- Biomedical Research (2014) Volume 25, Issue 4
Retinal nerve fiber layer thickness in cases with mild cognitive impairment and Alzheimer -Type Dementia.
Gokcen Akar1*, Eren Gozke1, Yasemin Agirman2 and Sevda Aydin Kurna21Fatih Sultan Mehmet Education and Research Hospital, Department of Neurology
2Fatih Sultan Mehmet Education and Research Hospital, Department of Ophtalmology
Accepted date: August 05 2014
Abstract
The aim of this study was to compare mean retinal nerve fiber layer thickness (MRNFLT) in cases with mild cognitive impairment (MCI) and mild and moderate Alzheimer type dementia (ATD) with normal individuals. We evaluated 54 eyes of 27 patients with the diagnosis of mild and moderate ATD and MCI and 32 eyes of 16 people, as control group who are compatible with the other age group. Diagnosis of ATD was made according to Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) and diagnosis of MCI was made according to Petersen criteria. Mini mental state examination (MMSE) was applied to patient and control group. There was no significantly difference in terms of age and gender between groups. MRNFLT were significantly decreased in MCI group than control group. In general patient group MRNFLT levels were not significantly lower than the control group, but general trend was a decrease direction. Correlation was not detected between cognitive decline and MRNFLT loss degree. Statistically significant difference was not observed in MRNLFT values, between MCI, mild and moderate ATD patient groups. RNFLT measurement with HRT is an easy method which may be useful to show the retinal effects of the disease in patients with MCI and mild and moderate Alzheimer type dementia. Our data show that; RNFLT is reduced in patients with MCI. However, the increase in the severity of cognitive decline does not effect on RNFLT.
Keywords
Alzheimer-Type Dementia, Mild Cognitive Disorder, Heidelberg Retinal Tomography (HRT), Retinal fiber layer thickness
Introduction
Dementia refers to a general loss in cognitive functions. In degenerative dementia, a progressive deterioration in cognition, and functionality is observed. Alzheimer- type dementia (ATD) is the most frequently seen type of dementia, a progressive neurodegenerative disease, resulting in deterioration in cognitive functions, decrease in the daily living activities, behavioural, and psychologic disorders [1].
Mild cognitive ımpairment (MCI) is defined as a clinical state between normal aging, and ATD characterized by mild memory decline, but preservation of other cognitive functions, and maintenance of daily living activities. This syndrome may be considered as an early stage of ATD. Although, these people experience episodes of amnesia more frequently than expected from their age, still diagnostic criteria of ATD are not met. Nowadays, people diagnosed as MCI are believed to have a higher risk of suffering from ATD [2]. Terminologically, ATD refers to a clinical diagnosis. However, definition of Alzheimer’s disease (AH) includes histopathologically confirmed cases.
As patient ages cognitive, and visual functions decline, and daily living activities are impaired. Studies performed up to now have linked decline in visual functions in ATD, to degenerative changes in primary visual cortex. Besides, some studies have demonstrated loss of retinal ganglion cells, and their axons as an evidence of precortical involvement [3].
Heidelberg retinal tomography test (HRT) is a noninvasive, immediately, and easily applicable modality which measures amplitudes of laser beams sent to the optic disc, and retina surface which back scatter from these tissues to the device [4].
In this study, our aim was to measure retinal nerve fiber layer thickness (RNFLT) using HRT in patients diagnosed as ATD, and MCI, and compare the values obtained with those of the normal subjects.
Materials and Methods
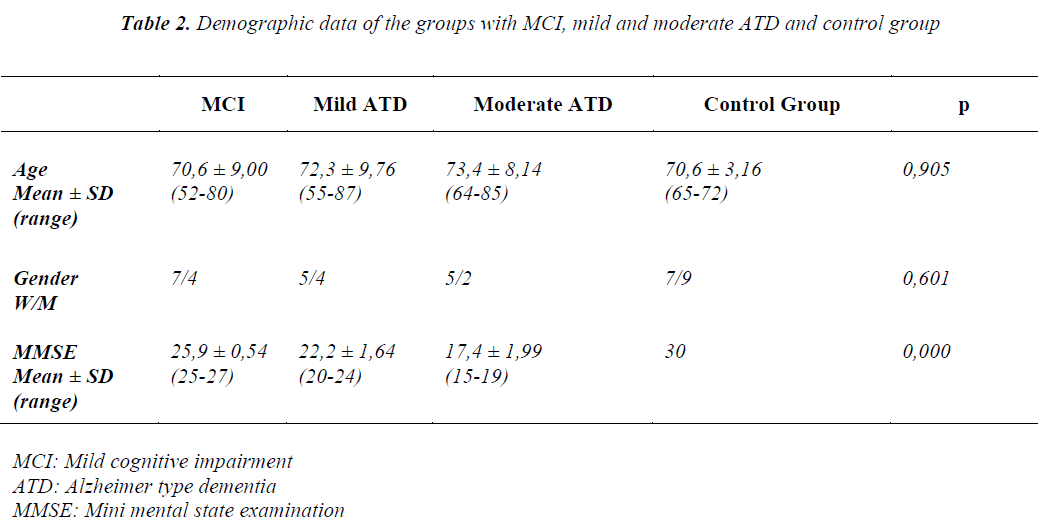
We evaluated 54 eyes of a total of 27 patients (10 men, and 17 women) with established diagnoses of ATD, and MCI based on DSM -IV (Diagnostic and Statistical Manual of Mental Disorders, 4th edition) , and Peterson’s MCI criteria, respectively [5-8]. Mini-mental state test (MMSE) was used to evaluate cognitive decline. The patients were evaluated, and categorized in 3 groups based on MMSE scores as MCI (n=11; 25-27 pts), mild (n=9; 20-24 pts), and moderate (n=7; 19-11 pts) ATD. As a control group, 32 eyes of 16 healthy (9 men, and 7 women) participants with MMSE scores of 30 pts were included in the study. During the selection process of the patient, and the control groups, cases with ocular disease, and systemic diseases which might affect visual functions were excluded from the study.
All cases underwent detailed neurological examinations. Their routine laboratory tests, and cranial magnetic resonance imagings (MRI) were evaluated. Patients with metabolic, nutritional, and alcoholism –related diseases, psychiatric diseases, and other neurological disorders were excluded from the study. An opthalmologist performed visual acuity, refraction, ocular motility, pupillar, and fundal tests, and evaluated intraocular pressure (IOP). Visual acuities of all cases were over 5/10 dioptres with IOPs lower than 22 mm Hg. Cases with macular degeneration, glaucoma, and cataract were not included in the study.
Optical nerve head analysis was conducted using a confocal scanning laser opthalmoscope [Heidelberg Retina Tomograph 3 (HRT 3, Heidelberg Engineering GmbH 69221 Dossenheim Germany)]. Preprocedural midriasis was not induced. During scanning, lens of the camera was adjusted so that the blue bars were set at maximal or nearly maximal luminance in order to obtain a clear image of the retinal vasculature.
Scanning was performed at an angle of 15 x 15° on a reference plane of 50 microns. The device yielded 64 successive images with a higher spatial resolution of were performed using an image analysis program. Standard printouts of the HRT were obtained, and mean RNLT was recorded in millimetres. Incomplaint patients were excluded from the study.
Statistical analysis
Statistical analysis was performed using an SPSS 18.0.0 package program. For the evaluation of data, descriptive statistical methods (mean, standard deviation), and for intergroup comparisons Kruskal Wallis, and Mann Whiney U tests were used. P< 0.05 was considered as the level of significance.
Results
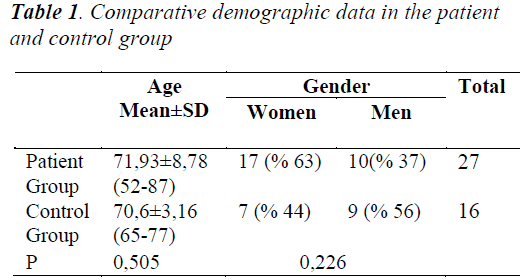
Mean RNFLT values of 16 healthy persons (32 eyes), and a total of 27 patients (54 eyes) evaluated as having diagnoses of MCI (n=11), mild (n=9) , and moderate (n=7) ATD were measured. Mean ages of the patient, and the control groups were 71,9 ± 8,7, and 70,6 ± 3,1 years of age. The patient group consisted of 17 females, and 10 males, while the control group comprised of 7 women, and 9 men. Comparative demographic data in the patient, and the control groups were summarized in Table 1. A significant difference was not detected between the patient, and the control groups as for gender, and ages of the study participants.
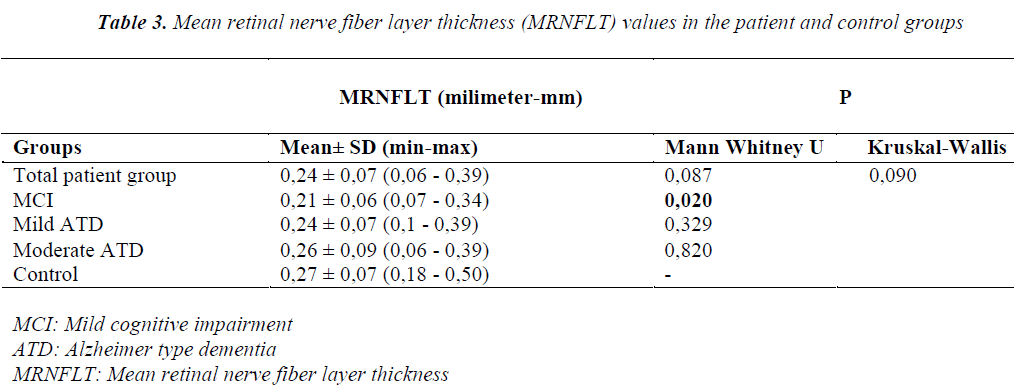
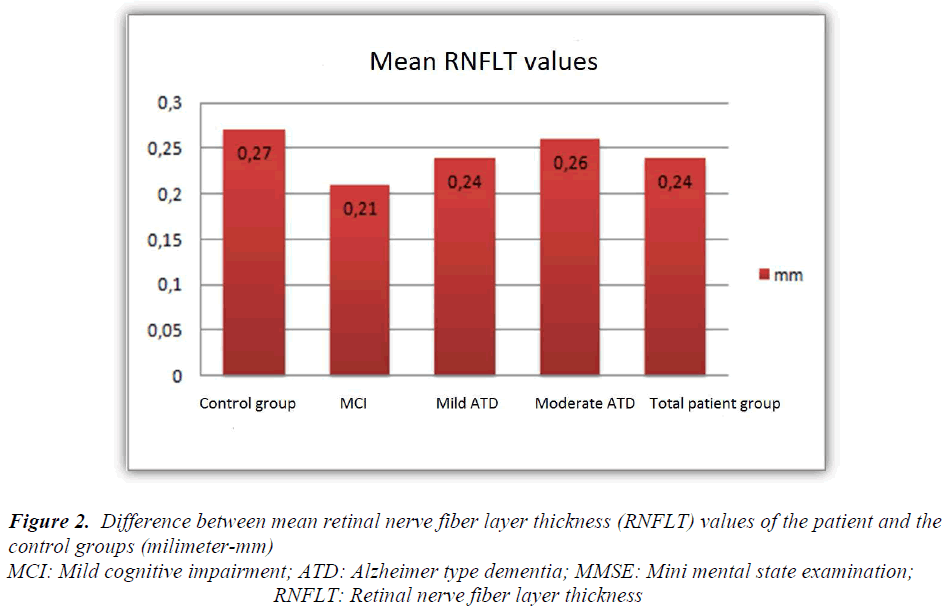
Difference between mean RNFLT values of the MCI, and the control groups was more prominent than that observed in comparisons between mild, and moderate ATD, and the control groups (Figure 2). Table 3 contains p values obtained as a result of comparisons between the control, and the patient groups. When all groups were evaluated in combination, a statistically significant difference between RNFLT values was not detected. Regarding RNFLT, in intergroup comparisons, a statistically significant difference was not observed between ATD groups combined (mild, and moderate), and the control group. (p>0.05). However mean RNFLT value of the MCI group was found to be statistically significantly lower than that of the control group (p=0.02).
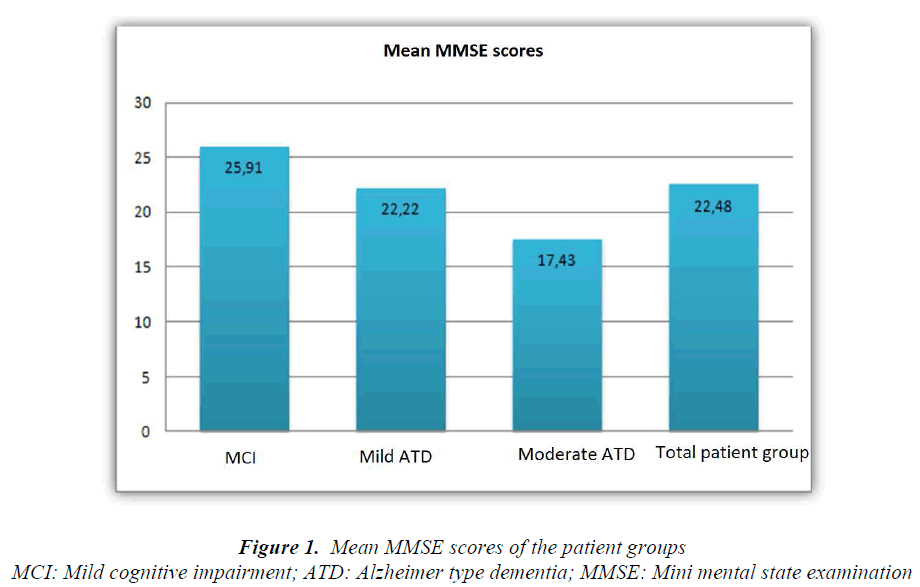
P values estimated as a result of comparisons between RNFLT values are shown in Table 4. Values displayed in the Table indicate absence of any statistically significant difference between patient groups as for RNFLT values (p>0.05). 384x384 pixels, saved them on the hard drive, and computed a single topographic image from this series. After the scanning procedure, the same physician drew the contour line of the optic disc margin for all cases by marking an average of 5-6 points on the external edge of the disc border, and optic disc topographic measurements Demographic data of the groups with MCI, mild, and moderate ATD were summarized in Table 2. Any statistically significant difference was not found between patient groups as for gender, and ages of the participants (p>0.05). Mean MMSE scores of the patient groups are shown in Figure 1.
Discussion
Several studies have attributed visual decline to the degeneration of primary visual cortex, and higher cortical levels [3,9,10]. Still, some studies have indicated loss of retinal ganglion cells, and axons as an evidence of precortical involvement. However, special, up-to-date imaging techniques (optical coherence tomography-OCT, laser polarimetry, pattern electroretinogram-PERG, HRT) have also sustained contradictory opinions.
Iseri et al. measured RNFLT using OCT in cases with ATD, and detected significant decreases in RNFLT relative to the control group. They also revealed a correlation between the decrease in macular volume, and degree of the cognitive decline [11].
Characteristic findings of cortical degeneration in Alzheimer’s disease are especially seen in the areas of visual association. Histopathological studies have demonstrated the presence of pathological markers (neurofibrillary tangles, senile plaques, beta amyloid, tau, amyloid precursor hemoglobin –APP) in subcortical visual centers (lateral geniculate nucleus, and superior colliculus) [3]. These alterations have not been observed in retina. On the other hand decreases in the number of retinal ganglion cells have been noted.
Morphological studies performed on the optic nerve have predominantly demonstrated loss of retinal ganglion cells [12]. This phenomenon might be the result of cortical retrograde degeneration as an integrated part of the primary process. In their postmortem histopathological studies, Blanks et al detected a 25 % neuron loss in the retinal ganglion cell layer in patients with Alzheimer’s disease [12].
Paquet et al. couldn’t find a significant difference between cases with MCI, and mild ATD regarding RNFLT values measured by optical coherence tomography, while they detected significantly lower RNFLT values in patients with MCI, and moderate ATD. Any correlation was not found between between MMSE scores, age of the patients, and RNFLT values in cases with ATD. In the control group of the study, female participants were more numerous than the male participants. However, in consideration of previous studies, the authors indicated that RNFLT values had not been influenced by the gender of the patients, and suggested RNFLT measurement as a structural marker of axon loss in the optic nerve [13].
In a study where decrease in RNFLT was demonstrated in cases with Alzheimer- type dementia using OCT, PERG revealed the presence of a correlation between this morphological abnormality, and retinal dysfunction with age-related proportional decrease in RNFLT in the control group. Estimated RNFLT values in ATD were lower than those found in the age-matched control group. In this situation, as a generally accepted corollary, decrease in RNFLT could not be attributed to advanced age [14].
Patients diagnosed as Alzheimer-type dementia have described visual symptoms as the disease deteriorates including blurred vision, inability to engage in reading for longer periods, impaired depth perception, and difficulty in identifying familiar faces, and objects. According to the study performed by Sadun et al, visual dysfunction onsets during routine visual acuity tests without any evidence of any ocular disease [9]. In a study by Mountjoy et al. ATD- related visual defects had been attributed to the cerebral atrophy which is the characteristic feature of the disease [10]. In histopathological analyses conducted by Johnson et al in cases with Alzheimer’s disease, degenerative changes were encountered in primary visual cortex [15].
Balazsi et al. underscored potential diagnostic deficits, and errors encountered in histopathological studies [16]. Number of optic nerve fibers in specimens harvested from autopsied cases can not be precisely enumerated. Besides, structural, and functional characteristics of the vision can not be assessed through examination of these specimens. Retinal tissue loses secondary to histologically defined Alzheimer’s disease might reflect histopathologic alterations occurring in more advanced stages of the disease. This finding might account for the failure to demonstrate ATD-related degeneration using in vivo techniques. Optic nerve damage detected in histopathological studies appears secondary to neuronal atrophy in cortical regions, which might reflect severe retrograde degeneration of the optic nerve fibers. In other words, the disease affects primarily visual cortex, and it also has an indirect impact on retinal ganglion cells as an outcome of retrograde degeneration. However, retrograd degeneration can be detected in the advanced stages of the disease. According to this study, visual dysfunction developed in parallel with the disease does not become evident at least in the early stages of ATD because of decrease in retinal nerve fiber layer thickness [16].
In a study conducted in 9 patients diagnosed as an earlystage ATD, RNFLT was measured using OCT, and retinal venous blood flow was evaluated. As an outcome of the study, in the patient group decrease in RNFLT, stenotic retinal veins, and decrease in venous blood pressure were demonstrated only in the superior quadrant [17].
In our study, difference between the MCI, and the control groups as for mean RNFLT values was more prominent relative to the difference between the groups with both types of ATD, and the control groups. Any statistically significant difference was not seen between the control and mild, and moderate ATD groups. However mean RNFLT value of the MCI group was found to be statistically significantly lower than that of the control group. This phenomenon suggests us lack of any correlation between the severity of cognitive decline, and loss of RNLFT. When the patient group in general was compared with the control group, though not statistically significant, mean RNFLT values tend to be lower in the former group.
In conclusion, controversy exists related to the necessity of measurements of RNFLT for the diagnosis of MCI, and early stage ATD. Relative to the other techniques examining optical nerve, HRT is a superior, simple, and easily applicable test in the examination of optical nerve head. Therefore, HRT can be useful in the diagnosis of MCI, mild, and moderate ATD. The data we retrieved, have demonstrated that RNFLT values decreased without any correlation between the increased severity of cognitive decline, and decreased RNFLT values.
References
- Hendrie HC. Epidemiology of Dementia and Alzheimer's Disease. Am J Geriatr Psychiatry 1998; 6:3-18.
- Bakar M. Hafif Kognitif Bozukluk. Demans Dergisi 2002; 2: 97-104
- Justino L, Kergoat M, Bergman H, Chertkow H, Robillard A, Kergoat H. Neuroretinal function is normal in early dementia of Alzheimer type. Neurobiology of Aging 2001; 22: 691-695.
- Mikelberg FS, Parfitt CM, Swindale NV. Ability of the Heidelberg Retina Tomography to detect early glaucomatous visual field loss. J Glaucoma 1995; 4: 242-247.
- Eker E. Alzheimer Hastaligi. Turkiye’de Sik Karsilasilan Psikiyatrik Hastaliklar Sempozyum Dizisi 2008 (Mart); 62: 85-110
- Petersen RC, Doody R, Kurz A, Mohs RC, Morris JC, Rabins PV, Ritchie K, Rossor M, Thal L, Winblad B. Current concepts in mild cognitive impairment. Arch Neurol. 2001; 58: 1985-1992.
- Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004; 256: 183-194.
- Petersen RC, Stevens JC, Ganguli M, Tangalos EG, Cummings JL, DeKosky ST. Practice parameter: early detection of dementia: mild cognitive impairment (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2001; 56: 1133-1142.
- Sadun AA, Bassi CJ. Optic nerve damage in Alzheimer’s disease. Ophthalmology 1990; 97:9–17.
- Mountjoy CQ, Roth M, Evans NJ, Evans HM. Cortical neuronal counts in normal elderly controls and demented patients. Neurobiol Aging 1983; 4: 1-11.
- Iseri P, Altinas Ö, Tokay T, Yüksel N. Relationship between cognitive impairment. Journal of Neuro- Ophtalmology 2006 Mar; 26 (1): 18-24.
- Blanks JC, Hinton DR, Sadun AA, Miller CA. Retinal ganglion cell degeneration in Alzheimer’s disease. Brain Res. 1989 Nov 6; 501 (2): 364-372.
- Paquet C, Boissonnot M, Roger F, Dighiero P, Gil R, Hugon J. Abnormal retinal thickness in patients with mild cognitive impairment and Alzheimer’s disease. Neuroscience Letters 2007 Jun 13; 420 (2): 97-99.
- Parisi V, Restuccia R, Fattapposta F, Concetta M, Bucci M. G, Pierelli F. Morphological and functional retinal impairment in Alzheimer's disease patients. Clinical Neurophysiology 2001 Oct; 112 (10): 1860-1867.
- Kergoat H, Kergoat MJ, Justino L, Chertkow H, Robillard A, Bergman H. An evaluation of the retinal nevre fiber layer thickness by scanning by laser polarimetry in individuals with dementia of Alzheimer type. Acta Ophtalmol Scand 2001; 79: 187-191
- Balazsi AG, Bootman J, Drance SM, Schulzer M, Douglas GR. The effect of age on the nerve fiber population of the human optic nerve. Am J Ophthalmol 1984; 97: 760-766.
- Berisha F, Feke G, Trempe C, McMeel JW, Schepens C L. Retinal Abnormalities in early Alzheimer’s Disease. Invest Ophthalmol Vis Sci. 2007 May; 48 (5): 2285-2289.