Research Article - Archives of General Internal Medicine (2025) Volume 9, Issue 5
Comparative efficacy of duloxetine, gabapentin, and pregabalin in long-term diabetic neuropathy management.
Athira Nair1*, Manoj Pallapothu2, Rithish Nimmagadda3, Vamsi Polepalli4, Anuja Nandanapu5, Shravya Edavaluru6, Juber Shaikh7
1Department of Surgery, Pontiac General Hospital, Michigan, United States
2Department of Neurology, Pontiac General Hospital, Michigan, United States
3,4Department of Internal Medicine, Maimonides Medical Centre, New York, United States
5Department of Internal Medicine, Kamineni Academy of Medical Sciences and Research Centre, Hyderabad, India
6Department of Neurology, Kamineni Academy of Medical Sciences and Research Centre, Hyderabad, India
7Department of Neurology, Prisma Health/University of South Carolina, Columbia, South America
- *Corresponding Author:
- Athira Nair Department of Surgery, Pontiac General Hospital, Michigan, United States E-mail: athira.r.nair13@gmail.com
Received: 20-Sep-2024, Manuscript No. AAAGIM-24-148415; Editor assigned: 23-Sep-2024, AAAGIM-24-148415 (PQ); Reviewed: 07-Oct-2024, QC No. AAAGIM-24-148415; Revised: 12-Aug-2025, Manuscript No. AAAGIM-24-148415 (R); Published: 19-Aug-2025, DOI: 10.35841/aaagim-9.5.200
Citation: Nair A, Pallapothu M, Nimmagadda R, et al. Comparative efficacy of duloxetine, gabapentin, and pregabalin in long-term diabetic neuropathy management. Arch Gen Intern Med. 2025;9(5):200.
Abstract
Numerous millions of individuals worldwide have excruciating diabetic peripheral neuropathy. The FDA has officially approved pregabalin and duloxetine hydrochloride as treatments for painful diabetic peripheral neuropathy. It is generally established that as individual therapeutic drugs, duloxetine, pregabalin, and gabapentin are safe for treating uncomfortable diabetic peripheral neuropathy. We evaluated the effectiveness, side effects, and tolerability of duloxetine to those of pregabalin and the often-recommended duloxetine-gabapentin combination treatment in this literature article. To compare these three presently used medications statistically, we examined several sources, including PubMed, Science Direct, AAFP Articles, Medscape, Cochrane Library, Cureus, Medline Plus, and a few other databases. Duloxetine or pregabalin are used as alternative treatments when a patient is not responding well to gabapentin. Which alternative would be best for the patient is still unclear. It is preferable to use duloxetine, which belongs to a distinct class of medications if a patient is on another anticonvulsant. Pregabalin is a preferable option if the patient is taking an antidepressant to avoid serotonin syndrome. This literature evaluation compares these three medications for the management of uncomfortable diabetic peripheral neuropathy.
Keywords
Diabetic peripheral neuropathy, Peripheral neuropathy, Duloxetine, Gabapentin, Pregabalin, Diabetic neuropathy management.
Introduction
Diabetic peripheral neuropathy is a painful medical ailment that affects 442 million people throughout the world [1]. About onefourth of patients suffering from type two diabetes mellitus are affected by painful peripheral neuropathy which has a major impact on their quality of life [2]. Peripheral neuropathy has serious consequences in terms of morbidity and death. The yearly incidence rate is about two percent and it affects 30 percent of hospitalized and 20 percent of diabetic patients living in the community [3]. It presents as a stocking glove of searing pain, paresthesia, and numbness that advances proximally from the feet and hands.
The therapeutic approach available for neuropathic pain includes a variety of pharmacological and non-pharmacological treatments, it is difficult to treat and usually necessitates a multidisciplinary treatment [4]. Maintaining hemoglobin A1c greater than or equal to seven, daily physical activity, and weight reduction are the cornerstones of diabetic distal symmetrical polyneuropathy therapy, and they should be undertaken early in the disease's progression [5]. In most patients, pharmacotherapy is the primary tool for successful pain control [4]. The US Food and Drug Administration has now authorized two drugs for the treatment of diabetic peripheral neuropathy: Duloxetine (duloxetine hydrochloride) and pregabalin [6].
The first-line therapy choice for neuropathic pain is pregabalin and as a substitute therapeutic choice is gabapentin [2,7]. Pregabalin binds to the two-delta subunit of calcium channels, similar to gabapentin, limiting neurotransmitter release [4]. It effectively relieves the symptoms of numerous forms of neuropathic pain and is a first-line treatment medication with exceptional safety and efficacy. Its efficacy in treating symptoms like allodynia and hyperalgesia has been demonstrated in preclinical investigations in numerous animal models of neuropathic pain [8].
Duloxetine inhibits serotonin (5-HT) and norepinephrine reuptake in the central nervous system, thereby elevating these neurotransmitters. Thus, duloxetine tends to reduce pain perception by lowering pain signals reaching the brain [6]. It is approved for the treatment of chronic depression, urinary stress incontinence, and diabetic peripheral neuropathic pain. Various studies have been carried out to see whether duloxetine can aid alleviate neuropathic and nociceptive pain [9].
When choosing therapy for painful diabetic peripheral neuropathy, clinicians should carefully assess the patient's objectives and functional state, as well as the medication's possible side effects. Furthermore, pharmaceutical doses and treatment durations should be controlled and titrated based on regular patient input on pain alleviation, increased function, and side effects. The need to consult a neurologist (ideally one who specializes in peripheral neuropathy) for treating diabetic neuropathy patients cannot be overstated. The focus of this research was to assess the efficacy of Duloxetine (DLX) to the anticonvulsants Pregabalin (PGB) and Gabapentin (GBP) in the management of Diabetic Peripheral Neuropathic Pain (DPNP).
Materials and Methods
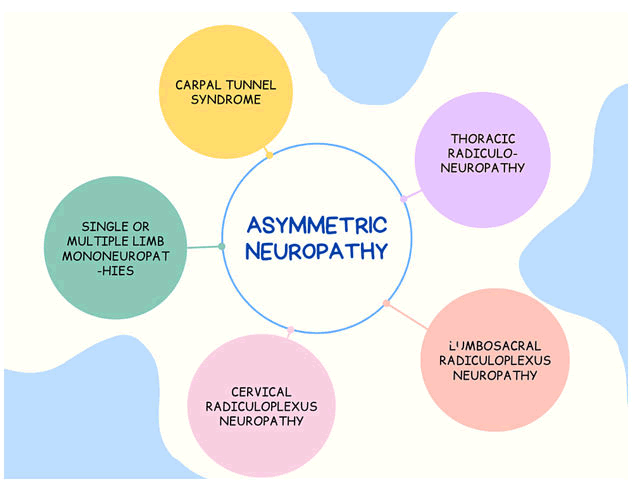
Neuropathic pain is attributed to the high utilization and expense of medical services. In this section, we will learn more about painful Diabetic Peripheral Neuropathy (DPN), duloxetine, pregabalin, gabapentin, and the comparison between these three therapeutic agents. More than 14 million Americans have diabetes mellitus. Painful diabetic neuropathy is thought to affect 20 to 24 percent of these people. There is a lack of information on the risk factors for Painful diabetic neuropathy, however, long-term diabetes and poor glycemic control are likely major contributors. The general action, temperament, versatility, business, social intelligence, rest, recreational interests, and joy of life may all be adversely affected by difficult diabetic neuropathy. They appear in at least 20% of persons with type one diabetes after 20 years of illness, according to several sizable observational cohorts and other research. When type two diabetes is first diagnosed, at least 10% to 15% of people may develop Distal Symmetrical Polyneuropathy (DSPN), with rates rising to 50% after 10 years of illness. Sensory symptoms include negative or positive, permeate or localized; frequently sneaky in the beginning, and exhibiting a stocking-and-glove allocation in the lower extremity. When sensory neuropathy coexists with motor weakness, it might be distal, proximal, or more localized (sensorimotor neuropathy). Autonomic neuropathy risks that could potentially affect the digestive, urogenital, and cardiac systems as well as the sweat glands. Asymmetrical neuropathies can present as carpal tunnel, mononeuropathies, thoracic, lumbosacral, or cervical radiculoneuropathies (Figure 1).
Figure 1. Asymmetric neuropathy pain
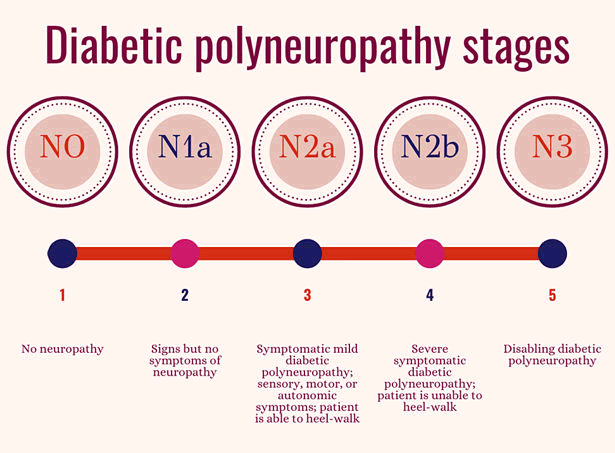
Diabetic Polyneuropathy (DPN) occurs in a few stages. About 26% of DPN patients experience distal symmetric polyneuropathy, characterized by stocking-glove-like searing pain, paresthesia, and numbness that advances proximally. Both the prevalence and severity of painful DPN are influenced by poorly managed blood glucose levels, particularly by increased variance in glucose levels. Improved glucose management in people with type one diabetes significantly decreased the chance of developing DPN, according to a 2012 Cochrane review of two randomized controlled trials (RCTs; n=1,228) (risk difference=1.84 percent; 95 percent Confidence Interval (CI), 1.11 to 2.56). However, a comparative study of four RCTs involving people with type two diabetes failed to demonstrate a statistically significant reduction in the rate of DPN with improved glucose management. Age, cigarette usage, hypertension, obesity, dyslipidemia, and peripheral artery disease are all cardiovascular risk factors that are linked to an increased incidence of painful DPN. It is crucial that we comprehend the most effective methods for diagnosing and treating this problem given the expanding prevalence of severe diabetic neuropathy. Diagnostic procedures in this area are developing quickly. Diabetic neuropathy diagnosis should focus on random/fasting glucose level, HbA1c, CBC-CMP, vitamin deficiency mainly B12 and folate, ESR, thyroid, etc. Checking for nerve conduction studies, skin/nerve biopsies, single-photon emission CT, MRI, and electrophysiologic studies may help to further evaluate the diagnosis. Stages of DPN is shown in Figure 2.
Figure 2. Stages of diabetic polyneuropathy.
Results and Discussion
The treatment of diabetic neuropathy should start as soon as diabetes is diagnosed. Because failing to detect diabetic polyneuropathy can have catastrophic repercussions, such as disability and amputation, the primary care physician has to be vigilant for the onset of neuropathy or even its existence at the time of initial diabetes diagnosis. The only drugs recognized by the FDA as effective in treating painful peripheral neuropathy are pregabalin and duloxetine. According to recommended clinical practices, these drugs should be taken into consideration as the first line of therapy together with gabapentin.
Duloxetine (brand name Cymbalta) comes under a group of drugs known as selective Serotonin and Norepinephrine Reuptake Inhibitors (SNRIs). Adults with depression and generalized anxiety disorder are treated with duloxetine which can also be helpful for some forms of pain and to treat enuresis. 60 mg doses of duloxetine are frequently prescribed. According to reasonably trustworthy statistics, duloxetine reduced pain in excruciating diabetic peripheral neuropathy. Duloxetine 60 mg/ day compared to placebo increased the likelihood of a 50% or greater improvement in diabetic peripheral neuropathic pain by slightly more than one point five times. It functions by raising levels of serotonin and norepinephrine, two naturally occurring chemicals in the brain that support mental equilibrium and slow the transmission of pain impulses.
Pregabalin (Lyrica) may be utilized as first-line therapy for painful peripheral neuropathy and has also been licensed to treat extensive diabetic peripheral neuropathic pain. Comparing the two medications, pregabalin, and gabapentin (Neurontin) against a placebo, both show a demonstrated but limited effectiveness. Pregabalin shares the same properties and mode of action as its predecessor gabapentin since it is chemically a Gamma-Aminobutyric Acid (GABA). The effectiveness of pregabalin for treating and controlling acute pain is comparable to that of gabapentin; however, pregabalin is superior to its predecessor because of its higher bioavailability, linear pharmacokinetics, lower dosage modulation, and quicker time to adequate pain-relieving impact. Additionally, it appears that pregabalin has a better chance of effectiveness in treating intense neuropathic pain. It has been believed that pregabalin and gabapentin appear to alleviate pain by altering the central nervous system's calcium channel α-2-δ subunit, this might explain their antinociceptive and antiepileptic properties. For pregabalin and gabapentin, respectively, the number needed to treat is seven point seven and six point three. Pregabalin and gabapentin both have comparable adverse effect profiles and are often adequately handled. The most prevalent symptoms, emerging in around 25% of patients, are dizziness and drowsiness.
In spite of the fact that the security and tolerability of duloxetine, pregabalin, and gabapentin as treatments for Diabetic Polyneuropathy (DPNP) have been set up, no distribution has however compared duloxetine to those of pregabalin and the regularly endorsed duloxetine furthermore gabapentin combination. A randomized study was done on 692 patients who were diagnosed with diabetic peripheral neuropathy and were at least over 18 years of age. During the initial screening, 285 patients were dropped out due to failing the screening. The 407 patients who were still in the study were randomly given Pregabalin 300 mg/day (for 134 patients), Duloxetine 60 mg/day (for 138 patients) and Duloxetine 60 Mg-Gabapentin >900 mg combined per day (for 135 patients). 38, 51, and 36 patients discontinued the study due to adverse effects, consent withdrawal, lack of efficacy, lack of follow-up, physician decision, protocol violation, and sponsor decision. The remaining 96 (71.6%), 87 (63%), and 99 (73.3%) patients from pregabalin, duloxetine, and pregabalingabapentin combined therapy respectively were able to complete the study. Although some people are little hesitant to undertake a treatment like this due to potential adverse effects, whether they have general neuropathic pain or diabetic neuropathic pain. However, this risk should be evaluated and addressed with a pain consultant so that it can be quantified. Unexpectedly, the majority (56%) reported a family member with Peripheral Diabetic Neuropathy (PDN), suggesting a possible hereditary propensity to the development of painful neuropathy. Patients who received duloxetine (30%) experienced withdrawals for any reason, compared to those who received placebo (28%); the NNTp for all causes of withdrawal with duloxetine as opposed to placebo was 26 (13 to 426). Fewer patients (four percent) who took duloxetine than those who took a placebo (nine percent) experienced withdrawals due to lack of effectiveness; the NNTp for such withdrawals with duloxetine, as opposed to a placebo, was 17 (12 to 35). When pregabalin was compared to a placebo, serious adverse effects were uncommon and did not differ between pregabalin users and those who took placebo pills. However, less severe adverse effects were frequent, especially fatigue and dizziness. Six to seven out of ten pregabalin users will have one or more side effects, but so will five to six out of ten placebo users.
Overall, the 150 mg dose did not result in an excess of adverse events, with the exception of drowsiness in patients with neuropathic pain following shingles, according to the data, which demonstrate a slight increase in the risk of adverse events at a daily dose of 600 mg compared with 300 mg. There were no variations in 24 h PS when duloxetine and pregabalin were indirectly compared. Pregabalin had a considerable advantage in PGI-I/C, but duloxetine had an advantage in dizziness. There were no statistically significant differences between gabapentin and duloxetine.
Various adverse effects were noticed in patients from each group. Nephrotoxicity, insomnia, hyperhidrosis, and decreased appetite were reported significantly more frequently in patients receiving duloxetine compared to those receiving pregabalin; insomnia was reported significantly more frequently in patients receiving duloxetine compared to those receiving duloxetine plus gabapentin; peripheral edema was reported significantly more frequently in patients receiving pregabalin compared to those receiving duloxetine and duloxetine plus gabapentin (p 0.001 for each); and at the end of the study, there was a significant difference in weight change between the treatment groups: Patients in the pregabalin group gained weight on average (1.0 0.04 kg), whereas those in the duloxetine and duloxetine plus gabapentin groups lost weight on average (2.39 0.04 and 1.06 0.04 kg, respectively).
In an additional 12-week, unrestricted research that included individuals experiencing painful diabetic peripheral neuropathy, who had already received gabapentin (900 mg/d) treatment but had an unsatisfactory result (characterized as an everyday torment value of equal to or more than 4 on a measure from the 0 to 10 focuses). On September 28, 2006, the first participant got enlisted, and on August 26, 2009, the final patient visit was placed. Duloxetine monotherapy (n=138), pregabalin monotherapy (n=134), or a combination of duloxetine and gabapentin (n=135) were given at random to the patients. The main goal was to compare the efficacy of duloxetine and pregabalin on each week's average of the checklist constant pain ratings (0-10 scale) at the ultimate end. For duloxetine and pregabalin, the mean change in the pain rating at the endpoint was -2.6 and -2.1, respectively. Noninferiority was established by the 97.5 percent lower confidence level, which was a -0.05 difference in means. In terms of side effects, pregabalin was more frequently associated with peripheral edema than duloxetine; nausea, hyperhidrosis, decreased appetite, and vomiting were more frequently associated with duloxetine plus gabapentin than pregabalin, and insomnia was more frequently associated with duloxetine than duloxetine plus gabapentin.
Patients in the duloxetine group dropped the greatest weight at the 12-week mark, with an average loss of 2.39 kg. Pregabalin patients gained on average 1 kg, while those on duloxetine with gabapentin decreased on average 1.06 kg. In the end, there were significant differences between the groups (p 0.01 for each comparison). For duloxetine and pregabalin, the mean change in the pain rating at the endpoint was -2.6 and -2.1, respectively. Low dosages of pregabalin and gabapentin utilized in these study trials may have contributed to both anticonvulsants' less effective performance in treating patients with painful diabetic peripheral neuropathy compared to duloxetine. Duloxetine's non-inferiority was established by the 0.05 difference in means that represented the 97.5 percent lower confidence level. Lethargy/somnolence, peripheral edema, and constipation were the most frequently mentioned side effects in the pregabalin group, whereas orthostatic hypotension and constipation were the most frequently mentioned side effects in the duloxetine group, as mentioned in Figure 3 below.
Figure 3. Comparing adverse effects of pregabalin and duloxetine.
Concerns about the possibility of misuse and dependence may act as a deterrent to taking painkillers as prescribed and obtaining therapeutic levels. Physicians must be able to identify possible signs of abuse in their patients as well as comprehend and address their concerns. Pregabalin and gabapentin recreational misuse is being reported more frequently and is a global problem in both the scientific literature and the general press. Current research reveals that the risk of establishing dependent on pregabalin is low in individuals without a history of substance addiction, even if doctors should be aware of the possibility for medication misuse or theft. As a twice-daily or three times-daily medications for the treatment of neuropathic pain brought on by Diabetic Peripheral Neuropathy (DPN), Post-Therapeutic Neuropathy (PHN), and spinal cord injury, Extended Release (ER) pregabalin has been given FDA approval. For the treatment of neuropathic pain connected to DPN and PHN, ER pregabalin is authorised as a once-daily dose. Although oncedaily dosage is convenient with the ER formulation in theory, pregabalin's variable absorption is a possible problem since its stomach retention duration is influenced by calorie consumption. The limited pregabalin absorption window is blamed for this heterogeneity. Duloxetine hydrochloride, the active component in Cymbalta, has also been approved for the treatment of moderate to severe stress urinary incontinence, another condition thought to respond to medication that alters serotonin and norepinephrine levels, in addition to depression and pain brought on by diabetic peripheral neuropathy. Duloxetine may be more effective in reducing pain in persons with DPNP who were not properly managed with gabapentin and did not also take antidepressants concurrently. Contrarily, switching to duloxetine or pregabalin in nonresponders to gabapentin who are already taking an antidepressant may result in comparable pain reductions.
The Researched Abuse, Diversion and Addiction-Related Surveillance (RADARS®) system gives a comprehensive picture of abuse patterns in various nations. It can also help to inform healthcare prescribers about the dangers of abuse and misuse in various patient populations and the necessity of keeping an eye out for abuse signs in patients. Pregabalin's lifetime misuse rates are modest compared to those of other medicines, such as opioids and benzodiazepines, according to recent real-world data from the RADARS® System. Additionally, pregabalin abuse or misuse was commonly reported by participants together with abuse of one or more additional medicines. When managing pharmacologic therapy, clinicians should take the patient's age, health-related qualityof- life objectives, physical function, and comorbidities into account, as well as any potential negative effects of pharmaceutical usage. Additionally, regular patient input about pain relief, increased function, and side effects should be used to adjust drug doses and the length of therapy. Pregabalin overdose should be noted that, unless combined with opioids or other sedatives, such as benzodiazepines, an overdose typically results in drowsiness and, rarely, seizures rather than serious toxicity. These statistics should be known to prescribers of healthcare in order to help them make treatment decisions, especially for vulnerable patients. Based on these findings, we advise that gabapentinoids be avoided in patients with present or prior drug use problems or, if necessary, taken cautiously under thorough therapeutic and prescription monitoring. When comparing the cost-effectiveness duloxetine started off starting pregabalin because it was more efficient and less expensive. A combination of desipramine and duloxetine first predominated gabapentin for a lengthy period of time. Costing $23,468 and $25,979, respectively, desipramine and duloxetine produce 3.05 and 3.16 QALYs. In comparison to desipramine, duloxetine had an increased cost-effectiveness ratio of $22,867/QALY gained. The model was most sensitive to the adherence threshold and utility for mild pain, according to a one-way sensitivity analysis. According to PSA, in 56.3 percent of the simulations, duloxetine was the most costeffective choice, followed by desipramine in 29.2 percent, gabapentin in 14.4 percent, and pregabalin in 0.1 percent, at a Willingness-to-Pay (WTP) of $50,000/QALY. When WTP is more than $22,867/QALY, duloxetine is the most economical alternative for PDN. For PDN patients, decision-makers could start with duloxetine.
Conclusion
Painful diabetic peripheral neuropathy has been successfully treated with pregabalin, duloxetine, and gabapentin. There were notable differences in wooziness, favouring duloxetine, but no quantitatively notable differences existed between gabapentin and duloxetine. So, the doctor may relax and give the patient duloxetine if they are not compatible with gabapentin. A research comparing duloxetine and pregabalin found that both had few side effects, indicating a modest advantage for duloxetine over pregabalin. Sexual dysfunction was reportedly recorded in the pregabalin group, which was a significant downside that caused individuals to stop using the medication. Pregabalin usage was frequently reported to be hampered by decreased appetite, peripheral edema, and weariness, whereas the duloxetine group experienced constipation and orthostatic hypotension as side effects. Duloxetine is therefore preferable to pregabalin in patients who are not responding to gabapentin and is in no way inferior to the other two therapy alternatives. Although all three of these drugs are effective in treating painful diabetic peripheral neuropathy, this literature evaluation may assist doctors and patients in deciding which drug is most effective for a given patient.
Limitations
We conducted an extensive bibliometric analysis in platform supported, but since we only incorporated studies from a few databases, we cannot completely deny the possibility that we missed relevant studies. Only a few authors really made the comparison between duloxetine and pregabalin/gabapentin. We discovered from earlier trials that all three of these therapeutic alternatives are excellent for treating neuropathic pain. To get a thorough comparison between these, we should try to undertake additional studies on this subject.
Ethics Approval and Consent to Participate
Not applicable to a review article.
Consent for Publication
All authors gave their consent for publication.
Availability of Data and Materials
Not applicable as no novel data were generated for this review article.
Competing Interests
No author has any conflict of interest or competing interests to declare.
Funding
No funding was received to perform this study.
Acknowledgment
None.
References
- Hagedorn JM, Engle AM, George TK, et al. An overview of painful diabetic peripheral neuropathy: Diagnosis and treatment advancements. Diabetes Res Clin. 2022;188:109928-0168.
[Crossref] [Google Scholar] [PubMed]
- Snyder MJ, Gibbs LM, Lindsay TJ. Treating Painful Diabetic Peripheral Neuropathy: An Update. Am Fam Physician. 2016;94(3):227-34.
[Google Scholar] [PubMed]
- Duby JJ, Campbell RK, Setter SM, et al. Diabetic Neuropathy: An intensive review. Am J Health Syst Pharm. 2004;61(2):160-73.
[Crossref] [Google Scholar] [PubMed]
- Macone A, Otis JAD. Neuropathic Pain. Medscape. 2018;38(6):644-53.
- Cernea S, Raz I. Management of diabetic neuropathy. Metabolism J. 2021;123:154867.
[Crossref] [Google Scholar] [PubMed]
- Irving G, Tanenberg RJ, Raskin J, et al. Comparative Safety and Tolerability of Duloxetine vs. Pregabalin vs. Duloxetine Plus Gabapentin in Patients with Diabetic Peripheral Neuropathic Pain. Int J Clin Pract. 2014;68(9):1130-40.
[Crossref] [Google Scholar] [PubMed]
- Yamada T, Mitsuboshi S, Makino J, et al. Risk of Pregabalin-Induced Hypoglycemia: Analysis of the Japanese Adverse Drug Event Report Database. J Clin Pharmacol. 2022;62(6):756-61.
[Crossref] [Google Scholar] [PubMed]
- Verma V, Singh N, Singh Jaggi A. Pregabalin in neuropathic pain: evidences and possible mechanisms. Curr Neuropharmacol. 2014;12(1):44-56.
[Crossref] [Google Scholar] [PubMed]
- Lunn MPT, Hughes RAC, Wiffen PJ. Duloxetine for treating painful neuropathy, chronic pain or fibromyalgia. Cochrane Database Syst Rev. 2014;1:7115.
[Crossref] [Google Scholar] [PubMed]