Research Article - Biomedical Research (2017) Volume 28, Issue 21
Clinical analysis of submucosal tunneling endoscopic resection in treating esophageal submucosal tumors
Fu Xinjuan1,2, Shi Xiuju1, Liu Hualin1, Liu Hui1, Li Bin1 and Xu Hongwei1*
1Department of Gastroenterology, Shandong Provincial Hospital of Shandong University, Jinan, Shandong Province, PR China
2Department of Gastroenterology, the Affiliated Qingdao Hiser Hospital of Qingdao University, Jinan, Shandong Province, PR China
- *Corresponding Author:
- Xu Hongwei
Department of Gastroenterology
Shandong Provincial Hospital of Shandong University, PR China
Accepted on September 18, 2017
Abstract
Background: Inspired by peroral Endoscopic Myotome (POEM), a new technique named Submucosal Tunneling Endoscopic Resection (STER) was developed for the treatment of submucosal gastrointestinal tumors.
Objective: To evaluate the clinical security and the feasibility of Submucosal Tunneling Endoscopic Resection (STER) for treating the submucosal tumors of esophageal.
Methods: Collecting cases during 2011 November to 2014 December in Shandong Provincial Hospital, all patients were diagnosed as esophageal submucosal tumors by Ultrasonography Endoscopic (EUS), and Computed Tomography (CT) scan. 58 patients were treated with STER, and 30 patients were treated with ESE, to observe two group’s indicators such as operation time, intraoperative and postoperative complications, tumor size, pathology and postoperative hospitalization days.
Results: All 58 patients’ tumors were resected en bloc successfully by STER at one time, the en bloc resection rate was 100%, the mean operation time was 63.24 ± 15.10 min, a bit shorter than the ESE group (71.97 ± 25.46 min), and there was significant difference between two groups (t=2.018, P=0.047). The mean size of the tumors was 2.07 ± 0.8 cm, larger than ESE group (1.28 ± 0.63 cm), and there was significant difference between two groups (t=4.659, P=0.013). No intraoperative perforation occurred in STER, and fevers 9 cases (15.8%), subcutaneous emphysema 3 cases (5.3%) and pneumothorax 2 cases (3.5%) were observed post operation, all patients recovered after conservative treatment. No delayed bleeding and secondary infection and esophageal fistula. The mean postoperative hospitalization days were 5.72 ± 2.24 d, no significant difference was observed compared with ESE group (6.27 ± 3.10 d). The incidence rate of fever, subcutaneous emphysema and pneumothorax of STER group were lower than the ESE group, but no statistical significance was observed. Three months later, gastroscope or Ultrasonography (EUS) review showed wound healing, and no residual tumor or relapse.
Conclusion: STER is a safe and feasible method for treating esophageal sumucosal tumors, and it worth of clinical promotion.
Keywords
Submucosal tunneling endoscopic resection, Endoscopic submucosal excavation, Esophageal tumor, Sumucosal tumor, Muscularis propria.
Introduction
With the increase of people’s health awareness and the development of Endoscopic diagnosis technology, upper gastrointestinal submucosal tumor draws more attention of the medical profession, the diagnostic rate of esophageal submucosal tumor increased significantly. Submucosal tumor such as leiomyoma, lipoma, stromal tumor, etc., most of which are benign tumors, a few have malignant potential or obstruction symptom and need active treatment. It’s difficult for the patients to accept surgical intervention because of more invasion and higher cost. Endoscopic treatment technologies such as Endoscopic Mucosal Resection (EMR), Endoscopic Submucosal Dissection (ESD) can be effectively treated tumors which derived from the mucous membrane layer and part of submucosa and Endoscopic Submucosal Excavation (ESE) is used for removal tumors derived from the muscularis propria which is just an extension of ESD technique. For lack of serosal layer, ESE risk high for larger esophagus submucosal tumors, especially for tumors originated from the muscularis propria, because it may lead to perforation, bleeding and other complications. And Submucosal Tunneling Endoscopic Resection (STER) technology [1] can establish tunnel effectively by using the space between submucosa and the muscularis propria, and then resect the tumor completely in the full space without destroying the integrity of the mucous membrane, and so decrease the risk of perforation and gets good operability [2]. STER is a new emerging technology in recent years, our study was to investigate the safety and efficacy of STER in treating esophageal submucosal tumor by retrospectively analyzing.
Materials and Methods
Materials
Patient information: 88 inpatients came from the Shandong Provincial Hospital during November 2011 to December 2014, all patients were diagnosed as esophageal submucosal tumors by ultrasonic gastroscopy and CT. 58 patients received STER (male 33 cases, female 25 cases, range 26-77 y old, mean 9 ± 10.50 y old), 30 patients received ESE (male 16 cases, female 14 cases, range 27-75 y old, mean 49.30 ± 11.22 y old). Routine blood, clotting mechanism, liver and kidney function were checked before operation, no taboo, all patients signed informed consent. This study was approved by the ethics committee of the Shandong provincial hospital.
Endoscopic device: Olympus 260 host, GIF-Q260J treatment gastroscope, transparent cap (D-201-11802, Olympus), endoscopic needle (25 g, Olympus NM-200L-0525), IT2 knife (KD-611L), 650 knife (dual knife, KD-650L), HOOK knife (KD-620-LR), thermal coagulation forceps (FD-410-LR), argon plasma coagulation (APC, ERBE), CO2 gas pump, etc.
Methods
Preoperative preparation: preoperative fasting 8 h, all patients were treated endoscopic operation with left lateral position under the propanol intravenous anesthesia and tracheal intubation, electrocardiogram monitoring and the CO2 air pump.
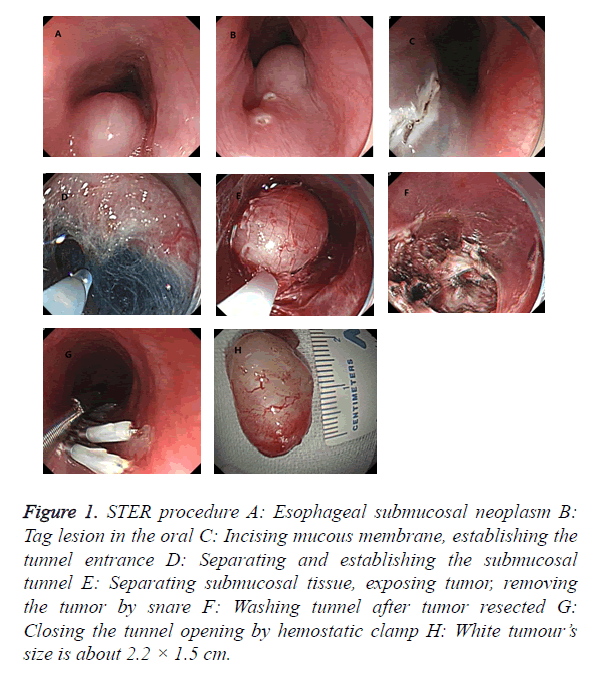
STER procedure as follows: (1) Transparent cap was added to gastroscopy front, injecting glycerol fructose indigo carmine into the submucosa about 4-6 cm proximal to the lesions at the oral side. (2) Incising mucous membrane by HOOK knife and establishing a submucosal tunnel entrance about 2 cm. (3) Establishing a submucosal tunnel, separating the tumor gradually and stripping the tumor completely by HOOK knife, then taking the mass out with a snare. (4) Endoscopic exit the tunnel, closing the tunnel incision site by hemostatic clamps (Figure 1).
Figure 1. STER procedure A: Esophageal submucosal neoplasm B: Tag lesion in the oral C: Incising mucous membrane, establishing the tunnel entrance D: Separating and establishing the submucosal tunnel E: Separating submucosal tissue, exposing tumor, removing the tumor by snare F: Washing tunnel after tumor resected G: Closing the tunnel opening by hemostatic clamp H: White tumour’s size is about 2.2 × 1.5 cm.
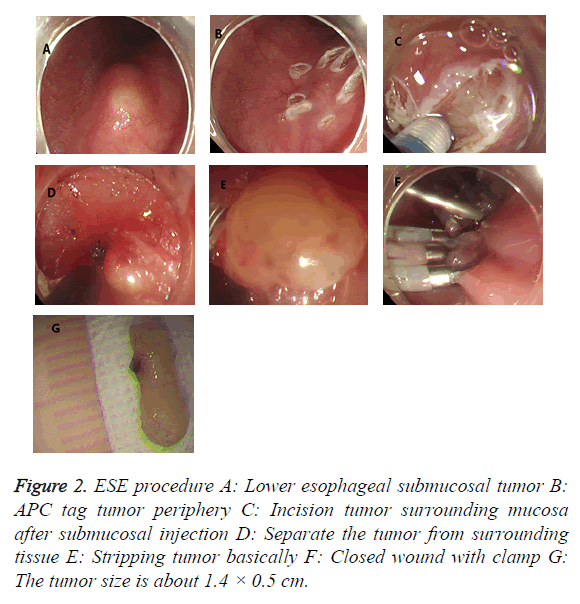
ESE procedure: (1) Installing the transparent cap to the upper front and marking the tumor edge with argon knife. (2) Injecting submucous multipoint along the side of the tag with a mixed solution of epinephrine, methylene blue and glycerol fructose. (3) Cutting the mucous membrane along the tumor edge with a HOOK knife, cutting submucosa and exposing tumors with IT knife, and removing tumors after it was completely exposed, then closed the incision with clips. The electrocautery was adopted if hemostasis during operation (Figure 2).
Postoperative management
All resection specimens were sent to pathological examination. It was necessary to retain the gastrointestinal decompression tube, fasting 1-3 d and monitor vital signs such as chest pains, breathing difficulty, fever, subcutaneous emphysema abdominal pain and dark bowel. Proton pump inhibitors and antibiotics were applied regularly, and proton pump inhibitors were applied 4 to 6 w after discharge. Gastroscope and ultrasonic gastroscopy were carried out 3 to 6 months after operation to observe wound healing, residual tumor and recurrence, and so on.
Statistical processing
With SPSS19.0 software, using independent sample t test to compare two groups’ operation time, tumor size, postoperative hospitalization days, etc. with the using chi-square test, Fisher exact probability method to compare two groups’ complication rates. P<0.05 was considered was statistically significant.
Results
Comparison of two groups operation situation
29 cases of 30 treated with esophageal ESE were resected the tumor completely, 1 case fail to be resected for larger lesions (3.5 × 1.5 cm) and adhesion, en bloc resection rate was 96.7%. Average operation time was 71.97 ± 25.46 min, range 45-160 min, average tumor size was 1.28 ± 0.63 cm, range 0.6 × 0.3 cm to 1.8 × 2.0 cm. One case appeared small perforation (3.3%) intraoperative and closed by clamp. 6 cases of postoperative fever (20%), 4 case of subcutaneous emphysema (13.3%), 2 cases of pneumothorax (6.7%), all patients recovered by conservative treatment, no delayed hemorrhage and secondary infection cases and no esophageal fistula was found. Average postoperative hospitalization days were 6.27 ± 3.10 d, range 3 to 15 d.
All 58 cases were successfully implemented STER and the en bloc resection rate was 100%. Operation time was from 40 to 120 min, average 63.24 ± 15.10 min, a bit shorter than the ESE group (t=2.018, P=0.047). Tumor size was about 0.8 × 1.3 cm to 5.5 × 3.5 cm, average 2.07 ± 0.81 cm, larger than ESE group, and there was significant difference between two groups (t=4.659, P=0.000). No perforation occurred intraoperative. After operation, fever in 9 cases (15.8%), subcutaneous emphysema in 3 patients (5.3%), pneumothorax in 2 cases (3.4%) appeared, the incidence of fever, subcutaneous emphysema and pneumothorax were slightly lower than ESE group and no significant difference, all patients recovered after conservative treatment. No delayed bleeding and secondary infection and esophageal fistula. Postoperative hospitalization days were from 3 to 11 d, average 5.72 ± 2.24 d (Tables 1 and 2).
| Groups | ESE | STER | t | P |
|---|---|---|---|---|
| Operation time (min) | 71.97 ± 25.46 | 63.24 ± 15.10 | 2.018 | 0.047 |
| Tumor size (cm) | 1.28 ± 0.63 | 2.07 ± 0.81 | 4.659 | 0.000 |
| Postoperative hospitalization days | 6.27 ± 3.10 | 5.74 ± 2.24 | 0.913 | 0.364 |
Table 1. Comparison of two groups in operation time, tumor size and postoperative hospitalization days.
| Groups | ESE | STER | χ2 | P |
|---|---|---|---|---|
| Fever | 6 (20.0%) | 9 (15.8%) | 0.281 | 0.596 |
| Subcutaneous emphysema | 4 (13.3%) | 3 (5.3%) | 0.857 | 0.355 |
| Pneumothorax | 2 (6.7%) | 2 (3.4%) | 0.022 | 0.883 |
Table 2. Comparison of two groups in incidence of complications.
The postoperative pathology and follow-up
ESE groups: leiomyoma 28 cases (93.3%), stromal tumor 1 case (3.3%) and lipoma in 1 case (3.3%).
STER group: leiomyoma 53 cases (91.4%), stromal tumor 3 case (5.2%), and lipoma in 2 case (3.4%).
Three month later, 2 groups of patients were reviewed with gastroscopy or Ultrasonography (EUS) and no residual tumor or relapse or narrow were found.
Discussion
Gastrointestinal submucosal tumor mainly refers to the following tumor which originated from mucous membrane layer, the submucosa and muscularis propria. Esophageal submucosal tumors are usually benign lesions. Leiomyoma is the most common. Generally, it hasn’t obvious clinical symptoms and happen been found by endoscopic examination. Some bigger tumors may cause obstruction symptoms, and those tumors derived from gastrointestinal mesenchymal tissue have malignant potential, follow-up or actively processing were recommended, if the tumor is greater than 2 cm, it is recommended removal [3]. Usually, surgical resection could made definite effect, but it maybe has large trauma and high risk, and subsequent high incidence of complications frustrated us. With the development of endoscopic techniques, endoscopic treatment field gradually widened, ESE and STER were all used for submucosal tumors recent years.
In 1996, Gotoda first removed mucosal lesion completely through IT knife and named Endoscopic Submucosal Dissection (ESD) [4], initially it is used for removal of mucosa lesion larger than 2 cm in diameter. Later, Pinghong [5] put forward resecting submucosal tumors with ESD technology and called it Endoscopic Submucosal Excavation (ESE), which was applied for submucosal tumors derived from muscularis propria [6]. Multicenter study abroad [7] showed that the en bloc resection rate of ESD achieve 96.7% and the incidence rate of perforation 5.25%. While [8] reported that intraoperative hemorrhage rate of stomach ESD was 12% (20/167), delayed hemorrhage rate was 1.8% (3/167). For larger tumors (>2 cm) from esophageal muscular propria, ESD wound closed more difficult, also the risk of perforation and bleeding increased, especially pneumothorax. Submucosal Tunneling Endoscopic Resection (STER) technique developed on the basis of the Peroral Endoscopic Myotomy (POEM) and was applied to submucosal tumors resection, which was first reported by Xu [1] in 2011. The technique establishes an artificial tubular tunnel in the digestive tract between mucosa layer and muscular propria by endoscopy, then diagnosis and treatment in the submucosa, muscular propria serous layer was implanted in the tunnel. This method makes the esophagus mucous membrane incision and the surgical wound is not in the same level, which can not only resect of the tumor completely, but also maintain the integrity of the mucous membrane, then avoid damage to nearby tissue and organs and reduce the possibility of delayed perforation. Due to the tunnel space can up and down extension, operation can be done smoothly and fast and perforation risk reduced significantly compared with ESE.
Our study stated that tumor size will affect the operation choice, STER group’s tumor is greater than the ESE group, also illustrated that the ESE is unfavorable for larger tumors. Restricted by esophageal tunnel space, it is difficult to operate if tumor diameter is more than 3 cm, but in STER group the biggest tumor resected is about 5.5 × 3.5 cm, which broke the general scholar’s view that STER is mainly suitable for the tumors whose diameter is about 3.5 cm or less [9]. Second, the middle and lower esophagus have high tumor incidence, tumors location affect operation choice. If tumors located upper esophagus especially less than 20cm from incisors, submucosal tunnel is difficult to build and ESE is preferable. If tumors position is low and lies in the border between cardiac stomach bottom, ESE often have residual tumors and operation difficulty is bigger because tumors at this position often extends to the bottom of the stomach which lobulated or have pseudopodia, and STER is preferable. Our research showed that STER patients’ incidence of fever, subcutaneous emphysema and pneumothorax were slightly lower compared with ESE group, the incidence of postoperative fever and subcutaneous emphysema of the upper esophageal submucosal tumors is higher than the middle and lower esophagus, whose cases were of small size.
As other microscopic minimally invasive treatment, STER also has some common complications such as mediastinum and subcutaneous emphysema, pneumothorax, pneumoperitoneum, bleeding, perforation, secondary infection, and so on. Esophageal lacks of serous layer, outer longitudinal muscle and outer membrane were liable to be damaged when separating the basal part of the tumors derived from the muscularis propria, and may result in such complications as subcutaneous emphysema, pneumomediastinum or pneumothorax, etc. Also, some tumors located in the deep muscularis need full thickness resection and hard to avoid perforation. Due to the tunnel entrance and surgical wound is not in the same level, we clip the entrance of the tunnel when perforation happened and full thickness perforation wouldn’t generally appear. Our study showed 9 cases of postoperative fever, some cases of pneumothorax, subcutaneous emphysema and free gas under diaphragm, who recovered by conservative treatment, no severe perforation and other complications happened. Using CO2 gas pump infusion, for CO2 is normal metabolism product and can quickly be absorbed in the organization whose absorption rate is about 150 times that of air, so the clinical symptoms of subcutaneous emphysema, pneumothorax and pneumoperitoneum can be recovered quickly after surgery [10,11].
STER, ESE are all the effective and safe minimally invasive treatments for resecting upper gastrointestinal submucosal tumors, and STER has an advantage in large submucosal tumor resection because it can maintain the integrity of the digestive tract mucosa and reduce complications. But STER is a new technology appearing in recent years and there may be deviation in the data because of the limited sample size. We look forward to multicenter, large sample research which can provide more reliable data for clinical work.
STER can preserve the mucosa integrity technically, but in our study, perforation rate of two means hasn’t large difference, maybe we need more patients.
References
- Xu MD, Cai MY, Zhou PH. Submucosal tunneling endoscopic resection: a new technique for treating upper GI submucosal tumors originating from the muscularis propria layer (with videos). Gastrointest Endosc 2012; 75: 195-199.
- Nishida T, Kawai N, Yamaguchi S, Nishida Y. Submucosal tumors: comprehensive guide for the diagnosis and therapy of gastrointestinal submucosal tumors. Dig Endosc 2013; 25: 479-489.
- Demetri GD, von Mehren M, Antonescu CR, DeMatteo RP, Ganjoo KN, Maki RG. NCCN Task Force report: Update on the management of patients with gastrointestinal stromal tumors. J Natl Compr Canc Netw 2010; 8: 1-41.
- Gotoda T, Kondo H, Ono H. A new Endoscopic Mucosal Resection (EMR) procedure using a insulation-tipped diathermic (IT) knife for rectal flat lesions report of two cases. Gastrointest Endosc 1999; 50: 560-563.
- Ping-Hong Z, Qiang S, Yun-Shi Z. New progress in endoscopic treatment of esophageal diseases. World J Gastroenterol 2013; 19: 6962-6968.
- Huang LY, Cui J, Liu YX, Wu CR, Yi DL. Endoscopic therapy for gastric stromal tumors originating from the muscularis propria. World J Gastroenterol 2012; 18: 3465-3471.
- Tsujii Y, Nishida T, Nishlyama O. Clinical outcomes of endoscopic submucosal dissection for superficial esophageal neoplasms: a multicenter retrospective cohort study. Endoscopy 2015.
- Jeon SW, Jung MK, Cho CM. Predictors of immediate bleeding during endoscopic submucosal dissection in gastric lesions. Surg Endosc 2009; 23: 1974-1979.
- Xu MD, Yao LQ, Zhou PH. The submucosal tunnel endoscopic resection of the tumor treatment is the result of upper gastrointestinal submucosal tumor of a preliminary study to the muscularis propria. Dig Endosc 2011; 28: 606-610.
- Bassan MS, Holt B, Moss A. Carbon dioxide insufflation reduces number of post-procedure admissions after endoscopic resection of large colonic lesions: a prospective cohort study. Gastrointest Endosc 2013; 77: 90-95.
- Maeda Y, Hirasawa D, Fujita N, Obana T, Sugawara T, Ohira T. A pilot study to assess mediastinal emphysema after esophageal endoscopic submucosal dissection with carbon dioxide insufflation. Endoscopy 2012; 44: 565-571.