- Biomedical Research (2012) Volume 23, Issue 2
Variations in vitamin C levels in leukemias
K. N. Pujari1*, S. P. Jadkar1, S. N. Mashal2, G. J. Belwalkar3, Aruna kulkarni4, C. G. Patil5 and V. B. Tuljapurkar61Department of Biochemistry, Government Medical College, Miraj, India
2Department of Pathology, Government Medical College, Miraj, India
3Department of Biochemistry, Bharati Vidyapeeth Deemed University, Sangli, India
4Department of Biochemistry, Grant Medical College, Mumbai, India
5Department of Preventive and Social Medicine, Government Medical College, Miraj, India
6Department of Medical Oncology, Shri Shiddhivinayak Ganapati Cancer Hospital, Miraj, India
- *Corresponding Author:
- K. N. Pujari
Department of Biochemistry
Government medical College, Miraj 416410, India
Accepted date: January 27 2012
Abstract
Vitamin C is a six-carbon lactone that is synthesized from glucose in the liver of most mammalian species, but not by humans. Vitamin C is an electron donor and therefore a reducing agent. All known physiological and biochemical actions of vitamin C are due to its action as an electron donor and therefore it is good free radical scavenger. In humans, vitamin C acts as an electron donor for eight different enzymes. These vitamin C dependent enzymes required for collagen hydroxylation, synthesis of carnitine, norepinephrine, and peptide hormones and tyrosine metabolism. We have estimated the vitamin C levels in the plasma of the leukemic patients. The overall mean vitamin C levels in the leukemic patients were significantly low (0.25 ± 0.04) as compared to that of the normal control (0.90 ± 0.07). In respect to sex, the mean vitamin C levels in males (0.25 ± 0.04) were found to low than that in females (0.26 ± 0.04) however, we found significant difference only in patients with chronic leukemias. Whereas in respect to age, the mean vitamin C levels were found to elevated with increase in age (in < 10 years value was 0.23 ± 0.03, whereas in age >51 years value was 0.26 ± 0.04) indicating the extent of the free radicals might have stimulated the production of the antioxidant. We observed highly significant (p<0.001) vitamin C levels in all leukemic patients (AML, ALL, CML and CLL) with respect to age. Our results suggest that oxidative stress in leukemic patients causes the deficiency in antioxidant vitamin C, which arise as a result of enormous production of reactive oxygen species in the system.
Keywords
Leukemia, AML, ALL, CML, Vitamin C
Introduction
Vitamin C (ascorbic acid) is a six-carbon lactone that is synthesized from glucose in the liver of most mammalian species, but not by humans, non-human primates and guinea pigs. These species do not have the enzyme gulonolactone oxidase, which is essential for synthesis of the ascorbic acid immediate precursor 2-keto-lgulonolactone. The DNA encoding for gulonolactone oxidase has undergone substantial mutation, resulting in the absence of a functional enzyme [1,2]. Consequently, when humans do not ingest vitamin C in their diets, a deficiency state occurs with a wide spectrum of clinical manifestations. Clinical expression of vitamin C deficiency, scurvy, is a lethal condition unless appropriately treated. Thus, humans must ingest vitamin C to survive. Vitamin C is an electron donor and therefore a reducing agent. All known physiological and biochemical actions of vitamin C are due to its action as an electron donor. It donates two electrons from a double bond between the second and third carbons of the 6-carbon molecule. It is also called an antioxidant because, by donating its electrons, it prevents other compounds from being oxidized. However, by the very nature of this reaction, vitamin C itself is oxidized in the process. It is noteworthy that when vitamin C donates electrons, they are lost sequentially. The species formed after the loss of one electron is a free radical, semidehydroascorbic acid or ascorbyl radical. As compared to other free radicals (a species with an unpaired electron), ascorbyl radical is relatively stable with a half-life of 10-5 seconds and is fairly unreactive. This property explains why ascorbate may be a preferred antioxidant. In simple terms, a reactive and possibly harmful free radical can interact with ascorbate. The reactive free radical is reduced, and the ascorbyl radical formed in its place is less reactive. Reduction of a reactive free radical with formation of a less reactive compound is sometimes called free radical scavenging or quenching. Ascorbate is therefore a good free radical scavenger due to its chemical properties [3,4].
In humans, vitamin C acts as an electron donor for eight different enzymes [5]. At least for some of the enzymes, ascorbate adds electrons sequentially, with formation of the ascorbyl radical intermediate. Of the eight enzymes, three participate in collagen hydroxylation [6–8]. These reactions add hydroxyl groups to the amino acids proline or lysine in the collagen molecule, thereby greatly increasing stability of the collagen molecule triple helix structure. Two other vitamin C dependent enzymes are necessary for synthesis of carnitine [9,10]. Carnitine is essential for the transport of fatty acids into mitochondria for ATP generation. The remaining three vitamin C dependent enzymes have the following functions: one participates in the biosynthesis of norepinephrine from dopamine [11,12], one adds amide groups to peptide hormones, greatly increasing their stability [13,14], and one modulates tyrosine metabolism [15,16]. In this study we measured plasma vitamin C (ascorbic acid) levels in patients with leukemia.
Materials and Methods
Present study was carried out in the Department of Biochemistry, Government Medical College, Miraj and Department of Medical Oncology, Shri Siddhivinayak Ganpati Cancer Hospital, Miraj, Maharashtra (India). Study protocol was approved by ethical committee of Government Medical College, Miraj.
Sample Size
Study cases: The study group includes a total 191 subjects. This includes patients as well as control.
Patients
Total 131 patients with confirmed diagnosis of leukemia were selected for this study. The patients in the study were those who referred to Department of Medical Oncology, Shri Siddhivinayak Ganapati Cancer Hospital, Miraj.
Patients were grouped according to type of leukemia as;
1. AML ( Acute myeloid leukemia) : 36 patients
2. ALL (Acute lymphoblastic leukemia): 37 patients
3. CML (Chronic myeloid leukemia): 28 patients
4. CLL (Chronic lymphoblastic leukemia): 30 patients.
Control
The 60 healthy control were taken in all age groups with both genders (compared to leukemia patients) attending the OPD of the Shri Siddhivinayak Ganapati Cancer Hospital and Government Medical College and Hospital, Miraj during the same period.
The patients and healthy controls having history of smoking, alcoholism and other diseases which alters serum vitamin C concentration such as cancers, scurvy etc no such concurrent or past history of diseases were excluded from the study.
Collection of blood samples
Informed consent was obtained from the participants. Blood samples were collected from 131 patients with confirmed diagnosis of leukemia, attending Medical Oncology Department of Shri Siddhivinayak Ganpati Cancer Hospital. In addition blood samples were also obtained from 60 healthy age and sex matched randomly selected individuals from general population to serve as controls. A plasma vitamin C levels were measured by phosphotungstic acid reaction described by Ayekyaw [17] and the levels were expressed as mg/dl
The data were evaluated statistically by using student ‘t’ and ‘F’ test, ‘F’ value was calculated by Minitab and SPSS software.
Results
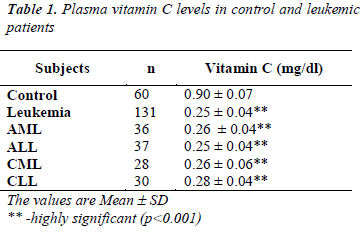
We investigated the levels of ascorbic acid in plasma of leukemic patients and healthy controls and are given in table 1 and level of it in all patients (AML, ALL, CML and CLL) were found to be significantly decreased (p<0.001) as compared to the control. Lowest decrease is found in ALL patients (Table 1).
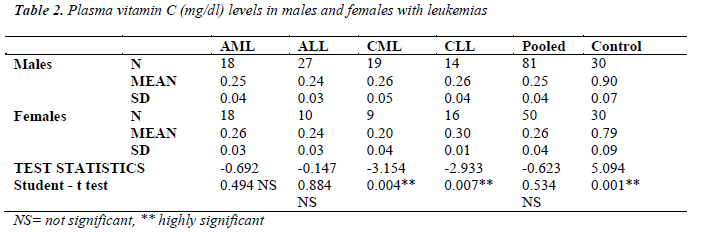
Table 2 shows the vitamin C levels in males and females with leukemias. The significant sex difference in serum vitamin C levels are observed only in chronic leukemias.
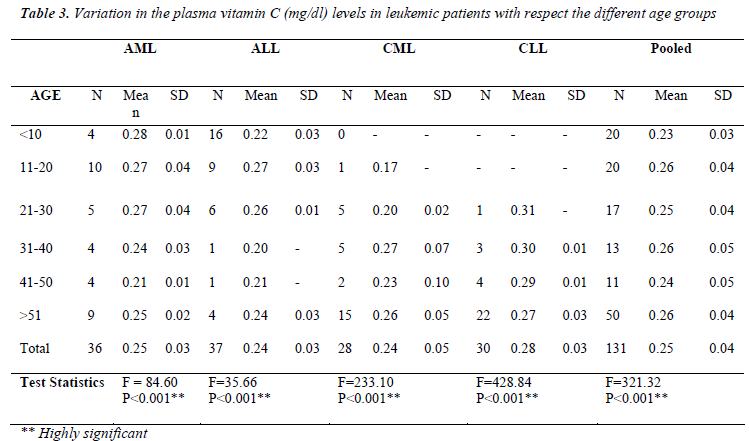
Table 3 shows the variation in serum vitamin C levels in leukemic patients with respect the different age groups. In the age group between less than 10 years, we found lowest mean vitamin C levels in pooled patients (0.23 ± 0.03 to 0.26 ± 0.04). We found highly significant (p<0.001) trend in vitamin C levels with respect to age in all leukemic patients (AML, ALL, CML, and CLL).
Discussion
Ascorbic acid (vitamin C) is one of the important and essential vitamin for human health. It is needed for many physiological functions in human biology. Many health benefits have been attributed to ascorbic acid namely antioxidant, anti-atherogenic and anti-carcinogenic activity. The relation between ascorbic acid and cancer is still a debatable as the molecular mechanism underlying anticarcinogenic activity of ascorbic acid is not clearly elucidated.
A significant lower level of ascorbic acid was found in plasma from patients with leukemia than normal control this may be due increased utilization (Table 1).
Cancer patients are significantly deficient of vitamin C; it indicates increased requirement and utilization of this substance to potenciate resistance mechanism like secondary anticancer mechanism of Vitamin C. The tendency of leukemic cells to proliferate is restrained by highly viscous intracellular glycosaminoglycans and to overcome the above situation cells releases enzyme hyaluronidase. For the synthesis of hyaluronides, vitamin C is utilized and hence total vitamin C levels may decrease [18].
It is hard clinical fact that leukemics have pathologically low levels of ascorbic acid in their blood plasma. It is not only the biochemical stresses of the leukemic disease which lowers the plasma ascorbic acid levels, but also the abnormally large volumes of white blood cells which scavenge and remove the remaining ascorbic acid from the blood serum. The white blood cells, which selectively absorb up to 40 times more ascorbic acid from the blood serum than red cells, do a fine job in removing the final traces of ascorbic acid from the blood serum and trap it within their cells and make it unavailable to the tissues [19].
Liebes et al (1981) studied ascorbic acid content in Blymphocytes of CLL cases and they showed higher levels of vitamin C in B-lymphocytes than normal [20].
The fact that there is no ascorbic acid left in the blood plasma deprives the tissues of the body of this most important and essential metabolite. The synthesis of collagen for maintaining the strength of the tissues, the blood vessels and capillaries is dependent on an adequate supply of ascorbic acid in the blood plasma. If the blood plasma is deficient in ascorbic acid, collagen production will stop and the mechanical strength of the tissues, blood vessels and capillaries will weaken and hemorrhage will result [19].
First line of defence against infection, phagocytosis is also an ascorbic acid dependent process. Phagocytosis is the gobbling up and digestion of invading bacteria in the blood stream and tissues by white blood cells. Under normal conditions, with good supply of ascorbic acid in the blood serum, any injury or bacteria getting into the tissues attracts hordes of white cells to the area; and they immediately go to work swallowing and digesting the bacteria and foreign material [19].
Leukemia is not the neoplastic disease itself, but hemorrhage and infection. This is a statistical fact. Both lack of resistance to infections and hemorrhaging are symptoms of scurvy. Leukemics are suffering from uncorrected hypoascorbemia or severe chronic subclinical scurvy in addition to leukemia; and they require high levels of ascorbic acid amounting to many grams per day to conquer their severe hypoascorbemia [19].
The significant sex difference in vitamin C levels are observed only in CML and CLL patients (table 2). Earlier, it was observed that total antioxidant status (TAS) was lower in females than in males; however, there was significant decrease in TAS levels with age in male but not in female indicating the genetic difference found in management of oxidative status [21]. This might explain the significant sex difference found in vitamin C levels in present study in the case of CML and CLL. Both types of acute leukemias (AML and ALL) have exhibited reduced vitamin C levels, though sex difference was not apparent. This indicates failure of antioxidant mechanism to counteract the extensive oxidative damage resulting in leukemia.
Table 3 shows the variation of vitamin C levels in the leukemic patients with different age groups. In the age group between 0-10 years and 41-50 years there is a sudden increase in the mean vitamin C levels of pooled patients. Whereas in the age 31-40 years there is sudden rise in vitamin C levels. Various studies have reported the process of aging associated with the degree of the antioxidant activity. Casado et al [22] reported the antioxidant enzyme were increased in the diseases of the aged individuals such as cardiovascular diseases, myomas, chronic obstructive pulmonary disease and acute cerebral accident, but the antioxidant enzyme SOD levels were seen to be decreasing with the process of aging. Whereas in another report [23] it has been shown that the excessive production of free radicals in the organism and the imbalance between the concentrations of these and the antioxidant defenses was related to the processes such as aging and the development of several diseases such as cancer. Alexander Michels [24] studied the effect of aging on Vitamin C Status. He observed that vitamin C levels appear to decline in the elderly; this has been observed not only in human plasma, but also in plasma and tissues in rats. Why do vitamin C levels decline with age? The most straightforward answer is that vitamin C uptake declines with age. In other words, our bodies don't have the same capacity to absorb vitamin C, so vitamin C levels in the plasma and tissues become lower. This has been supported by a recent meta-analysis, which showed that the elderly had much lower plasma vitamin C levels than younger adults following a given dose of vitamin C. This suggests that vitamin C absorption in the gut declines with age. Vitamin C absorption is governed by the same process that allows vitamin C to enter cells—membrane transport. The mechanism responsible for vitamin C transport in the gut and all other tissues is a Sodiumdependent Vitamin C Transporter known as SVCT. Two isoforms of this protein, SVCT1 and SVCT2, are known to exist in every mammal, including humans. In particular, nobody knows how expression or activity of this protein changes with age. He found that the decline in agerelated vitamin C levels is due to a loss of vitamin C transport, primarily due to changes in SVCT activity [23].
In our previous study the levels of oxidative stress markers- Malonyldialdehyde (MDA), SOD and catalase in patients with leukemia and we found increased MDA and decreased SOD and catalase levels [24,25]. In this way Vitamin C is utilized by proliferating malignant cells and also neutralizing oxidative stress markers and resulting in decrease levels of it.
Conclusion
Our study suggests that deficiency of Vitamin C in leukemia is due to increased utilization causing hypovitaminosis.
References
- Nishikimi M, Fukuyama R, Minoshima S, Shimizu N, Yagi. Cloning and chromosomal mapping of the human nonfunctional gene for L-gulono-gamma-lactone oxidase, the enzyme for Lascorbic acid biosynthesis missing in man. J Biol Chem 1994; 269: 13685-13688.
- Nishikimi M, Yagi K. Biochemistry and molecular biology of ascorbic acid biosynthesis. Subcell Biochem 1996; 25: 17-39.
- Buettner GR, Moseley PL, EPR spin trapping of free radicals produced by bleomycin and ascorbate. Free Radic Res Commun 1993; 19: S89-S93.
- Bielski BH, Richter HW, Chan PC. Some properties of the ascorbate free radical. Ann N Y Acad Sci 1975; 258: 231-237.
- Levine M, Rumsey SC, Wang Y, Park JB, Daruwala R. Vitamin C. In Stipanuk MH (ed): “Biochemical and Physiological Aspects of Human Nutrition.” Philadelphia: W B Saunders, pp 541-567, 2000.
- Prockop DJ, Kivirikko KI. Collagens: molecular biology, diseases, and potentials for therapy. Annu Rev Biochem 1995; 64: 403-434.
- Peterkofsky B. Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. Am J Clin Nutr 1991; 54: 1135S-1140S.
- Kivirikko KI, Myllyla R. Post-translational processing of procollagens. Ann N Y Acad Sci 1985; 460: 187-201.
- Rebouche CJ: Ascorbic acid and carnitine biosynthesis. Am J Clin Nutr 1991; 54: 1147S-1152S.
- Dunn WA, Rettura G, Seifter E, Englard S. Carnitine biosynthesis from gamma-butyrobetaine and from exogenous protein-bound 6-N-trimethyl-L-lysine by the perfused guinea pig liver. Effect of ascorbate deficiency on the in situ activity of ammabutyrobetaine hydroxylase. J Biol Chem 1984; 259: 10764-10770.
- Levine M, Dhariwal KR, Washko P, Welch R, Wang YH, Cantilena CC, Yu R: Ascorbic acid and reaction kinetics in situ: a new approach to vitamin requirements. J Nutr Sci Vitaminol 1992; (Tokyo) Spec No: 169-172.
- Kaufman S: Dopamine-beta-hydroxylase. J Psychiatr Res 1974; 11: 303-316.
- Eipper BA, Milgram SL, Husten EJ, Yun HY, Mains RE: Peptidylglycine alpha-amidating monooxygenase: a multifunctional protein with catalytic, processing, and routing domains. Protein Sci 1993; 2: 489-497.
- Eipper BA, Stoffers DA, Mains RE. The biosynthesis of neuropeptides: peptide alpha-amidation. Annu Rev Neurosci 1992; 15: 57-85.
- Englard S, Seifter S. The biochemical functions of ascorbic acid. Annu Rev Nutr 1986; 6: 365-406.
- Lindblad B, Lindstedt G, Lindstedt S: The mechanism of enzymic formation of homogentisate from phydroxyphenylpyruvate. J Am Chem Soc 1970; 92: 7446-7449.
- Ayekyaw. Simple colorimetric method for ascorbic acid in blood plasma. Clin. Chimica. Acta 1978; 86: 153-157.
- Chan A. Partners in defense: vitamin E and vitamin C. Can J Physiol Pharmacol 1993; 71: 725-731.
- Irwin S. Massive doses of ascorbic acid in the control of leukemic mortality. www.google.com.
- Liebes L, Krigel R, Kuo S, Nevrla D, Pelle E, Silber R. Increased ascorbic acid content in chronic lymphocytic leukemia B lymphocytes. Proc. Natl. Sci. USA 1981; 78 (10): 6481-6484.
- Wang XL, Rainwater DL, VandeBerg JF, Mitchell BD, Mahaney MC. Genetic contributions to plasma total antioxidant activity; Arterioscler Thromb Vasc Biol 2001; 21: 1190-1195.
- Cadado A, de la Torre R, Lopez-Fernandez E, Carrascosa D, Venarucci D. Departmento de Fisiopatologia Y. Superoxide dismutase and catalase levels in diseases of the aged. Gac Med Mex 1998 Sep-Oct; 134 (5): 539-544.
- Kesavulu MM, Rao BK, Giri R, Vijaya J, Subramanyam G. Alterations of antioxidant enzymes and oxidative stress markers in aging.Kasapoglu M, Ozben T. Apparao C. Exp Gerontol 2001; 36: 209.
- Alexander Michels. Aging Affects Vitamin C Status. www.google.com.
- Pujari KN, Kulkarni A, Tuljapurkar VB, Joshi RM, Mujawar A. Free radical and antioxidant enzymes in acute leukemia. The antiseptic April 2008, 105 (4): 168-169.
- Pujari KN, Aruna Kulkarni, Tuljapurkar VB, Joshi RM, Mujawar A. Lipid peroxidation and antioxidant enzymes in chronic leukemia. Spectrum: Journal of Medical Research. Jan- Dec 2007: 4 (1 & 2): 60-63.