The detritivore diet reveals a high concentration of phytobenthos in the intestinal content which coincides with the diatomological flora found in the anterior part of the large intestine, however, selectivity is not shown in the consumption of these species, which constitute part of the food chain of the polychaete A. reesei [56]. This food source reported above and the granulometry of the medium allows A. reesei to build the U-shaped habitat, made with grains of sand, cemented by a mucilaginous substance, secreted by the same polychaete, which takes 6 h to produce construction [53]. These same authors observe a regeneration speed of the organism segments with growth between 2.40 and 3.50 cm/month, under laboratory conditions, a speed considered low.
Morphometry and biomass
The size and weight of polychaetes taken from El Agallito beach, an area with less extraction than El Salado, maybe the effect of capture pressure, since polychaetes, as a survival mechanism of the population, tend to reproduce at sizes smaller than usual [54]. In the Gulf of Montijo find banks with a density of 0.38 ind/m2, with an average length of 0.73 m and a weight of 32.21 g in the same place, report density in extraction period of 2.45 ind/m2 to 12.85 ind/m2, while in closed season 0.52 ind/ m2, to 1, 68 ind/m2, also indicate the average length of 1.06 m and weight of 40.45 g in extraction period and 0.94 m in length and 29 g of weight in the closed period [15]. It should be noted that the difference in size, weight, and density between periods is due to the relationship with the amplitude of the tide, which reveals a greater number of banks in the extraction season.
Pollution
It found trace metals in the musculature of polychaetes A. reesei, in extraction banks, adjacent to the mouth of rivers in Parita Bay, Panama, whose basin irrigates important areas of cultivation of sugar cane, rice, and cucurbits with average concentrations of Fe, 581, 98 μg.gˉ¹, followed by Mn, with 25.71 μg.gˉ¹, Zn, 16.05 μg.gˉ¹ and Cu, 3.52 μg.gˉ¹, whose values do not pose a danger, as they are below the minimum limits considered dangerous [10]. The previous samples conversion of organic and inorganic material is analyzed by atomic absorption spectrophotometry with flame atomization, analyzed by mathematical algorithms [57-62].
Conclusion
This review is framed in the impact of the use of polychaetes in aquaculture, especially Americonuphis reesei in the shrimp industry in Central America, principally in Panama. It must be paid attention to the difficulty of maintaining polychaete fisheries due to the scarce information on the natural history of the species, the lack of data regarding the impact of the capture of these organisms, and the inherent obstacles to biosecurity in export. Due to the high demand for polychaetes for use in aquaculture, Spain imports live polychaetes of the Asian species Perinereis aibuhitensis and the American Glycera dibranchiata, however, their introduction runs the risk of producing effects on biodiversity, which, as they are exotic species. On the other hand, there is a strong suspicion that the use of polychaetes in the shrimp industry could be the source of infection of diseases such as microsporidiosis and acute hepatopancreatic necrosis, as polychaetes are hosts, which would make them passive carriers of these diseases and the possibility of WSSV transmission to shrimp. The transmission of WSSV to Litopenaeus vannamei SPF in the presence of polychaetes of the genus Dendronereis.
In the case of A. reesei in Panama, the extraction in periods when the population is in a greater reproductive period responds to the capture season authorized by law, which is controversial with the studies reported about the life cycle. Everything seems to indicate that the capture for the commercial interest, during the periods of the greater amplitude of the monthly spring tide, is a premium over the protection of the resource, which should be guaranteed for sustainability over time. Due to the above described, it is suggested to change the closed period between October and December, months in which a higher frequency of reproduction has been demonstrated.
References
- Hutchings P. Biodiversity and functioning of polychaetes in benthic sediments. Biodivers Conserv. 1998; 7: 1133-45.
- Bruschetti M. Role of reef-building, ecosystem engineering polychaetes in shallow-water ecosystems. Diversity. 2019; 11: 168.
- Varadharajan D, Soundarapandian P. Contribution of polychaetes in feeding capability of commercially important crabs, southeast coast of India. J Mar Sci Res Dev. 2013; 3: 123.
- Iwamatsu S, Suzuki A, Sato M. Nereid polychaetes as the major diet of migratory shorebirds on the estuarine tidal flats of Fujimae-Higata in Japan. Zool Sci. 2007; 24: 676-85
- Fauchald K. The polychaete worms. Definition and keys to the orders, families and genera. Nat Hist Mus. 1997; 28:188.
- Olive PJW. Polychaete aquaculture and polychaete science: a mutual synergism. Hydrobiologia. 1999; 402: 175-93.
- Rojas RW, Vargas JA. Abundancia, biomasa y relaciones sedimentarias de Americonuphis reesei (Polychaeta: Onuphidae) en el Golfo de Nicoya, Costa Rica. Rev Biol Trop. 2008; 56 (S4): 59-82.
- Luna IG, Villalaz JR, López IE. Desarrollo gonadal del poliqueto Americonuphis reesei en las playas de El Agallito de Chitré y El Salado de Aguadulce (Onuphidae: Polychaeta). Scientia. 2001; 16(2): 21-8.
- Cano M, De León K, Ramos C, et al. Sequence analysis of 5.8s Rdna and internal transcriber spacers of the ribosomal region of Americonuphis reesei (Polychaeta: Onuphidae) in the Republic of Panama. First Report. Int J Genomics Proteomics. 2014; 7(1).
- Rodríguez D, Villalaz J, Goti I, et al. Metales trazas (Fe, Mn, Zn, Cu) en el poliquetoAmericonuphis reesei (Clase: Polychaeta), en la playa El Salado, Aguadulce, Panamá. Rev Mar Cost. 2020; 12(1): 57-72.
- Rojas R. Gestión de la pesquería de Americonuphis reesei Fauchald 1973 en el Golfo de Nicoya, Costa Rica: Recomendaciones para un rendimiento sostenible. Tesis Maestría, Universidad de Costa Rica. 2007; pp: 161.
- Vega A, Robles Y, Torres L. Análisis biológico y pesquero de la extracción del poliqueto Americonuphis reesei en el golfo de Montijo, Pacífico Panameño. Tecnociencia. 2014; 16(2): 77-93.
- Gómez JA, Murillo E, Villaláz JR, et al. Determinación de lípidos en el poliqueto Americonuphis reesei en las playas El Salado, Aguadulce y Agallito, Chitré. Scientia. 2001; 16(4): 37-44.
- Villalaz J, Gómez JA, Muñoz E. Determinación de proteínas en el poliqueto Americonuphis reesei en las playas El Salado, Aguadulce y Agallito en Chitré. Scientia. 2001; 16(2): 29-36.
- Rodríguez D, Goti I. Relaciones biométricas y sedimentarias de Americonuphiss reesei en períodos de extracción y veda, golfo de Montijo, Panamá. Vis Antataura. 2020; 4(2): 35-48.
- D’Croz L, Wong L, Justine G. Prostaglandins and related compounds from the polychaete worm Americonuphis reeseiFauchald (Onuphidae) as possible inducers of gonad maturation in Penaeid shrimps. Rev Biol Trop. 1988; 36(2): 331-2.
- Paxton H. Generic revision and relationships of the family Onuphidae (Annelida: Polychaeta). Rec Aust Mus. 1986; 38(1): 1-74.
- Beesley PL, Ross GJB, Glasby CUJ, et al. Polychaetes & allies: the southern synthesis. CSIRO publishing. 2000; pp:465.
- Fauchald K, Reimer AA. Clave de poliquetos panameños con la inclusión de una clave para todas las familias del mundo. Bol Inst Oceanogr Univ Oriente. 1975; 14(1): 71-94
- Rivera CG, Romero MY. Distribución de poliquetos (Annelida: Polychaeta) en la zona costera de El Salvador. Resultado del crucero de investigación R/V Urracá del Instituto Smithsonian de Investigaciones Tropicales. Tesis de Licenciatura, Universidad de El Salvador. 2002; pp: 68.
- Fauchald K. Polychaete from Central American sandy beaches. Bull South Calif Acad Sci. 1973; 72(1): 19-31.
- Andrade J, Charzeddine L, Cruz DLO, et al. Aspectos biométricos en el poliqueto tubícola Americonuphis magna Andrews (Annelida: Polychaeta) en la península de Araya, Venezuela. Mem Fund La Salle Cienc Nat. 1999; 59(151): 29-44.
- Delgado-Blas VH. Distribución espacial y temporal de poliquetos (Polychaeta) bénticos de la plataforma continental de Tamaulipas, Golfo de México. Rev Biol Trop. 2001; 49(1): 141-7.
- Gaceta oficial. Decreto Ejecutivo 4. Ministerio de Comercio e Industrias. Reglamenta la extracción, comercialización y exportación de poliquetos. Gaceta Oficial 23224. 1997.
- Gaceta oficial. Resuelto Autoridad de los Recursos Acuáticos de Panamá. Establece la Certificación para plantas de procesamiento de productos pesqueros no tradicionales. Gaceta Oficial 25946. 2007.
- Luna IG, Villalaz JR, López IE. Distribución de la biomasa y los parámetros morfométricos en las poblaciones del poliqueto Americonuphis reesei en las playas de El Salado de Aguadulce y El Agallito de Chitré. Scientia. 2001; 16: 53-64.
- Spadafora A. Evaluación preliminar de la extracción del recurso poliqueto, área de Aguadulce, meses de octubre, noviembre y diciembre. Informe Técnico, Ministerio de Comercio e Industrias. 1994.
- INEC. Cantidad y valor del desembarque de la pesca industrial, Artesanal y del cultivo de camarones en la república. 2011
- Valverde RA. Estudio sobre la producción pesquera industrial, comercialización y exportación, en la República de Panamá desde 1995 al 2008. Centros. 2012; 1(1): 116-147.
- INEC. Anuario de Comercio Exterior, Volumen II- Exportación: Año 2016.
- ARAP. Directorio de exportadores de pescados y mariscos de Panamá. 2017.
- VERITRADE SF. Información de comercio exterior de latinoamérica y el mundo.
- VERITRADE. SF. Comercio exterior importaciones y exportaciones de SEA TECHNOLOGY INC.
- IDIAP. El estado de la biodiversidad para la alimentación y la agricultura en Panamá. 2016; pp:265.
- Gómez JA, Herrera R, Ríos V, et al. Análisis del sedimento y organismos de la infauna de playa El Salado–Aguadulce. Tecnociencia. 2001; 3(1): 81-94.
- Villalaz JR, Vega C, Ávila Y, et al. Análisis temporal de macroinvertebrados bentónicos em la playa El Agallitio, Chitré. Tecnociencia. 2002; 4(2):111-26.
- Rodríguez D. Aspectos biométricos y relaciones sedimentarias de Americonuphis reesi en el Golfo de Montijo, Pacífico panameño. Tesis Licenciatura, Universidad de Panamá. 2018; pp: 36.
- Sivadas S, Ingole B, Nanajkar M. Benthic polychaete as good indicators of anthropogenic impact. Indian J Mar Sci. 2010; 39(2): 201-11.
- Fernández V, Londoño M. Poliquetos (Annelida: Polychaeta) como indicadores biológicos de contaminación marina: casos em Colombia. Gestión y Ambiente. 2015; 18(1): 189-204.
- Pombo A, Baptista T, Granada L, et al. Insight into aquaculture’s potential of marine annelid worms and ecological concerns: a review. Rev Aquac. 2018; 1-15.
- Cole VJ, Chick RC, Hutchings PA. A review of global fisheries for polychaete worms as a resource for recreational fishers: diversity, sustainability, and research needs. Rev Fish Biol Fish. 2018; 28: 543-65.
- Olive PJW. Polychaeta as a world resource: a review of patterns of exploitation as sea angling baits and the potential for aquaculture based. En Dauvin, JC, Laubeir, CL. y Reish, DJ. (edn). Actes de la 4ème Conférence Internationale des polychaètes. Mem Mus Hist Nat. 1994; 162: 593-603.
- US Patent. Patent No.: US 7,004,109 B2. Aquaculture of Marine Worms.2006.
- León-González JA, Cornejo-Rodríguez MH, Degraer S. A new species of Australonuphis (Polychaeta: Onuphiae) from the eastern Pacific. J Mar Biol Assoc. UK. 2008; 88(4): 739-42.
- Naranjo AP, Tobías FJ, Borroy RS, et al. Producción de poliquetos libes de enfermedades para su uso como alimento vivo en la industria camaronera de Ecuador. Aquacultura. 2019; 129: 44-8.
- Naranjo AP, Tobías FJ. Production of polychaetes for Ecuador’s shrimp industry. Global Aquaculture Alliance. 2019.
- Tehcprempreecha S, Khongchareonporn N, Chaicharoenpong C, et al. Nutritional composition of farmed and wild sandworms, Perinereis nutia. Anim Feed Sci Technol. 2011; 169: 265-9.
- Safarik M, Redden AM, Shreider MJ. Density-dependent growth of the polychaete Diopatra aciculate. Sci mar. 2006; 70(S3): 337-41.
- Brown N, Eddy S, Plaud S. Utilization of waste from a marine recirculating fish culture system as a feed source for the polychaete worms, Nereis virens. Aquaculture. 2011; 322-323: 177-83.
- Wang H, Seekamp I, Malzahn A, et al. Growth and nutritional composition of the polychaete Hediste diversicolor (OF Müller, 1976) cultivated on waste from land-based salmon smolt aquaculture. Aquaculture. 2019; 502: 232-41
- SUMACUA. Poliquetos (Americonuphis reesei), Ficha Técnica. Corporación de Suministros Acuícolas, Hondura .
- Bharath J, Priya A, Stella C. A comparative study on the nutritional value of three polychaete species used in shrimp aquaculture. Sustain Agr Food and Env Res. 2020; 8.
- Luna IG, López IE. Aspectos de la alimentación, regeneración y comportamiento de la construcción de los tubos del poliqueto Americonuphis reesei. Scientia. 2001; 16: 7-13.
- Luna IG, López IE. Comportamiento del consumo de oxígeno en Americonuphis reesei durante el período de estudio (Onuphidae: Polychaeta). Scientia. 2001; 16:15-19.
- Grimaldo M, Goti I, Trejos E. Comparación de dos comunidades zooplanctónicas del pacífico panameño. Scientia. 2001; 16(2): 65-72.
- Soler A, Pérez MI, Aguilar E. Fitobentos de las playas El Salado y Agallito. Scientia. 2001; 16(2): 69-79.
- Leone O, Tapia E, Fuentes MV, et al. Aplicación de la polarografía de pulso diferencial y redisolución anódica en la determinación de metales pesados Zn, Cd, Pb, Cu, en el río Matasnillo (Panamá). Scientia. 2017; 27(1):53-64.
- Cole VJ, Chick RC, Hutchings PA. A review of global fisheries for polychaete worms as a resource for recreational fishers: diversity, sustainability, and research needs. Rev Fish Biol Fish. 2018; 28: 543-65.
- Costa FP, Gil J, Passos AM, et al. The Market features of imported non-indigenous polychaetes in Portugal and consequent ecological concerns. In Sardá, R, San Martín, G, López, E, Martín, D y George D (edN). Scientific advances in polychaete research. Sci Mar. 2006; 70(S3):287-92.
- Desrina H. Verreth JAJ, Verdegem MCJ. Polychaetes as potential risks for shrimp pathogen transmission. Asian Fish Sci. 31S:155-67.
- Desrina H. On the role of the polychaete Dendronereis spp. In the transmission of white spot syndrome virus in shrimp ponds. Ph.D. Thesis, Wageningen University, pp:181.
- Haryadi D, Vereth JAJ, Verdegem MCJ, et al. Transmission of White Spot Syndrome Virus (WSSV) from Dendronereis spp. (Peters) (Nereididae) to penaeid shrimp. J Fis Dis. 38(5):419-28.