- Biomedical Research (2010) Volume 21, Issue 1
Salivary malondialdeyde and antioxidant status in oral squamous cell carcinoma patients and smokers
Arathi A1, Benedicta D’Souza*, Sayanthan M, Raksha S, Buthesh GA, Jisha K, Hegde MC1, Vivian D’SouzaDepartment of Biochemistry, Centre for basic sciences, Kasturba Medical College, Manipal University, Bejai, Mangalore, India.
1Department of ENT, KMC Hospital, Attavar, Mangalore, India.
- Corresponding Author:
- Benedicta D’Souza
Department of Biochemistry
Centre for Basic Sciences
Kasturba Medical College, Manipal University
Bejai, Mangalore 575004
India.
E-mail: benedicta_7@yahoo.com
Accepted Date: June 03 2009
Abstract
Free radical mediated oxidative stress has been implicated in pathogenesis of Oral Squamous Cell Carcinoma ( OSCC). The present study was conducted in saliva to estimate the level of lipid peroxidation product- malondialdehyde ( MDA) and antioxidants uric acid, vitamin C, and total antioxidant activity in 25 OSCC patients. The results were compared with age and sex matched 25 smokers and 25 normal healthy control subjects. pH of the saliva was also measured in all the study groups. Highly significant increase in MDA and decrease in antioxidants was observed in OSCC patients and smokers. pH of saliva decreased significantly in smokers. This study suggests that there is increased oxidative stress and antioxidant defense mechanism is impaired in OSCC patients and smokers. Supplementation of antioxidants might be beneficial to reduce the oxidative stress in OSCC patients and smokers.
Keywords
Malondialdehyde, Antioxidants, Oral squamous cell carcinoma, Smokers
Introduction
Oral sqamous cell carcinoma is the sixth most common cancer in men and the 14th most common cancer in women. On an average only half of those diagnosed with this disease will survive more than five years [1,2]. Free radicals such as reactive oxygen species (ROS) and reactive nitrogen species(RNS), which induce oxidative and nitrative stress are principal inducers of OSCC. ROS such as superoxide radicals (O2), hydroxyl radicals(OH.) hydrogen peroxide (H2O2) and RNS in the form of nitrosamines (NO3 and NO2) play a key role in human cancer development [3]. Reactive free radicals are able to damage proteins, lipids, carbohydrates and nucleotides in the tissue. ROS can cause DNA base alterations, strand breaks, damage tumor suppressor genes and enhance expression of proto oncogenes [4]. The OSCC inducing ROS and RNS originate mainly from smoking, alcohol, food and drink which enter into the oral cavity through the mouth. The oral cavity is armed with salivary antioxidant system containing both enzymatic and nonenzymatic components including peroxidase, superoxide dismutase, glutathione s-transferase and uric acid [5].
Saliva is a diagnostic tool for many oral and systemic diseases [6,16]. Moreover reports on salivary antioxidants are very scanty. Therefore the current study was planned to evaluate the oxidative stress and antioxidant profile of the saliva in OSCC patients and smokers. Malondialdehyde (MDA) was estimated as the marker of lipid peroxidation. Uric acid, vitamin C and total antioxidant activity (TAA) was measured to assess the antioxidant status of OSCC patients ,smokers and controls.
The salivary pH test is a simple test to measure the susceptibility of cancer, heart disease and many other degenerative diaseases. Since the pH of saliva offers us a window through which we can see the overall pH balance in the body, we have also measured the pH of saliva of all the subjects enrolled in the present study.
Materials and Methods
This study was conducted on patients presenting to the Otorhinolaryngology OPD in Kasturba Medical College Hospital, Attavar and Government Wenlock Hospital, Mangalore, between the periods of July 2005 and July 2007. The study was approved by Institutional Ethical Committee of KMC Mangalore. Three groups of people were enrolled in the study after informed consent was obtained from the patients. There were 25 subjects in each group and hence a total of 75 subjects were studied.
All the patients were subjected to an ENT examination. From all the subjects saliva was collected and subjected to biochemical analysis.
Group I is the Control group: 25 healthy subjects with no history of tobacco related habits, no history of alcohol consumption and no associated lesions between the age of 46-65years of both sexes.
Group II is the Smokers group: 25 subjects of the same age and sex, with history of smoking more than 10 cigarettes per day for a period of 10-20years with no history of alcohol consumption and no associated lesions.
Group III is the Malignant group: 25 age and sex matched OSCC patients. All the 25 cases of biopsy proven malignancies were squamous cell carcinomas. There were 15 cases of moderately differentiated OSCCs and 10 were well differentiated OSCCs. None of the cases were poorly differentiated OSCC.
Tumour staging
16 out of the 25 cases (64%) fell into the category of stage IV, while 5 cases were stage III and only 4 cases were stage II. None of the cases had presented in stage I of the malignancy.
Exclusion criteria
Patients with head and neck malignancies, cases with salivary gland, nasopharyngeal malignancies, cases with history of chronic alcohol consumption were excluded from the study
Saliva collection
Saliva was collected shortly after the diagnosis and before administration of definitive therapy. All the subjects enrolled in the study were asked to rinse their mouth with 10ml of water and saliva was collected in sterile bottles and was used for biochemical analysis.
pH: Intra oral pH of patients and controls were measured by the quantitative litmus paper test.The litmus paper was inserted into the saliva and the reading was taken by noting the color change. The color was compared with the pH chart
The colour blue indicated an alkaline pH while pink indi-cated an acidic pH.
MDA: Lipid peroxidation product-MDA was analysed by thiobarbituric acid(TBA) reaction[7].
Uric acid: This was estimated by uricase method of Ka-basakalian et al. [8].
Vitamin C (Ascorbic Acid): This was measured by 2,4-Dinitrophenyl hydrazine method [9].
Total antioxidant activity (TAA): This was measured by the method of Koracevic D et al [10].
Statistical analysis
Data was analysed using the SPSS statistical software. The analysis of the variants (ANOVA) was done to compare the parameters between malignant cases, smokers and control. Chi square test, Kruskal wallis test was done to analyse the data.
Results
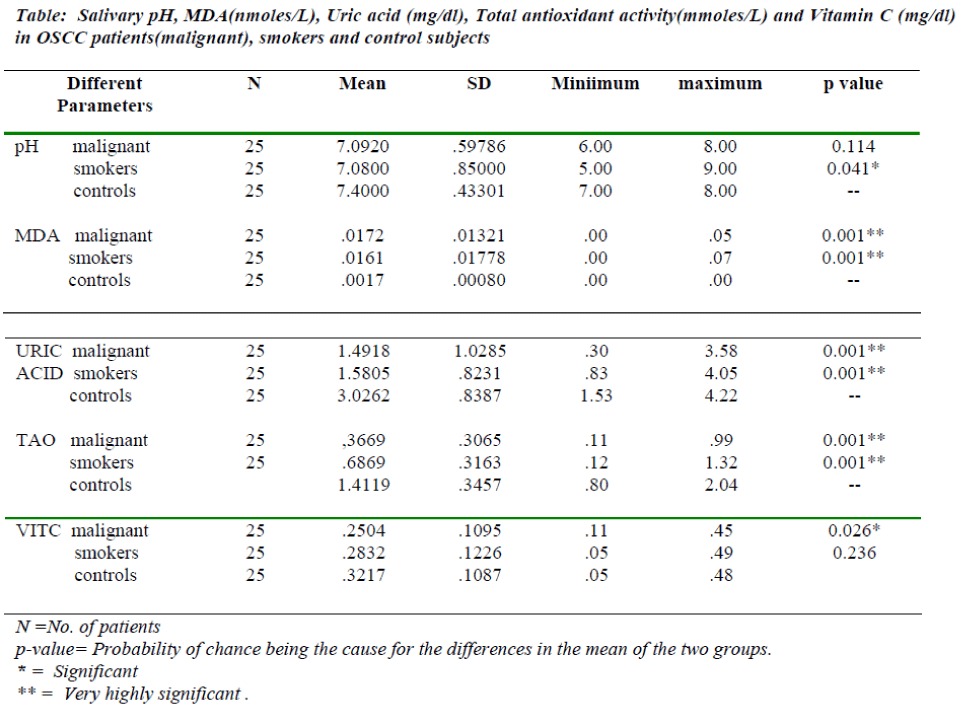
The mean pH of saliva of OSCC patients, smokers and normals is given in the table 1. Decrease in salivary pH found in smokers group is statistically significant when compared to controls where as it not significant in OSCC patients. Salivary MDA is highly increased in OSCC patients & smokers when compared to normal subjects. Salivary vitamin C decreased significantly in OSCC patients compared to control subjects where as in smokers group, the decrease in vitamin C is not very significant. Salivary Uric acid and TAO activity is significantly decreased in OSCC patients and smokers when compared with control subjects (Table 1).
Discussion
The present study indicates that there is decrease in saliva pH in OSCC patients and smokers when compared to control subjects. When healthy, the pH of blood is 7.4, the pH of spinal fluid is 7.4. In our study we have found that the pH of saliva is 7.4 in normal subjects. Thus the pH of saliva parallels the extracellular fluid pH. The pH of the healthy person is in the slightly alkaline range. All degenerative diseases including cancer, heart disease, osteoporosis, arthritis, kidney and gall stones and tooth decay are associated with excess acidity in the body. The body has a homeostatic mechanism which maintains a constant pH 7.4 in the blood by depositing and withdrawing acid and alkaline minerals from other locations including body fluids and saliva. Therefore pH of saliva offers us a window through which we can see overall pH balance in our bodies. In our study we have found decrease in saliva pH(7.09) in OSCC patients and smokers(7.08), where as pH of normal healthy control subjects remains 7.4 [11].
We have also found in our study that malondialdehyde which is a marker for lipid peroxidation is greatly increased in the saliva of OSCC patients and smokers. The salivary antioxidants like Uric acid, Vitamin C and total antioxidant activity are significantly reduced in OSCC patients and smokers. Increase in MDA indicates that there is increase in lipid peroxidation and free radical generation. Oxidative damage caused by the free radicals is thought to be a basic mechanism underlying many diverse pathological conditions such as diabetes, malaria, atherosclerosis, central nervous system disorders, carcinogenesis and aging[12,13,14,15].
Reactive free radicals are able to produce chemical modifications and damage to proteins, lipids ,carbohydrates and nucleotides in the tissues. It is also known that free radicals are mediators for tissue damage in neoplastic diseases[16]. The development of cancer is multifactorial, depending on the extent on DNA damage that is proportional to the magnitude of oxidative stress. This oxidative stress reflects the net effect of ROS on the one hand and the effectiveness of antioxidant defense on the other. ROS is involved in the initiation and promotion of multistep carcinogenesis but it is inhibited by antioxidants[17]. When this equilibrium is broken either by a reduction in the levels of antioxidants or by enhancement of ROS, the DNA is oxidized and cancer evolves. In our study we have found that MDA which is the marker for oxidative stress is highly increased in OSCC patients. Moreover the antioxidants like uric acid , vitamin c and total antioxidant activities are greatly reduced in these patients. Uric acid which is the end product of purine metabolism has been regarded as the potent endogenous water soluble antioxidant in saliva and free radical scavenger in humans[18]. Uric acid is particularly effective in quenching hydroxyl superoxide and peroxynitrite radicals and may serve a protective role by preventing lipid peroxidation[19]. Uric acid and total antioxidant activity is significantly reduced in OSCC patients which indicates that antioxidant defense is impaired in the saliva of oral squamous cell carcinoma patients
Our study also indicates that uric acid and total antioxidant activity is highly decreased and MDA levels are significantly increased in the smokers group. The OSCC including ROS may originate mainly from smoking . Naglar et al[20] states that exposure to cigarette smoke/ tobacco is responsible for 90% cases of oral cancer. There is a constant and direct attack of various cigarette smoke reagents on the oral epithelial cells, which gradually accumulate and cause a stepwise malig transformation . It has been suggested that ROS in the inhaled cigarette smoke induce this process initially expressed by dysplastic lesions of the mucosa. These are then transformed into insitu carcinoma lesions and eventually result in full blown metastasizing OSCC [21,22]. In line with these authors we have also found that salivary MDA is very high in smokers and OSCC patients which indicates that there is increase in production of ROS.
There is also a decrease in antioxidant profile as it is evident from decreased uric acid ,vitamin c, and total antioxidant activity. Therefore treatment with antioxidants like vitamin E, vitamin C might be beneficial to reduce MDA in smokers and OSCC patients.
References
- Myers JN, Elkins T, Roberts D, Byers RM. Squamous cell carcinoma of the tongue in adults: increasing incidence and factors that predict treatment outcomes. Otolaryngol Head Neck Surg 2000;122: 44-51
- Hinerman RW, Mendenhall WM, Morris CG, Amdur RJ, Werning JW, Villaret DB. Post operative irradiation for sqamous cell carcinoma of the oral cavity: 35 years experience. Head Neck 2004; 26: 984-994.
- Ma N, Tagawa T, Hiraku Y, Murata M, Ding X, Kawanishi S. 8-Nitroguanine formation in oral leukoplakia, a premalignant lesion. Nitric oxide 2006; 14: 137-143
- Liu RH, Hotchriss JH. Potential genotoxicity of chronically elevated nitric oxide: a review. Mutat Res.1995; 339: 73-89
- Nagler RM, Klein I, Zarzhevsky N, Drigues N, Reznick AZ. Characterisation of differentiated anti oxidant profile of human saliva. Free Radic. Biol.Med. 2002; 32: 268-277.
- Guyton AC, John E. Hall. Text book of medical physiology. WB Saunders Co. 11th Ed. 2006; p 738-740.
- Stocks J, Dormandy TL. The Auto oxidation of human red cells lipid reduced by hydrogen peroxide. Brit J. Hematol 1971; 20: 95-111.
- Kabasakalian P, Kalliney S, West Cott A. Determination of uric acid in serum with the use of uricase and a tribromophenol amino antipyrene chromogen. Clin Chem. 1973;19: 522-4
- Teitz NW. Methods of determination of ascorbic acid. In : Text book of clinical chemistry edited by N.W Teitz, WB Saunders company, Philadelphia, London, Toronto 1986; p960-962
- Koracevic D, Koracevic G, Djordjevic V, Andrejevic S, CoskV. Method for the determination of antioxidant activity in human fluids. J Clin.Pathol. 2001; 54: 356-361 .
- Scott WG-Brown. Otorynolaryngology, Basic Sciences 6th edition (Ed. Allan G. Kerr) Anatomy and physiology of salivary glands. 1997; Vol.1: 9, p 1-16
- D?Souza B, D?Souza V. Oxidative injury and antioxidant vitamins E and C in Schizophrenia. Ind . J.clin.Biochem.2003,18(1) p 87-90.
- Akila, D?Souza B, Vishwanath P, D?Souza V. Oxidative injury and antioxidants in coronary artery bypass graft surgery: off pump CABG significantly reduces oxidative stress. Clinica Chemica Acta 2007;375: 147-152
- Prasannachandra, D?Souza V, D?souza B. Comparative study in lipid peroxidation and anti oxidant vitamins E and C in falciparum and vivax malaria. Ind. J. of Clin. Biochem.2006;21(2): 103-106
- Akila, Vishwanath P, Harishchandra H, D?souza V, D?Souza B. Age related changes in lipid peroxidation and anti oxidants in elderly people. Ind.J.Clin. Biochem 2007;22(1): 131-134.
- Balwant R, Simmi K, Rajanish J, Anand SC. Salivary lipid peroxidation product malonaldehyde in various dental diseases. World J Med.Sci 2006; 2: 100-101
- Sun Y. Free radicals, anti oxidant enzymes and carcino-genesis. Free Radic. Biol. Med.1990; 8: 583-8599
- D?Souza B, D?Souza V. Sowmya S, Seema G, Hemalatha CN, Anu M and Sanjana G. A comparative study on oxidative stress and antioxidant status in ischemic stroke patients with and without diabetes. Ind .J of Clin.Biochem. 2008; 23 (3): 218-222.
- Ames BN, Cathcart R,Schweisrs E, Hochstein P.Uric acid provides an antioxidant defense in humans against oxidant and radical caused ageing and cancer: a hypothesis. Proc.Natl.Acad.Sci USA. 1981; 73: 6852-6862.
- Nagler R, Reznick A. Cigarette smoke effects an salivary antioxidants and oral cancer. Novel concepts. IMAJ 2004; 6: 691-694.
- Hershkovich O, Shafat I, Nagler R M, Nagler R, Lis-chinsky S, Diamond E, et al. Age related changes in salivary antioxidant profile: possible modifications for oral cancer. J. gerontal A Biol. Sci Med Sci 2007; 62 (4): 361-366
- Bahar G, Feinmesser R, Shpitzer T, Popovtzer A, Nagler RM. Salivary analysis in oral cancer patients. DNA and protein oxidation, reactive nitrogen species and antioxidant profile. Cancer 2007; 109 (1): 54-59