Research Article - Biomedical Research (2017) Volume 28, Issue 19
Surgical therapeutic strategy for giant pituitary adenomas
Han-Shun Deng, Zhi-Quan Ding, Sheng-fan Zhang, Zhi-Qiang Fa and Qing-Hua Wang*
Department of Neurosurgery, Zhujiang Hospital, Southern Medical University, Guangzhou, PR China
- *Corresponding Author:
- Qing-Hua Wang
Department of Neurosurgery
Zhujiang Hospital
Southern Medical University, PR China
E-mail: wqh1123@126.com
Accepted date: September 14, 2017
Abstract
To discuss the methods and experience of surgical treatment for giant pituitary adenomas. A total of 246 giant pituitary adenomas were included from January 2004 to January 2015. Massive resection of giant pituitary adenomas was accomplished in all cases. Photon knife radiotherapy was given postoperatively, with follow-up for over a year. Among the included cases, 173 cases were cured, 57 cases were improved, and 16 cases relapsed. There were 115 cases who had transient profuse urination after surgery. Hypopituitarism occurred in 124 cases, most of which already had hypopituitarism before surgery. Ten cases had postoperative intracranial hemorrhage; 8 cases had intracranial infection; 2 cases had cerebrospinal fluid leakage; 36 cases had transient severe internal milieu disorder; 4 cases died because of intracranial hemorrhage and lung infection. Giant pituitary adenomas should be treated by surgery because of its invasiveness, but we do not advocate a complete removal of the tumor. Intraoperative protection of the pituitary stalk is crucial for preventing postoperative hypopituitarism. Postoperative radiotherapy is an important means to cure and prevent recurrence. Different surgical methods should be adopted according to the growth pattern of giant pituitary adenomas. Transsphenoidal surgery is fit for giant pituitary adenomas which grow within the sphenoid sinus. Transcranial surgery is appropriate for giant pituitary adenomas which grow into the suprasellar and parasellar regions. For those growing within the sphenoid sinus while penetrating diaphragma sellae and growing into the suprasellar and parasellar regions, transcranial surgery should be first performed to remove the portion of tumor in the suprasellar and parasellar regions. This will be followed by staging transsphenoidal surgery to remove the portion of tumor which grows within the sphenoid sinus and in the intrasellar region.
Keywords
Giant pituitary adenomas, Transcranial surgery, Transsphenoidal surgery, Diaphragma sellae
Introduction
Pituitary adenomas, as common benign tumors in the sellar region, arise from the anterior pituitary gland. Its incidence is about 1/100,000, accounting for 8%-15% of all intracranial tumors. Pituitary adenoma is next only to gliocytoma and meningoma in terms of prevalence [1]. Those larger than 4cm are defined as giant pituitary adenomas [2], which exhibit high invasiveness and generally invade the cavernous sinus, sclerotin, chiasma opticum and internal carotid artery. Therefore, surgery for giant pituitary adenomas is highly risky [3], and postoperative recurrence is very likely. However, surgery remains the preferred treatment for giant pituitary adenomas. In this study, 246 cases with giant pituitary adenomas who were treated by surgery from January 2004 to January 2015 were included. The choice of different surgical strategies was discussed.
Materials and Methods
Clinical data
There were 121 males and 125 females, with a male-to-female ratio of 0.97:1. The cases were aged 18-70 years old, with an average of 41. The course of disease was 1 months to 8 years, with an average of 3.5 months. Clinical manifestations included headache, sexual dysfunction, menstrual disorder, galactorrhea and acromegaly. All 246 cases had vision or visual field disturbance to varying degree, and 35 cases were combined with hydrocephalus and an apparent increase of intracranial pressure, accounting for 14.23%.
Radiologic examinations
All 246 cases received CT and MRI scan of the sellar region. All tumors were located in the sellar region and grew into the sphenoid sinus and suprasellar and parasellar regions. The tumors were above 4 cm in diameter.
Endocrinological examinations
Endocrinological indicators increased in 82 cases, accounting for 33.33%. There were 68 cases who had a mild increase in Prolactin (PRL) level (30-60 μg/L), accounting for 27.64%. These cases took bromocriptine for over 3 months before surgery, which did not result in an apparent shrinkage of tumors. Growth Hormone (GH) level increased (>30 μg/L) in 14 cases (5.69%), who did not receive any medication. Before surgery 87 cases had hypopituitarism, accounting for 35.37%.
Surgical treatment massive resection of tumors was performed for all 246 cases. Those with hypopituitarism received cortisol and thyroxine supplement before surgery. Tumor size, position, growth pattern and invasion to the surrounding tissues were assessed by preoperative CT and MRI. Different surgical strategies, namely, transcranial surgery, microscopic endonasal transsphenoidal surgery, and combined transcranial surgery with transsphenoidal surgery, were given depending on patients’ conditions (Table 1). All cases received local radiotherapy (photon knife) after surgery, with follow-up for over a year.
| Surgical approach | Cases (%) |
|---|---|
| Transpterional approach | 74 (30.08%) |
| Interhemispheric-third ventricle approach | 39 (15.85%) |
| Subfrontal approach | 15 (6.10%) |
| Transsphenoidal approach | 84 (34.15%) |
| Combined transcranial surgery with transsphenoidal surgery | 34 (13.82%) |
| Total | 246 (100%) |
Table 1. Choice of different surgical approaches for giant pituitary adenomas.
Results
Pathology results
Postoperative immunohistochemistry indicated prolactinomas in 19 cases, GH-secreting pituitary adenoma in 14 cases, Thyroid-Stimulating Hormone (TSH)-secreting pituitary adenomas in 13 cases and Follicle Stimulating Hormone (FSH) secreting pituitary adenomas in 3 cases.
Treatment outcome
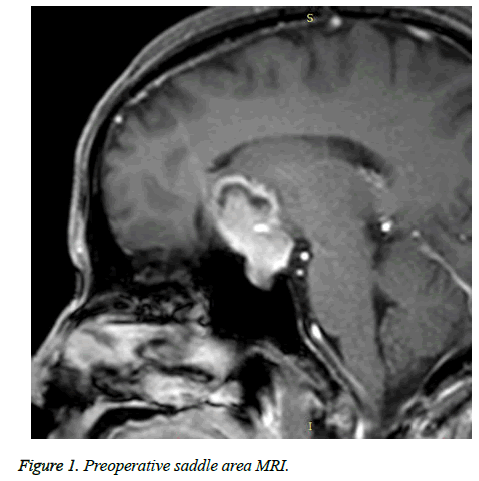
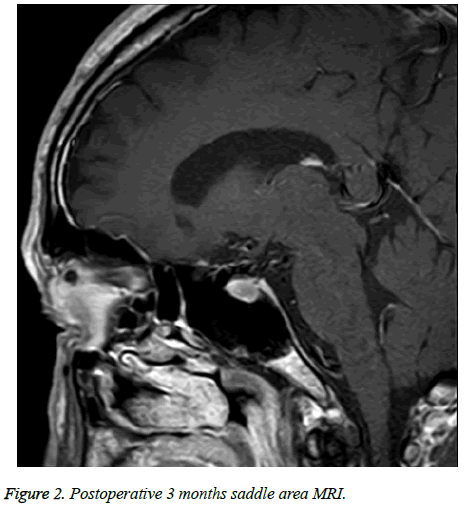
In accordance with Hardy’s criteria for surgical outcome for pituitary adenoma and patients’ recovery, outcome was divided into cured, improved and relapse. Of 246 cases, 173 cases were cured, 57 cases were improved and 16 cases relapsed (Table 2 and Figures 1 and 2).
| Approach/outcome | Cured | Improved | Relapse |
|---|---|---|---|
| Transcranial surgery (128 cases) | 96 cases (75.00%) | 24 cases (18.75%) | 8 cases (6.25%) |
| Microscopic endonasal transsphenoidal surgery (84 cases) | 51 cases (60.71%) | 25 cases (29.76%) | 8 cases (9.53%) |
| Combined transcranial surgery with transsphenoidal surgery (34 cases) | 26 cases (76.47%) | 8 cases (23.53%) | 0 case (0%) |
Table 2. Treatment outcome after surgeries using different approaches.
Postoperative complications
There were 115 cases who had transient profuse urination after surgery (46.75%), but none of them had permanent profuse urination; 124 cases had hypopituitarism after surgery (50.41%), and most of them already suffered from hypopituitarism before surgery; 10 cases had intracranial hemorrhage (4.07%); 8 cases had intracranial infection (3.25%); 2 cases had cerebrospinal fluid leakage (0.81%); 36 cases had transient severe internal millieu disorder (14.63%); 4 cases died (1.62%), including 3 cases with intracranial hemorrhage and 1 case with lung infection (Table 3).
| Approach/complication | Profuse urination | Electrolyte disturbance | Hypopituitarism | Intracranial infection | Intracranial hemorrhage | Cerebrospinal fluid leakage |
|---|---|---|---|---|---|---|
| Transpterional approach | 28 | 5 | 26 | 4 | 4 | 1 |
| Subfrontal approach | 5 | 5 | 4 | 0 | 3 | 0 |
| Interhemispheric approach | 10 | 0 | 14 | 0 | 0 | 0 |
| Transsphenoidal approach | 65 | 25 | 66 | 4 | 2 | 1 |
| Combined transcranial surgery with transsphenoidal surgery | 7 | 1 | 14 | 0 | 1 | 0 |
Table 3. Postoperative complications in giant pituitary adenomas (cases).
Discussion
The clinical treatment and basic research regarding pituitary tumors have undergone great advances in the past decade. Minimally invasive microneurosurgery and techniques for imaging diagnosis of pituitary tumors have been developed rapidly. Most pituitary adenomas can achieve good outcome after surgery. However, giant pituitary adenomas display high invasiveness and can invade peripheral nerves, blood vessels, brain and bones. Moreover, giant pituitary adenomas are associated with low total resection rate, high recurrence rate and high incidence of postoperative complications [4]. For giant pituitary adenomas, the proper choice of treatment and surgical approach remains one of the greatest challenges in neurosurgery.
Treatments for pituitary adenomas include medication, surgery and radiotherapy [5]. Giant pituitary adenomas can be hardly resected completely because of its invasiveness. The outcome of simple surgery alone is usually unsatisfactory, and adjuvant radiotherapy and medication are preferred after surgery [6].
Great progress has been made in medication for pituitary adenomas in recent years. Prolactinoma is the most common type of hormone-secreting pituitary tumor, for which dopamine receptor agonists are preferred. In China, the first-line drug for prolactinoma is bromocriptine. Cabergoline is used for those who are resistant or intolerant of bromocriptine to reduce the hormone level and tumor size [7]. Growth hormone receptor antagonists such as octreotide and lanreotide prove effective against GH- and TSH-secreting pituitary adenomas. We recommend medication as the first-line therapy for giant pituitary adenomas that do not cause cranial nerve compression or for patients who cannot tolerate surgery. But early surgery is recommended for those where the tumors already cause cranial nerve compression or who do not respond to medication.
Radiotherapy is an adjuvant therapy for pituitary adenomas if the surgery is performed with difficulty or if the recurrence risk is high [8]. Stereotactic gamma knife radiosurgery can reduce tumor size and lower surgical difficulty. Although it can help reduce the recurrence of pituitary adenomas, the risk of hypopituitarism, optic nerve injury and cerebral radiation necrosis still exists because of intensive gamma radiation [9]. The appropriate dose of radiotherapy depends on tumor type, tumor size, distance between the tumor and optic nerve and predicted risk of post-radiotherapy complications. The dose needed for improving the hormone levels is usually higher, and the duration of radiotherapy is longer. Moreover, radiotherapy usually achieves a better effect in non-functioning pituitary adenomas than in functioning pituitary adenomas [10]. When tumors already compress the optic nerve, the use of radiotherapy is usually restricted. It is generally believed that radiotherapy can be only safely applied when the distance between the tumor and optic chiasm is at least 3 mm. For these patients, we recommend surgical decompression or medication to reduce tumor size and to increase the distance from the optic chiasm before gamma knife radiation. Study has shown that the appropriate peripheral dose in gamma knife radiation is 15 Gy and that the incidence of hypopituitarism increases significantly above this threshold [11]. Photon knife is a threedimensional conformal radiation, which provides uniform irradiation of tumors at a high dose, thus reducing the injury to adjacent normal tissues and preserving the normal tissues [12]. The radiation dose for the periphery of the tumors should not be lower than 12 Gy and that for optic nerve and optic chiasm should not be lower than 10 Gy. Otherwise, the tumors can be hardly controlled, and the risk of vision and visual field injury increases.
Only complete resection under the microscope is possible for pituitary adenomas. In our study, all 246 cases received massive resection, followed by photon knife radiation 1-2 months after surgery. Sixteen cases relapsed, accounting for 6.50%. Eight cases received simple transcranial surgery and another eight cases received endonasal transsphenoidal approach. Thus for giant pituitary adenomas with high invasiveness, postoperative photon knife radiation is necessary to reduce recurrence.
The surgical approaches for pituitary adenomas fall into three categories: transcranial, transsphenoidal and combined approach. Transcranial surgery usually adopts subfrontal, transpterional or interhemispheric approach. The subfrontal approach is fit for pituitary adenomas that grow into the suprasellar region and dorsum sellae. Transpterional approach is usually used for pituitary adenomas that grow into the temporal lobe or parasellar region. For those located in the base of the third ventricle in the suprasellar region, the interhemispheric approach is more advantageous. Study has shown that about 90% of the pituitary adenomas can be resected via the endonasal transsphenoidal approach [13]. The endonasal transsphenoidal surgery is usually performed endoscopically or neuroendoscopically to avoid damage to the cavernous sinus, internal carotid artery and optic nerve and to reduce surgical trauma, complications and length of stay in hospital [14]. However, the endonasal transsphenoidal approach is difficult to expose and resect the suprasellar tumors. This is especially true for pituitary adenomas that are located in the suprasellar region, grow into the parasellar region or into the anterior fossa basilar and temporal lobe. However, if the portion of tumor located in the intrasellar region is very small, it can be hardly resected by surgery.
We suggest that the pear-shaped giant pituitary adenomas growing into the sphenoid sinus should be treated via the endonasal transsphenoidal approach. For the inverted pearshaped adenomas that grow into the suprasellar and parasellar regions, the transcranial surgery will be more appropriate. In our study, 128 patients had inverted pear-shaped giant pituitary adenomas, who were treated by the transcranial surgery. The cure rate was 75.00% (96/128), and improvement rate was 18.75% (24/128), and the recurrence rate was 6.25% (8/128) among these 128 patients. Endoscopic transsphenoidal approach was performed in 84 cases who had pear-shaped giant pituitary adenomas. Among them, the cure rate was 60.72% (51/84), the improvement rate was 29.76% (25/84), and the recurrence rate was 9.52% (8/84).
For giant pituitary adenomas that grow into the sphenoid sinus and penetrate the diaphragma sellae to go into the suprasellar and parasellar regions, we believe that the combined transcranial surgery with transsphenoidal surgery can achieve a complete resection of the tumors. All 34 cases who had such pituitary adenomas received combined transcranial surgery with transsphenoidal surgery. The cure rate was 76.47% (26/34) and the improvement rate was 23.53% (8/34), and none of the cases relapsed. Based on our clinical experience we suggest that transcranial surgery should be performed first to resect the suprasellar and parasellar lesions, which is followed by the staging transsphenoidal surgery to resect the lesions in the sphenoid sinus and intrasellar region. If the lesions in the sphenoid sinus and intrasellar region are first resected, there may be the risk of suprasellar lesion collapsing into the intrasellar region and compressing the diaphragma sellae, which can cause pituitary apoplexy. Moreover, the involvement and separation of suprasellar lesion with or from the brain tissues above it are likely to cause intracranial hemorrhage.
Conflict of Interest
The authors have declared that no competing interests exist.
References
- Cox L, Nelson H, Lockey R, Calabria C, Chacko T. Allergen immunotherapy: a practice parameter third update. J Allergy Clin Immunol 2011; 127: S1-55.
- Gondim JA, Almeida JP, Albuquerque LA, Gomes EF, Schops M. Giant pituitary adenomas: surgical outcomes of 50 cases operated on by the endonasal endoscopic approach. World Neurosurg 2014; 82: 281-290.
- Baumann F, Schmid C, Bernays RL. Intraoperative magnetic resonance imaging-guided transsphenoidal surgery for giant pituitary adenomas. Neurosurg Rev 2010; 33: 83-90.
- Sinha S, Sharma BS. Giant pituitary adenomas-an enigma revisited. Microsurgical treatment strategies and outcome in a series of 250 patients. Br J Neurosurg 2010; 24: 31-39.
- Ding D, Starke RM, Sheehan JP. Treatment paradigms for pituitary adenomas: defining the roles of radiosurgery and radiation therapy. J Neurooncol 2014; 117: 445-457.
- Yutaka OKI. Medical management of functioning pituitary adenoma: an update. Neurol Med Chir (Tokyo) 2014; 54: 958-965.
- Ogilvie CM, Milsom SR. Dopamine agonists in the treatment of prolactinoma: are they still first choice? Intern Med J 2011; 41: 156-161.
- Cortet-Rudelli C, Bonneville JF. Post-surgical management of non-functioning pituitary adenoma. Ann Endocrinol (Paris) 2015; 76: 228-238.
- Sheehan JP, Starke RM, Mathieu D, Young B, Sneed PK. Gamma Knife radiosurgery for the management of nonfunctioning pituitary adenomas: a multicenter study. J Neurosurg 2013; 119: 446-456.
- Minniti G, Clarke E, Scaringi C, Enrici RM. Stereotactic radiotherapy and radiosurgery for non-functioning and secreting pituitary adenomas. Rep Pract Oncol Radiother 2016; 21: 370-378.
- Mapek J, Jezkova J, Hana V. Is it possible to aviod hypopituitarism after irradiation of pituitary adenomas by the Le Ksell gamma knife. Eur J Endocrinol 2011; 164: 169-178.
- Toossi MT, Sabet LS, Soleymanifard S. A comparison of the doses received by normal cranial tissues during different simple model conventional radiotherapeutic approaches to pituitary tumours. Australas Phys Eng Sci Med 2016; 39: 517-524.
- Morgenstern LB, Hemphill JC, Anderson C. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2010; 41: 2108-2129.
- Chao-Hung K, Yu-Shu Y, Jau-Ching W. Primary endoscopic transnasal transsphenoidal surgery for giant pituitary adenoma. W Neurosurg 2016; 121-128.