Research Article - Biomedical Research (2019) Volume 30, Issue 1
An epidemiologic investigation of the prevalence of latent tuberculosis amongst students at Umm Al-Qura University: a pilot study.
Abdullah Aburiziza1*, Mahomed Farouk Mahomed2, Ayman Khalid Johargy2, Abdulaziz El-Zouki2 and Hassan S. Shaibah3,4
1Department of Pediatrics, Faculty of Medicine, Umm Al-Qura University, Makkah, Kingdom of Saudi Arabia
2Department of Medical Microbiology, Faculty of Medicine, Umm Al-Qura University, Makkah, Kingdom of Saudi Arabia
3Department of Anatomy, College of Medicine, Alfaisal University, Riyadh, Kingdom of Saudi Arabia
4Department of Anatomy, Faculty of Medicine, Umm Al-Qura University, Makkah, Kingdom of Saudi Arabia
- *Corresponding Author:
- Dr. Abdullah Jameel Aburiziza, MBChB, ABP, FAAP,
FAAAAI
Department of Pediatrics, Faculty of Medicine
Umm Al-Qura University, Makkah,
Kingdom of Saudi Arabia
E-mail: aburiziza@gmail.com
Accepted date: January 12, 2019
DOI: 10.35841/biomedicalresearch.30-18-1205
Visit for more related articles at Biomedical ResearchAbstract
Introduction: Mycobacterium tuberculosis, the prime etiological factor of Tuberculosis (TB), latently infects almost 33% of the global population. It is further buoyed by the emerging multidrug resistance and human immunodeficiency virus type I (HIV-1) pandemic, necessitating the development of novel control measures and stratagems for its specific diagnosis and prognosis. Although tuberculin skin test (TST) is the first choice for detecting latent Mycobacterium tuberculosis infection (LTBI), it cannot be regarded as the gold standard as it randomly gives false implication of the test and there is eccentric variation in its interpretation.
Objective: The current study was designed to assess the degree of sensitivity and specificity of IFN-c as QuantiFERON-TB Gold In-Tube (QFT-Gold IT) assay in the diagnosis of TB as compared to the TST.
Subjects and methods: The students from medical and paramedical students at Umm Al-Qura University were selected in the study and all selected students entered into the study before the commencement of their internship program. Pre-internship: A total of 47 blood samples were collected from medical and paramedical students at Umm Al-Qura University. Post-internship: On follow up after the internship program we managed to get back only 36 subjects of the 47, due to the unavailability of the subjects due to unanticipated reasons.
Results: Out of the 47 samples tested by ELISA test using QFT-Gold IT and TST, none of pre-internship subjects were positive in either of the test, however, of the 36 post-internship students only 1 (2.8%) tested positive in both tests.
Conclusion: In this study, the two tests, QFT-Gold IT and TST were found to be 100% correlated and it was found that QFT-Gold IT with remarkable sensitivity and specificity which is equivalent to that of the TST. Moreover, QFT-Gold IT is impervious to BCG vaccination and other similar variables but not TST. Since, the current study was only a pilot study and the sample size were very small, hence, further studies need to be done to determine the true sensitivity and specificity of the QFT-Gold IT.
Keywords
Mycobacterium tuberculosis, the prime etiological factor of TB, latently infects almost 33% of the global population. It is further buoyed by the emerging multidrug resistance and human immunodeficiency virus type I (HIV-1) pandemic, necessitating the development of novel control measures and stratagems for its specific diagnosis and prognosis [1]. Although tuberculin skin test (TST) is the first choice for detecting Mycobacterium tuberculosis infection (LTBI), it cannot be regarded as the gold standard as it randomly gives false implication of the test and there is eccentric variation in its interpretation. [2]. In spite of advances in the diagnosis, prognosis, treatment, control and prevention of Tuberculosis (TB), it is a re-emerging problem with rapid and complex evolution posing a threat to global public health. According to Global Tuberculosis Report of World Health Organization (2018), The TB is one of the top 10 rank causes of global deaths due to a single mycobacterial infection. There are millions of people continuously infected with Mycobacterium tuberculosis and get suffered with a fatal disease of TB every year which is more than HIV/AIDS. Surprisingly, it is estimated that approximately 23% of the world’s population (1.7 billion people) are reported to have a latent TB infection, and hence, these peoples are at high risk of developing active TB disease anytime during their lifetime [3]. Saudi Arabia, third-largest Arab country with a total estimated population of 29,195,895. Epidemiological Analysis of pulmonary TB in Saudi Arabia in 2017 reported that the total number of new cases of pulmonary tuberculosis in 2017G (1438H) was found to be 2095 with an overall incidence rate of 6.44 cases/100,000 population. These figures imply that it’s a country with moderate TB burden [4,5]. Mycobacterium tuberculosis infection, mainly airborne [3,4], is a leading root cause of mortality due single species infection and hence an ever-expanding world health emergency [5]. A positive “sputum smear” indicates higher risk of TB transmission and the rate of transmission is directly dependent on the bacillary density during respiratory secretion [5] and consequently those in close and persistent vicinity of the victims are at higher risk of getting infected [3-5]. Similarly, immunocompromised patients (absolute or relative) and the healthcare workers are more prone to Mycobacterium tuberculosis infection from the index case [3-5]. Once an individual is infected, the risk of progression from TB infection to TB disease depends on various factors such as age and immune system being the prime aspects [2]. In every immunodeficient older child, adult and untreated HIV-infected patients this risk is 5-10% whereas in infants (<2 y), its alarmingly between 40-50% [2,4]. The Mycobacterium tuberculosis infection spreads aggressively covering up to 50% of its growth in the first two years [4]. The individuals in whom the TB infection did not progress to TB disease enter a state of latency, latent tuberculosis infection “LTBI” [3-10] which can reactivate at later stage particularly if the individuals become immunocompromised, LTBI individuals have no clinical or radiological findings consistent with TB.
Health care workers especially medical and paramedical students are at higher risk of developing LTBI as they meet TB patients more frequently and therefore a contact screening must be carried out for early diagnosis of TB transmission [3-6,8]. Contact screening has distinguished significance as it helps in early detection of infected individuals in the community and characterizes the recent ongoing trends of transmission of infection within a set of population. Such transmissions can be controlled remarkably by treating the healthcare worker, medical and paramedical students infected with LTBI. Isoniazid (INH) therapy diminishes the infection of TB by 94% and should be administered daily for 9 months to all patients with LTBI [4].
National and international tourism, immigration and relocation of human population expedite the transmission of TB [11-13]. As per the data on global burden of TB, it is estimated that more than 1.7 billion people (approximately 25% of the global population) are estimated to be infected with Mycobacterium tuberculosis. The global incidence of TB was reported to be highest in 2003 and now gradually decreasing. This is reported that around 10 million individuals became ill with TB and 1.6 million died, according to the WHO report in 2017 [14]. Annually 3 million people visit Makkah to perform Hajj [15], most pilgrims are adults, often elderly and often from countries in south East Asia and Africa with high prevalence of Tuberculosis infection which reach to 40% of population [3-7,16]. This 6 weeks long pilgrimage has a lot of overcrowding which may enhance the spread of airborne infections [17]. More than half of the pilgrims have been reported to be suffering from cough [18-20] and it has been observed that the pulmonary TB is solely responsible for pneumonia in hospitalized pilgrims during the Hajj [21]. There is high risk of transmission of Mycobacterium tuberculosis infection during this pilgrimage, as reported in one of the study, the immune response to TB antigen was measured in pilgrims using Quanti-Feron TB assay prior to departure for Hajj and after 3 months of their return, 149 pilgrims were negative prior to the Hajj and 15 (10%) of these had a severe immune response to the TB antigen [22]. Additionally, Saudi Arabia has several development projects which attract multitude of international employees specifically from the underdeveloped or developing nations including Asian and African countries which have high prevalence of Tuberculosis. Envisaging the environmental and social circumstances in Makkah the probability of acquiring TB for the medical and paramedical students in Umm Al-Qura University is quite high. Therefore, preventive measures need to be taken including screening, early detection and preventive chemotherapy. Systemic review of rapid diagnostic tests for latent tuberculosis recommend studies to evaluate interferon assay and its comparison with tuberculin skin test in diagnosing such cases of latent tuberculosis.
Detection of LTBI by QuantiFERON-TB Gold Test
TST was the only practical and commercially accessible immunologic test for Mycobacterium tuberculosis infection available in the US by the end of 20th century [23]. Later, it was revealed that interferon gamma (IFN-γ) has a significant regulatory role in cell-mediated immune response against Mycobacterium tuberculosis infection and this led the foundation for the development of interferon gamma release assays (IGRAs) for the early detection of this infection [24].
For the diagnosis of Mycobacterium tuberculosis infection, Food and Drug Administration (FDA) approved QFT-TB test as the first IGRA to ease the detection in 2001. QFT-Gold IT was based on the enzyme-linked immunosorbent assay (ELISA) to compute the quantity of IFN-γ released in against to a purified protein derivative (PPD) [25]. Again in 2005, FDA approved QFT-TB Gold test as the second IGRA to estimate the immunological responsiveness of patients tested to Mycobacterium tuberculosis-specific antigens (ESAT-6 and CFP-10). The guidelines for QFT-TB Gold test also implied that it can be employed in all the conditions in which a TST has been recommended, including but not limited to the assessment of recent immigrants, contact investigations, health care workers and serial-testing surveillance programs organized for controlling such infections [26]. Nevertheless, IGRAs has limitation of accuracy in the measurement of IFN-γ viz. only a fresh sample of blood containing viable white blood cells can give accurate measurements. QFT-Gold IT, a novel blood test was designed to meet this limitation and was approved by FDA as the third IGRA in 2007. QFT-Gold IT employs blood collecting tubes of 1 ml volume which is specifically layered with Mycobacterium tuberculosis responsive antigens (ESAT-6, CFP-10 and TB 7.7 (p4), a negative and a positive control tube. All these antigens are encoded within two regions of the genome of Mycobacterium tuberculosis and are absent in all Bacillus Calmette-Guerin (BCG) strains apart from most of the non-tuberculous mycobacterial (NTM) species but M. kansasii, M. marinum and M. szulgai. This is what renders the assay to be highly accurate in identifying the infection of TB. Later in July 2008, FDA approved T-spot as the fourth IGRA. T-spot is based on an enzyme-linked immunospot assay (ELISpot) for calculating the increase in the population of cells secreting IFN-γ post-stimulation by antigen [27]. Both QFT-Gold IT and T-spot assays were approved by FDA as indirect tests for TB infection in conjunction with the evaluation of risk, radiography, and other medical and diagnostic assessments [28]. QFT-Gold IT and T-spot have several advantages over TST such as collection of blood sample in a single visit, quick retrieval of test results (<24 h) and they are unaffected by preceding BCG vaccination [29]. Nonetheless, QFT is unable to distinguish between freshly attained or old infection, or between LTBI and acute disease [30]. Although, the sensitivity of IGRA in pediatric victims is supposed to be equivalent to TST but there are reports claiming IGRAs to have lower sensitivity than TSTs in pediatric patients [25]. In a study involving 28 pediatric patients with culture-confirmed active TB (aged 4 months-7 years), assessment of sensitivity for TST, QFT-Gold IT, and T-spot were 100%, 93%, and 93% respectively [31]. Similarly, in other study comprising 25 child patients with culture-confirmed active TB, percentage of sensitivity were 83% for TST, 80% for QFT-Gold IT, and 58% for T-spot [32]. More intensive studies are needed to assess the efficacy of IGRAs in pediatric patients [33]. Our study was designed to investigate and evaluate the use of QFT in determining the prevalence of LTBI amongst medical and paramedical students at Umm Al-Qura University in (Makkah City) Saudi Arabia.
Methods
Sample collection
Written informed consent of participants was obtained prior to enrolment. In addition, information regarding gender, age, faculty program and year of study was recorded on a data form.
Pre-internship
“A total of 47 blood samples were collected from medical and paramedical students at Umm Al-Qura University. Students were selected and entered into the study before the commencement of their internship program”.
Post-internship
On follow up after the internship program we managed to get back only 36 subjects of the 47, due to the unavailability of the subjects due to unanticipated reasons.
Subjects of the study
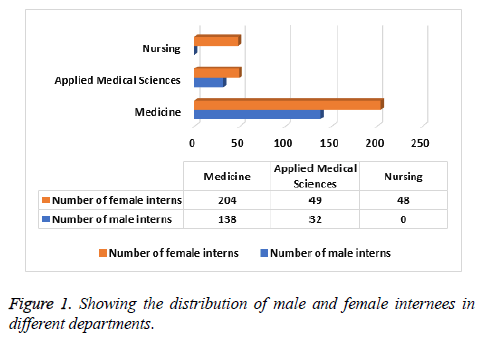
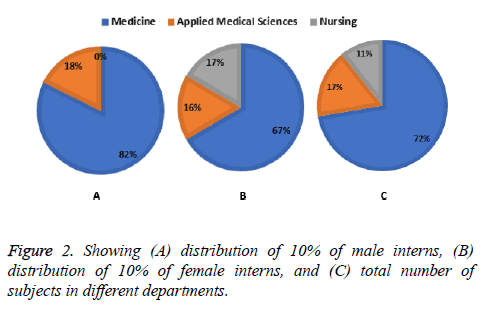
The registered number of students in internship year was obtained from the academic offices of three different department’s viz. Medicine, Applied Medical Sciences (Laboratory Medicine program) and Nursing, and then a 10% of total number of male students and 10% of female students were calculated from each to select a statistically representative number of students. Selection and calculation of number of subjects from the department of Medicine, Applied Medical Sciences and Nursing for this study is illustrated in Figures 1 and 2. Thus, total number of students selected was 47 out of which 34 were from Department of Medicine, 8 were from Department of Applied Medical Sciences and 5 were from Department of Nursing.
Procedure for QFT-Gold IT test
From each of the subject selected, 1 ml of blood was collected directly into each of 3 colored tubes i.e. negative control (gray), test tube (red) and positive control (purple) tubes. Succeeding the incubation of these tubes at 37°C for 16-24 h incubation period, centrifugation was carried out, the plasma is taken off and the quantitative estimation of IFN-c is measured by ELISA in terms of IU/ml. The non-visible reaction between the plasma samples and the conjugate shows a colored visible reaction after adding the enzyme substrate that indicates IFN-c existence in the patient’s plasma sample (Figure 3). The software implements a quality control assessment of the obtained results, generates a standard curve and provides the final test result for each subject as negative or positive.
Results
A total of 47 samples were tested by ELISA test using QFT-Gold IT, as well as the TST. None of the pre-internship subjects tested positive in either test. Of the 36 post-internship students only 1 tested positive in both tests.
Samples tested from pre-internship students
Total number of samples tested was 47, out which none were reported to be positive in either of the tests viz. TST or QFT-Gold IT test, thus the percent positive was 0/47=0% (Table 1) for each of them.
| Pre-internship | Post- internship | |
|---|---|---|
| Total samples | 47 | 36 |
| TST positive | 0 (0%) | 1 (2.8%) |
| QFT-Gold IT test positive | 0 (0%) | 1 (2.8%) |
Table 1: Results of ELISA QuantiFERON-TB Gold In-Tube (QFT-Gold IT) and tuberculin skin test (TST).
Samples tested from student’s post-internship
Out of a total number of samples tested (n=36), only one positive result was obtained for TST as well as the QFT-Gold IT test, and hence the percentage of positive sample was 1/36=2.8% for both (Table 1).
Discussion
The QFT-Gold IT assay is an ELISA that measures the production of IFN-c by T-cells after sensitization with Mycobacterium tuberculosis antigens using whole blood [34,35]. The aim of this study was to compare the sensitivity and specificity of QFT-Gold IT assay (Cellestis Limited, Carnegie, Victoria, Australia) with that of the TST. It also aimed to establish QFT-Gold IT assay as one of the novel diagnostic methods so that new control measures and strategies for specific diagnosis and prevention of TB transmission can be undertaken. This was a pilot study carried out on pre-intern and intern students from three different departments, namely, Nursing, Medicine and applied medical sciences at Umm Al-Qura University. The subjects were selected after seeking their consent and sitting together with the statistician to work out the sample size.
As evident from our results a cent percent correlation between the QFT-Gold IT assay and TST was observed. Although, it is only a pilot study with a very small sample size, but our results are in keeping with other study carried out in India on 726 healthcare workers; QFT-Gold IT positive in 40% and TST was positive in 41%. QFT-Gold IT assay result showed high congruence with that of TST and there was no effect of BCG on either of the tests [36]. In contrast, a study of 309 German contacts; 51% of whom were BCG vaccinated and 27% were foreign born. The 2/3rd of BCG vaccinated population and 18% of non-vaccinated were found to be TST positive, however only 9% and 11% were QFT-Gold IT test positive respectively. As observed QFT-Gold IT was linked with risk of TB whereas TST was only linked to BCG vaccination [37].
Similarly, in our results QFT-Gold IT revelations are less prone to reader bias and error as compared to TST. QFT-Gold IT test and TST interpretations were reasonably parallel, but their level of resemblance was negatively affected by preceding BCG vaccination, a prior positive TST and previous immune reactivity to non-tuberculous mycobacteria [38]. However, one out of the five sites included in this study reported poor concord [39].
A recent review article incorporated freshly reported data from 20 different studies into an updated meta-analysis on the IGRAs and its sensitivity and specificity, covering studies that assessed TSPOT, QuantiFERON-TB Gold and QuantiFERON-TB Gold In-Tube [40]. The pooled sensitivity was lowest in the QuantiFERON-TB Gold In-Tube (70% (CI, 63-78%) and highest in for T-SPOT (90% (CI, 86-93%)) while it was 78% (95% CI, 73-82%) for QuantiFERON-TB Gold [41]. Both QuantiFERON tests exhibited a pooled specificity of 99% among non-BCG-vaccinated subjects and 96% among BCG-vaccinated subjects however, the combined specificity of TSPOT was 93%. TST results were dissimilar, but specificity was constantly high (97%) in non-BCG-vaccinated subjects. Conclusively, this study revealed that the IGRAs, especially QFT-Gold and QFT-Gold In-Tube, possess exceptional specificity that is impassive to BCG vaccination [41,42].
ELISA-IGRA is supposed to be highly explicit for active TB, as demarcated by the number of patients without TB having negative reports of ELISA-IGRA. In a couple of related studies, the specificity of ELISA-IGRA, particularly of QFT ranged between 97-100% [43-47]. Our study showed strikingly similar results and cent percent correlation between QFT-Gold IT test and TST.
Conclusion
In this study, the both tests, QFT-Gold IT and TST were cent percent correlated and it was found that QFT-Gold IT with remarkable sensitivity and specificity which is equivalent to that of the TST. Moreover, QFT-Gold IT is impervious to BCG vaccination and other similar variables but not TST. However, this was only a pilot study and the sample size was very small, hence, further studies need to be done to determine the true sensitivity and specificity of the QFT-Gold IT.
References
- Raviglione M, Sulis G. Tuberculosis 2015: burden, challenges and strategy for control and elimination. Infect Dis Rep 2016; 8: 6570.
- Al-Orainey IO. Diagnosis of latent tuberculosis: can we do better? Ann Thorac Med 2009; 4: 5-9.
- World Health Organization. Global tuberculosis report 2018. World Health Organization.
- Pulmonary Tuberculosis by Region, Age Group, Sex and Nationality, 2017 G (1438 H), Annual Statistical Book, ISSN: 319-3228, Ministry of Health, Riyadh, Saudi Arabia.
- Alzohairy MA. Epidemiology of tuberculosis among migrant workers in Qassim area, Saudi Arabia. Res J Med Sci 2011; 5: 223-236.
- Nienhaus A, Schablon A, Preisser AM, Ringshausen FC, Diel R. Tuberculosis in healthcare workers-a narrative review from a German perspective. J Occup Med Toxicol 2014; 9: 9.
- Dutta NK, Karakousis PC. Latent tuberculosis infection: myths, models, and molecular mechanisms. Microbiol Mol Biol Rev 2014; 78: 343-371.
- Guirado E, Mbawuike U, Keiser TL, Arcos J, Azad AK, Wang SH. Characterization of host and microbial determinants in individuals with latent tuberculosis infection using a human granuloma model. MBio 2015; 6: 02537-14.
- Hunter RL. Tuberculosis as a three-act play: A new paradigm for the pathogenesis of pulmonary tuberculosis. Tuberculosis (Edinb) 2016; 97: 8-17.
- Committee on Infectious Diseases, American Academy of Pediatrics. Update on Tuberculosis Skin Testing of Children. Pediatrics 1996; 97: 282-284.
- Committee on Infectious Diseases, American Academy of Pediatrics. Screening for Tuberculosis in Infants and Children. Pediatrics 1994: 93; 131-134.
- Chang S, Wheeler LSM, Farrell KP. Public health impact of targeted tuberculosis screening in public schools. Am J Public Health 2002; 92: 1942-1945.
- World Health Organization. Guidance for national tuberculosis programs on the management of tuberculosis in Children, Geneva (Switzerland) World Health Organization 2006.
- World Health Organization. Global Tuberculosis Report 2017.
- Cobelens FG, van Deutekom H, Draayer-Jansen IW, Schepp-Beelen AC, van Gerven PJ, van Kessel RP, Mensen ME. Risk of infection with Mycobacterium tuberculosis in travellers to areas of high tuberculosis endemicity. Lancet 2000; 356: 461-465.
- Al-Jahdali H, Memish ZA, Menzies D. Tuberculosis in association with travel. Int J Antimicrob Agents 2003; 21: 125-130.
- Memish ZA, Ahmed QA. Mecca bound: the challenges ahead. J Travel Med 2002; 9: 202-210.
- Wilder-Smith A, Memish Z. Meningococcal disease and travel. Int J Antimicrob Agents 2003; 21: 102-106.
- Qureshi H, Gessner BD, Leboulleux D, Hasan H, Alam SE, Moulton LH. The incidence of vaccine preventable influenza-like illness and medication use among Pakistani pilgrims to the Haj in Saudi Arabia. Vaccine 2000; 18: 2956-2962.
- Wilder-Smith A, Barkham TM, Earnest A, Paton NI. Acquisition of W135 meningococcal carriage in Hajj pilgrims and transmission to household contacts: prospective study. BMJ 2002; 325: 365-366.
- Alzeer A, Mashlah A, Fakim N, Al-Sugair N, Al-Hedaithy M, Al-Majed S, Jamjoom G. Tuberculosis is the commonest cause of pneumonia requiring hospitalization during Hajj (pilgrimage to Makkah). J Infect 1998; 36: 303-306.
- Wilder-Smith A, Foo W, Earnest A, Paton NI. High risk of Mycobacterium tuberculosis infection during the Hajj pilgrimage. Trop Med Int Health 2005; 10: 336-339.
- American Thoracic Society, and the Centers for Disease Control and Prevention. Diagnostic standards and classification of tuberculosis in adults and children. Am J Respir Crit Care Med 2000; 161: 1376-1395.
- Streeton JA, Desem N, Jones SL. Sensitivity and specificity of a gamma interferon blood test for tuberculosis infection. Int J Tuberc Lung Dis 1998; 2: 443-450.
- Mazurek GH, Villarino ME. Guidelines for using the QuantiFERON-TB test for diagnosing latent Mycobacterium tuberculosis infection. MMWR 2003; 52: 15-18.
- Mazurek GH, Jereb J, Lobue P, Iademarco MF, Metchock B, Vernon A, Division of Tuberculosis Elimination, National Center for HIV, STD, and TB Prevention, Centers for Disease Control and Prevention (CDC). Guidelines for using the QuantiFERON-TB Gold test for detecting Mycobacterium tuberculosis infection, United States. MMWR Recomm Rep 2005; 54: 49-55.
- Pai M, Riley LW, Colford JM Jr. Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis 2004; 4: 761-776.
- Food and Drug Administration. QuantiFERON-TB Gold In-Tube. Available at http: //www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/PMAApprovals/ucm106548.htm.
- Food and Drug Administration. T-SPOT-TB 2019.
- Clinicians guide to QuantiFERON-TB. Cellestis Inc. 2001.
- Taylor RE, Cant AJ, Clark JE. Potential effect of NICE tuberculosis guidelines on paediatric tuberculosis screening. Arch Dis Child 2008; 93: 200-203.
- Detjen AK, Keil T, Roll S, Hauer B, Mauch H, Wahn U, Magdorf K. Interferon-gamma release assays improve the diagnosis of tuberculosis and nontuberculous mycobacterial disease in children in a country with a low incidence of tuberculosis. Clin Infect Dis 2007; 45: 322-328.
- Kampmann B, Whittaker E, Williams A, Walters S, Gordon A, Martinez-Alier N, Williams B, Crook A.M, Hutton AM, Anderson ST. Interferon-gamma release assays do not identify more children with active tuberculosis than the tuberculin skin test. Eur Respir J 2009; 33: 1374-1382
- Barnes PF. Diagnosing latent tuberculosis infection: turning glitter to gold. Am J Respir Crit Care Med 2004; 170: 5-6.
- Meier T, Eulenbruch HP, Wrighton-Smith P, Enders G, Regnath T. Sensitivity of a new commercial enzyme-linked immunospot assay (T SPOT-TB) for diagnosis of tuberculosis in clinical practice. Eur J Clin Microbiol Infect Dis 2005; 24: 529-536.
- Pai M, Gokhale K, Joshi R, Dogra S, Kalantri S,Mendiratta DK, Narang P, Daley CL, Granich RM, Mazurek GH, Reingold AL, Riley LW, Jr Colford JM. Mycobacterium tuberculosis infection in health care workers in rural India: comparison of a whole-blood interferon ? Assay with tuberculin skin testing. JAMA 2005; 293: 2746-2755.
- Diel R, Nienhaus A, Lange C, Meywald-Walter K, Forssbohm M, Schaberg T. Tuberculosis contact investigation with a new, specific blood test in a low-incidence population containing a high proportion of BCG-vaccinated persons. Respir Res 2006; 7: 77.
- Mazurek GH, LoBue PA, Daley CL, Bernardo J, Lardizabal AA, Bishai WR, Iademarco MF, Rothel JS. Comparison of a whole-blood interferon gamma assay with tuberculin skin testing for detecting latent Mycobacterium tuberculosis infection. JAMA 2001; 286: 1740-1747.
- Bellete B, Coberly J, Barnes GL, Ko C, Chaisson RE, Comstock GW, Bishai WR. Evaluation of a whole-blood interferon-gamma release assay for the detection of Mycobacterium tuberculosis infection in 2 study populations. Clin Infect Dis 2002; 34: 1449-1456.
- Bruins J, Gribnau JH, Bwire R. Investigation into typical and atypical tuberculin sensitivity in the Royal Netherlands Army, resulting in a more rational indication for isoniazid prophylaxis. Tuber Lung Dis 1995; 76: 540-544.
- QuantiFERON_-TB Gold (In Tube Method) package insert, Cellestis limited 2007.
- Pai M, Zwerling A, Menzies D. Systematic review: T-cell-based assays for the diagnosis of latent tuberculosis infection: an update. Ann Intern Med 2008; 149: 177-184.
- Brock I, Munk ME, Kok-Jensen A, Andersen P. Performance of whole blood IFN-gamma test for tuberculosis diagnosis based on PPD or the specific antigens ESAT-6 and CFP-10. Int J Tuberc Lung Dis 2001; 5: 462-467.
- Brock I, Weldingh K, Leyten EM, Arend SM, Ravn P, Andersen P. Specific T-cell epitopes for immunoassay-based diagnosis of Mycobacterium tuberculosis infection. J Clin Microbiol 2004; 42: 2379-2387.
- Mori T, Sakatani M, Yamagishi F, Takashima T, Kawabe Y, Nagao K, Shigeto E, Harada N, Mitarai S, Okada M, Suzuki K, Inoue Y, Tsuyuguchi K, Sasaki Y, Mazurek GH, Tsuyuguchi I. Specific detection of tuberculosis infection: an interferon-gamma-based assay using new antigens. Am J Respir Crit Care Med 2004; 170: 59-64.
- Ravn P, Munk ME, Andersen AB, Lundgren B, Lundgren JD, Nielsen LN, Kok-Jensen A, Andersen P, Weldingh K. Prospective evaluation of a whole-blood test using Mycobacterium tuberculosis-specific antigens ESAT-6 and CFP-10 for diagnosis of active tuberculosis. Clin Diagn Lab Immunol 2005; 12: 491-496.
- Chang PC, Wang PH, Chen KT. Use of the QuantiFERON-TB Gold In-Tube test in the diagnosis and monitoring of treatment efficacy in active pulmonary tuberculosis. Int J Environ Res Public Health 2017; 14: 236.