Research Article - Biomedical Research (2017) Volume 28, Issue 20
Relationship between platelet to lymphocyte ratio and coronary angiography timing in patients with NSTEMI
Sabri Seyis*, Seyda Gunay, Ibrahim Rencuzoglu
Istinye University Hospital Istanbul, Istanbul, Turkey
Accepted date: October 25, 2017
Abstract
Objective: To investigate the usefulness of PLR in predicting severity and complexity of coronary atherosclerosis as assessed by the SXscore in patients with Non ST elevation myocardial infarction NSTEMI who underwent nonurgent coronary angiography (CA).
Background: In NSTEMI patients, there is a debate whether early angiography and revascularization is beneficial compared with a more conservative approach. Platelet to lymphocyte ratio (PLR) is a new prognostic marker that gives idea about inflammation and aggregation pathways and may predict coronary atherosclerotic burden. PLR can be used to select patients who should go under angiography earlier due to excessive ischemic load.
Methods: We retrospectively evaluated 111 consecutive patients who presented with NSTEMI and underwent non-urgent coronary angiography between July 2016 and March 2017. The PLR was calculated as the ratio of the platelet count to the lymphocyte count. The Syntax scores SX of all patients were calculated. A low SX score was defined as ≤ 22, an intermediate score as 23 to 32, and a high score as ≥ 33.
Results: 92 patients (82.8%) had low SX scores (≤ 22), 19 patients (17.1%) had intermediate to high SX scores (≥ 23). Patients in the intermediate to high SX score group had significantly higher PLR 263 (219-366) vs. 117 (82-144.5) p<0.001. In ROC analysis PLR of 178 or lower predicted a low SX score defined as ≤ 22 with a sensitivity of 91.3% and specificity of 100%.
Conclusion: In patients with NSTEMI, PLR has significant association with syntax score and may be used for risk stratification and assessing optimal timing for coronary angiography.
Keywords
Platelet, Lymphocyte, NSTEMI, SYNTAX score.
Introduction
Coronary artery disease (CAD) is the leading cause of morbidity and mortality throughout the world [1,2]. It has a complex pathophysiology, and inflammation seems to play an important role in CAD [3]. Previous studies have shown that higher levels of inflammatory markers are associated with the severity of CAD and worse cardiovascular outcome [4]. Although endothelial damage has been known as the triggering factor for the formation of atherosclerotic plagues, inflammatory process is responsible in the initiation and progression of the atherosclerosis [3] Previous studies have demonstrated an association between high circulating platelet count and major adverse cardiovascular outcomes in patients with CAD as well as in healthy adults [5-7]. Platelet to lymphocyte ratio (PLR) is a new prognostic marker that integrates the risk prediction of these two parameters. It gives an idea about both the aggregation and inflammation pathways, and it may be more valuable than either platelet or lymphocyte count alone in the prediction of coronary atherosclerotic burden. The burden of coronary atherosclerosis is closely associated with prognosis in acute coronary syndrome (ACS) [8]. The SYNTAX score (SXscore) is an angiographic scoring system based on the severity and complexity of coronary lesions [9]. The SXscore has been shown to be able to predict mortality and morbidity at early and late follow-up in patients with ACS [10-12]. Patients with ACS may present an STsegment elevation myocardial infarction (STEMI), non STsegment elevation myocardial infarction (NSTEMI), or unstable angina. Current STEMI guidelines recommend urgent angiography followed by immediate mechanical opening of the infarct-related coronary artery to restore coronary flow (reperfusion therapy), a strategy that has been shown to reduce mortality. In NSTEMI patients, however, there has been continued debate over the last 10 years whether “immediate,” “urgent,” or “early” angiography and revascularization is more beneficial than a more “conservative” or “selective invasive” approach [13]. Since it is well known that inflammatory response is closely associated with the pathogenesis of coronary atherosclerosis, we aimed to investigated the usefulness of PLR in predicting severity and complexity of coronary atherosclerosis as assessed by the SXscore in patients with non-ST-segment ACS who underwent non-urgent coronary angiography (CA). By this way, we also investigated if PLR can be used to select patients who should go through angiography earlier due to excessive ischemic load.
Methods
The present study is a single-center and retrospectively designed study, consisting of eligible consecutive patients who were hospitalized at our institution because of NSTEMI and underwent non-urgent coronary angiography (CAG) between July 2016 and March 2017. A total of 111 age and gender matched patients were admitted for analysis. Exclusion criteria consisted of hematologic disorders, active infectious or inflammatory diseases, rheumatologic diseases, severe renal or liver disease, chronic renal disease, use of glucocorticoids, and malignancy. Since the SXscore has been used only for patients with native coronary artery lesions, patients with histories of CAD (bypass grafting and/or coronary stent history) were also excluded. NSTEMI was diagnosed when characteristic chest pain lasted ≥ 20 min with or without associated ST-segment depression ≥ 0.1 mV and/or T-wave inversion in 2 contiguous leads on the electrocardiogram or no electrocardiographic abnormalities and presence or absence of increased levels of troponin.
Hyperlipidaemia was defined as serum total cholesterol ≥ 240 mg/dL, serum triglyceride ≥ 200 mg/dL, low-density lipoprotein cholesterol ≥ 130 mg/dL, previously diagnosed hyperlipidaemia, or use of lipid-lowering medication. Hypertension was defined as a systolic blood pressure (SBP) of 140 mm Hg or higher, a diastolic blood pressure (DBP) of 90 mm Hg or higher, or taking antihypertensive medication. Diabetes mellitus was defined as a fasting plasma glucose ≥ 126 mg/dl, 2-hour plasma glucose during oral glucose tolerance test (75 g) ≥ 200 mg/dl, hemoglobin-A1c ≥ 6.5%, random plasma glucose ≥ 200 mg/dl, or the use of an antidiabetic agent. The total numbers of white blood cells (WBCs), lymphocytes, neutrophils, monocytes, platelets, platecrit, platelet distribution width, mean platelet volume, hemoglobin, hematocrit, and lipid profiles were measured for all patients. The PLR was calculated as the ratio of the platelet count to the lymphocyte count.
The SXscores of all patients were calculated by two independent experienced interventional cardiologists who were blinded to the identities and clinical information of the patients from baseline diagnostic CA. Each lesion with ≥ 50% diameter stenosis in vessels ≥ 1.5 mm in diameter was scored using version 2.1 of the on-line calculator at www.syntaxscore.com. The SXscore was defined as low if ≤ 22, intermediate if ≥ 23 and ≤ 32, and high if ≥ 33 [14]. By this definition, patients with SXscores ≥ 23 were considered to have moderate to severe CAD. Thus, the patients were divided into 2 groups, those with low SXscores (≤ 22) and those with intermediate or high SXscores (≥ 23).
All analyses were performed using R 3.3.2v program (open access). Continuous variables were defined as means and standard deviation; categorical variables were given as percentages. Hematologic parameters and the PLR, which was derived from these parameters, were checked for normal distribution using Shapiro-Wilks test. Based on the distribution pattern of variables, Independent Samples t-test (for normally distributed parameters) or Mann Whitney U test (for not normally distributed parameters) was used. Categorical variables were compared by the chi-square test. Pearson’s correlations were used to analyze the correlation between PLR and SXscore. Receiver-operating characteristic analyses were used to detect the cut-off value of PLR in the prediction of intermediate to high SXscore in order to select patients to be prioritized for coronary angiography; p values <0.05 were considered significant. The study protocol was approved by the local ethics committee.
Results
A total of 111 patients with non-ST-segment ACS who underwent non-urgent CA were enrolled in the study. The mean age was 57.7 ± 9.21 years; 28 of the patients (25.2%) were women. The sample was divided into two groups based on the results of CA and syntax scores. While 92 patients (82.8%) had low SXscores (≤ 22), 19 patients (17.1%) had intermediate to high SXscores (≥ 23). The differences in the baseline clinical characteristics of the patients with low and intermediate to high SXscores are outlined in Table 1. The groups were similar in age and sex. The presence of coronary risk factors such as hypertension, diabetes mellitus, and hyperlipidemia was also similar. Left ventricular ejection fraction (LVEF) values were significantly lower in patients with intermediate to high SXscores (≥ 23).
| SYNTAX Score | ||||
|---|---|---|---|---|
| = 22 (n=92) | = 23 (n=19) | p | ||
| Age | 57.58 ± 9.23 | 58.42 ± 9.17 | 0.717 | |
| Gender | Female | 23 (25) | 5 (26.32) | 0.904 |
| Male | 69 (75) | 14 (73.68) | ||
| Ejection Fraction | 44.73 ± 8.5 | 39.74 ± 8.07 | 0.021* | |
| Diabetes Mellitus | No | 47 (51.09) | 10 (52.63) | 0.999 |
| Yes | 45 (48.91) | 9 (47.37) | ||
| Hypertension | No | 37 (40.22) | 7 (36.84) | 0.999 |
| Yes | 55 (59.78) | 12 (63.16) | ||
| Hyperlipidemia | No | 54 (58.7) | 8 (42.11) | 0.212 |
| Yes | 38 (41.3) | 11 (57.89) | ||
Table 1: Baseline clinical and angiographic characteristics of the subjects.
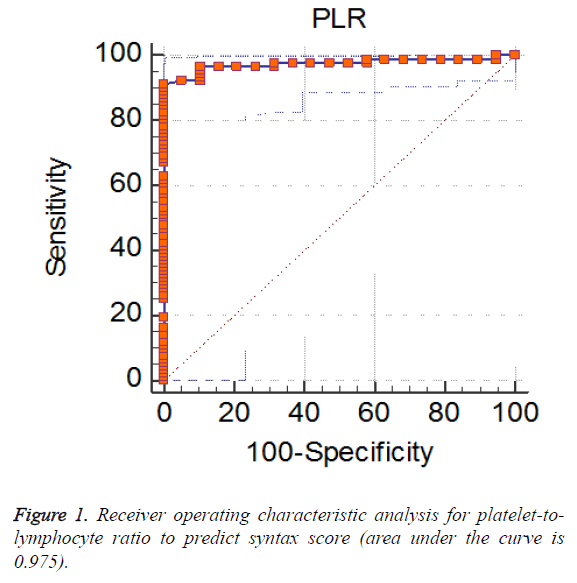
Baseline laboratory characteristics of the patients are given in Table 2. According to the admission laboratory results, patients in the intermediate to high SXscore group had significantly higher platelet to lymphocyte ratio (PLR), neutrophil to lymphocyte ratio (NLR) and neutrophil count. On the other hand, while platelet count was similar, lymphocyte and monocyte counts were significantly lower in the intermediate to high SXscore group. Receiver-operating characteristic analyses were used to detect the cut-off value of PLR (Tables 3). A PLR value of 178 or lower predicted a low SXscore defined as ≤ 22 with a sensitivity of 91.3% and specificity of 100% (Figure 1).
| SYNTAX Score | |||
|---|---|---|---|
| = 22 (n=92) | = 23 (n=19) | p | |
| Platelet | 253.08 ± 64.06 | 276.11 ± 106.08 | 0.372 |
| Platecrit | 0.26 ± 0.07 | 0.28 ± 0.1 | 0.243 |
| Platelet distribution width (PDW) | 13.8 (12.5-15.45) | 13.6 (11.8-15.7) | 0.925 |
| Mean platelet volume (MPV) | 10.67 ± 0.94 | 10.64 ± 0.91 | 0.897 |
| Hemoglobin-b | 13.16 ± 1.76 | 12.93 ± 1.51 | 0.595 |
| Hematocrit | 39.5 (36.1-42.8) | 40.5 (36.7-42.2) | 0.882 |
| WBC | 10764.13 ± 2980.84 | 10978.95 ± 3465.48 | 0.783 |
| Lymphocyte | 2.34 ± 0.84 | 1 ± 0.39 | p<0.001* |
| Neutrophil | 7.21 ± 3 | 9.43 ± 3.48 | 0.005* |
| Monocyte | 0.79 ± 0.27 | 0.5 ± 0.31 | p<0.001* |
| High density lipoprotein (HDL) | 38.24 ± 9.17 | 40.84 ± 11.08 | 0.280 |
| Low density lipoprotein (LDL) | 120.47 ± 35.37 | 134 ± 38.63 | 0.138 |
| Triglyceride | 131.5 (97.5-205.5) | 99 (77-191) | 0.062 |
| Platelet to Lymphocyte Ratio (PLR) | 117 (82-144.5) | 263 (219-366) | p<0.001* |
| Neutrophil to Lymphocyte Ratio (NLR) | 2.84 (2.1-4.495) | 9.63 (7.04-13.2) | p<0.001* |
Table 2: Baseline laboratory characteristics of the subjects.
| AUC (area under the ROC curve) | 0.975 |
| Standard error | 0.0135 |
| 95% Confidence interval | 0.926 to 0.995 |
| Z statistic | 35.272 |
| Significance level P (Area=0.5) | <0.0001 |
| Youden index | 0.913 |
| Criterion | = 178 |
| Sensitivity | 91.3 |
| Specificity | 100 |
Table 3: The receiver operating characteristic (ROC) curve analysis of platelet-to-lymphocyte ratio to predict syntax score.
Discussion
In the present study, we have shown that PLR at admission is an independent predictor of the prevalence of more complex coronary artery lesions (SXscore ≥ 23) in patients with NSTEMI. In addition, the study has shown that a cut-off value for PLR can be detected to predict the severity of CAD in patients with NSTEMI while they have not yet been evaluated with coronary angiography. Platelets and inflammation play an important role in the pathophysiology of CAD. The role of inflammation in CAD has been studied extensively, and a consistent relationship between various inflammatory markers and CVD has been established in the past [15-17]. In cases of sustained inflammation, lymphocyte counts decrease due to increased lymphocyte apoptosis. Also, ongoing inflammatory conditions lead to increased proliferation in megakaryocytic series and relative thrombocytosis.
Lymphocytopenia is a common finding in chronic inflammatory states because of increased lymphocyte apoptosis. Moreover, the leukocyte production in bone marrow makes a shift towards increasing neutrophils and decreasing lymphocytes in response to stress. Lymphocytes represent a more convenient immune response, while neutrophils cause a destructive inflammatory reaction [18]. The diagnostic and prognostic usefulness of a low lymphocyte count was demonstrated in patients with ACS and stable CAD, respectively [19,20]. The circulating platelets may contribute to the initiation of atheromatous plaque formation and trigger its complications [7]. High platelet and low lymphocyte counts in circulation have been suggested to be risk indicators of worse cardiovascular outcomes in previous studies [5-7,18-21]. A recently developed new prognostic marker, high PLR, integrates the predictive risk of these two parameters together. The advantage of PLR calculation could be that it reflects the condition of both aggregation and inflammatory pathways, and it may be more valuable than either platelet or lymphocyte count alone in the prediction of coronary atherosclerotic burden.
In this study, we also found a significant association between platelet lymphocyte ratio (PLR) and The SYNTAX score in patients with NSTEMI. The mechanisms of the relation between PLR and SXscore are not clear. One of the possible mechanisms may be an increased inflammatory response. Balta et al. demonstrated that PLR may be a useful inflammatory marker in clinical practice [22]. Platelets, as an acute phase reactant, may also increase in number in response to various stimuli (systemic infection, inflammatory conditions, bleeding, and tumors), resulting in overproduction of proinflammatory cytokines [23,24]. Higher platelet counts may reflect underlying inflammation, as several inflammatory mediators stimulate megakaryocytic proliferation and produce relative thrombocytosis. Another likely mechanism may be the increased physiologic stress induced by ACS. It has been proposed that in response to physiologic stress during myocardial ischemia or infarction, there is release of cortisol and catecholamine, redistribution of lymphocytes to lymphatic organs, and apoptosis, which lead to lymphopenia [25]. High level of physiologic stress means high levels of cortisol and catecholamine, which may translate into a lower lymphocyte count.
In patients with NSTEMI, optimal time for coronary angiography is not defined as clearly as in STEMI except in some circumstances. Current NSTEMI guidelines recommend the assessment of ischemic risk and bleeding risk using validated risk scores in each individual patient to decide on pharmacological and invasive management [26,27]. The guidelines recommend urgent or immediate transfer to the catheterization laboratory for patients with ongoing signs and symptoms of ischemia and for patients with hemodynamic or electric instability. An early invasive management in high-risk patients with non-ST-segment elevation acute coronary syndrome (NSTE-ACS) is recommended; high-risk features include the evidence of myocardial necrosis resulting in troponin elevations, ongoing myocardial ischemia with dynamic ST-segment changes on the electrocardiogram, the presence of diabetes mellitus, and a history of a recent percutaneous coronary intervention (PCI) or coronary artery bypass graft (CABG). Meta-analyses of the randomized control trials comparing an early invasive management strategy with a more conservative or selective invasive management have shown that an early invasive management reduced the composite of death or MI, in particular in patients with high ischemic risk scores [28,29]. In summary, early intervention is recommended in high-risk ACS patients and implemented in most centers.
According to our findings, higher PLR values on admission were significantly associated with intermediate to high SXscores in patients with NSTEMI. To our knowledge, this is the first study to have investigated the role of PLR as a predictor measure of CAD severity and ischemic load to give priority to patients in terms of coronary angiography timing in patients with NSTEMI although Kurtul et al. reported in a previous study that PLR at admission is significantly associated with the severity and complexity of coronary atherosclerosis in a heterogeneous patient population consisting both STEMI and NSTEMI [30].
Study Limitations
This study has a few limitations. Firstly, platelet functions were not measured and also PLR was based on a single measurement. It would be interesting to know if the PLR changes over time and if the PLR on subsequent tests remains a predictor of the severity of coronary disease. Second, we used a retrospective design for this study, which is not the best design to follow up the effect of coronary angiography timing in terms of both morbidity and mortality. A longer period of follow-up may be necessary to assess the accuracy of deciding the coronary angiography time based on PLR at admission. Finally, all patients were from a single center and the sample size was not large. Despite the limitations of the study, we observed that individuals with higher PLR are significantly more likely to have higher SYNTAX scores. Therefore, further large-scale, prospective studies are needed to gain more information about predicting CAD severity before performing coronary angiography in patients with NSTEMI.
Conclusion
In patients with NSTEMI, PLR has significant association with the SYNTAX score. Future research needs to investigate the relationship with longitudinal data to establish a decision in terms of timing of coronary angiography in patients undergoing non-urgent CAG. PLR is easy to access and inexpensive, so before performing coronary angiography, it appears to be additive to conventional risk factors and commonly used biomarkers for risk stratification and assessing optimal timing for coronary angiography. It can help to identify individuals at high risk for advanced CAD who might need an earlier therapeutic approach and closer clinical follow-up.
References
- Lopez AD, Mathers CD, Ezzati M. Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data. Lancet 2006; 367: 1747-1757.
- Onat A, Yazici M, Sari I. The risk factor survey of 2003 in western turkey indicates trend to declining coronary mortality and urban overall mortality. Arch Turk Soc Cardiol 2003; 31: 762-769.
- Libby P, Ridker PM, Hansson GK. Inflammation in atherosclerosis: from pathophysiology to practice. J Am Coll Cardiol 2009; 54: 2129-2138.
- Sönmez O, Ertaş G, Bacaksız A. Relation of neutrophil- to-lymphocyte ratio with the presence and complexiety of coronary artery disease: anobservational study. Anadolu Kardiyol Derg 2013; 13: 662-667.
- Nikolsky E, Grines CL, Cox DA, Garcia E, Tcheng JE, Sadeghi M. Impact of baseline platelet count in patients undergoing primary percutaneous coronary intervention in acute myocardial infarction (from the CADILLAC trial). Am J Cardiol 2007; 99: 1055-1061.
- Iijima R, Ndrepepa G, Mehilli J, Bruskina O, Schulz S, Schomig A. Relationship between platelet count and 30-day clinical outcomes after percutaneous coronary interventions. Pooled analysis of four ISAR trials. Thromb Haemost 2007; 98: 852-857.
- Thaulow E, Erikssen J, Sandvik L, Stormorken H, Cohn PF. Blood platelet count and function are related to total and cardiovascular death in apparently healthy men. Circulation 1991; 84: 613-617.
- Huang G, Zhao JL, Du H, Lan XB, Yin YH. Coronary score adds prognostic information for patients with acute coronary syndrome. Circ J 2010; 74: 490-495.
- Sianos G, Morel MA, Kappetein AP, Morice MC, Colombo A, Dawkins K, van den Brand M, Van Dyck N, Russell ME, Mohr FW, Serruys PW. The SYNTAX score: an angiographic tool grading the complexity of coronary artery disease. Eurointervention 2005; 1: 219-227.
- Valgimigli M, Serruys PW, Tsuchida K, Vaina S, Morel MA, van den Brand MJ, Colombo A, Morice MC, Dawkins K, de Bruyne B, Kornowski R, de Servi S, Guagliumi G, Jukema JW, Mohr FW, Kappetein AP, Wittebols K, Stoll HP, Boersma E, Parrinello G; ARTS II. Cyphering the complexity of coronary artery disease using the syntax score to predict clinical outcome in patients with three-vessel lumen obstruction undergoing percutaneous coronary intervention. Am J Cardiol 2007; 99: 1072e-1081.
- Capodanno D, Di Salvo ME, Cincotta G, Miano M, Tamburino C, Tamburino C. Usefulness of the SYNTAX score for predicting clinical outcome after percutaneous coronary intervention of unprotected left main coronary artery disease. Circ Cardiovasc Interv 2009; 2: 302-308.
- Magro M, Nauta S, Simsek C, Onuma Y, Garg S, van der Heide E, van der Giessen WJ, Boersma E, van Domburg RT, van Geuns RJ, Serruys PW. Value of the SYNTAX score in patients treated by primary percutaneous coronary intervention for acute ST-elevation myocardial infarction: the MI SYNTAX Score study. Am Heart J 2011; 161: 771-781.
- de Winter RJ, Tijssen JGP. Non-ST-segment elevation myocardial infarction revascularization for everyone? JACC: Cardiovascul Interven 2012.
- Yang CH, Hsieh MJ, Chen CC, Chang SH, Wang CY, Lee CH, Hsieh IC. SYNTAX score: an independent predictor of long-term cardiac mortality in patients with acute ST elevation myocardial infarction. Coron Artery Dis 2012; 23: 445e-449.
- Han YC, Yang TH, Kim DI, Jin HY, Chung SR, Seo JS. Neutrophil to lymphocyte ratio predicts long-term clinical outcomes in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Korean Circ J 2013; 43: 93-99.
- Tamhane UU, Aneja S, Montgomery D, Rogers EK, Eagle KA, Gurm HS. Association between admission neutrophil to lymphocyte ratio and outcomes in patients with acute coronary syndrome. Am J Cardiol 2008; 102: 653-657.
- Duffy BK, Gurm HS, Rajagopal V, Gupta R, Ellis SG, Bhatt DL. Usefulness of an elevated neutrophil to lymphocyte ratio in predicting long-term mortality after percutaneous coronary intervention. Am J Cardiol 2006; 97: 993-996.
- Zouridakis EG, Garcia-Moll X, Kaski JC. Usefulness of the blood lymphocyte count in predicting recurrent instability and death in patients with unstable angina pectoris. Am J Cardiol 2000; 86: 449-451.
- Ommen SR, Gibbons RJ, Hodge DO, Thomson SP. Usefulness of the lymphocyte concentration as a prognostic marker in coronary artery disease. Am J Cardiol 1997; 79: 812-814.
- Thomson SP, Gibbons RJ, Smars PA, Suman VJ, Pierre RV, Santrach PJ. Incremental value of the leukocyte differential and the rapid creatine kinase-MB isoenzyme for the early diagnosis of myocardial infarction. Ann Intern Med 1995; 122: 335-341.
- Ommen SR, Hodge DO, Rodeheffer RJ, McGregor CG, Thomson SP, Gibbons RJ. Predictive power of the relative lymphocyte concentration in patients with advanced heart failure. Circulation 1998; 97: 19-22.
- Balta S, Demırkol S, Kucuk U. The platelet lymphocyte ratio may be useful inflammatory indicator in clinical practice. Hemodial Int 2013; 17: 668-669.
- Waehre T, Damas JK, Yndestad A, Taskén K, Pedersen TM, Smith C, Halvorsen B, Frøland SS, Solum NO, Aukrust P. Effect of activated platelets on expression of cytokines in peripheral blood mononuclear cells-potential role of prostaglandin E2. Thromb Haemost 2004; 92: 1358-1367.
- Damas JK, Waehre T, Yndestad A, Otterdal K, Hognestad A, Solum NO, Gullestad L, Frøland SS, Aukrust P. Interleukin-7-mediated inflammation in unstable angina: possible role of chemokines and platelets. Circulation 2003; 107: 2670-2676.
- Thomson SP, McMahon LJ, Nugent CA. Endogenous cortisol: a regulator of the number of lymphocytes in peripheral blood. Clin Immunol Immunopathol 1980; 17: 506-514.
- Hamm CW, Bassand JP, Agewall S. ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: the Task Force for the management of acute coronary syndromes (ACS) in patients presenting without persistent ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J 2011; 32: 2999-3054.
- Wright RS, Anderson JL, Adams CD. 2011 ACCF/AHA focused update of the guidelines for the management of patients with unstable angina/non-ST-elevation myocardial infarction (updating the 2007 guideline): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2011; 57: 1920-1959.
- Mehta SR, Cannon CP, Fox KA. Routine vs selective invasive strategies in patients with acute coronary syndromes: a collaborative meta-analysis of randomized trials. JAMA 2005; 293: 2908-2917.
- Bavry AA, Kumbhani DJ, Rassi AN, Bhatt DL, Askari AT. Benefit of early invasive therapy in acute coronary syndromes: a meta-analysis of contemporary randomized clinical trials. J Am Coll Cardiol 2006; 48: 1319-1325.
- Kurtul A, Murat SN, Yarlioglues M. Association of platelet-to-lymphocyte ratio with severity and complexity of coronary artery disease in patients with acute coronary syndromes. Am J Cardiol 2014; 114: 972-978.