Case Report - Biomedical Research (2017) Volume 28, Issue 9
Malignant peritoneal mesothelioma with chronic hepatitis B cirrhosis
Hongwei Zhao1*, Na Liu1, Dingxin Wang1, Yuehong Yue2, Bingjie Li1, Xiaomeng Lang3, Lili He4, Qilu Hu5, Ziqian Dun6, Yeshan Zhu7, Jing Zhang8 and Jianping Liu3*1Department of Gastroenterology, Hebei General Hospital, No. 348, Heping West Road, Shijiazhuang, China
2Department of Neurology, Hebei General Hospital, No. 348, Heping West Road, Shijiazhuang, China
3Department of Spleen and Stomach, Traditional Chinese Medicine Hospital of Hebei Province, No.389, Zhongshan Road, Shijiazhuang, China
4Department of Geriatrics, Hebei General Hospital, No. 348, Heping West Road, Shijiazhuang, China
5Department of Oncology, Hebei General Hospital, No. 348, Heping West Road, Shijiazhuang, China
6Comprehensive Department, Shijiazhuang Fifth Hospital, No. 42, South Road Shijiazhuang, China
7Department of Spleen and Stomach, Chinese Medicine Hospital of Tangshan, Tangshan, China
8Department of Imaging, Hebei General Hospital, No. 348, Heping West Road, Shijiazhuang, China
- *Corresponding Author:
- HW Zhao
Department of Gastroenterology
Hebei General Hospital, China
- Liu JP
Department of Spleen and Stomach
Traditional Chinese Medicine Hospital of Hebei Province, China
Accepted on January 3, 2017
Abstract
Here, we report an extremely rare case of Malignant Peritoneal Mesothelioma (MPM) with chronic hepatitis B cirrhosis. Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) in a Middle-aged 58-year-old man showed peritoneal effusion, a smaller size liver, the abdominal cavity with obviously abnormal signal and unclear border around two huge mixed density shadows. Color Doppler ultrasound examination showed a hypoechoic mass located below the left kidney and right left kidney, respectively. Histopathological examination showed that the tumor was composed of spindle cells with large quantities collagen fibers. Whereas, Epithelial Membrane Antigen (EMA), Calretinim, AE1/AE33 showed negative results by immunohistochemical examination. A fibrous mesothelioma was strongly suspected. The patient refused surgery and did not receive chemotherapy. Currently, the patient has been taking Gleevec and has a relatively stable condition.
Keywords
Malignant peritoneal mesothelioma, Cirrhosis, Abdomen.
Introduction
Peritoneal mesothelioma is a relatively rare tumor but highly aggressive malignancy originated in the serosal linings of the peritoneal cavity [1]. Most reported cases of malignant peritoneal mesothelioma are diffuse type [2], and 33% of patients diagnosed with diffuse malignant peritoneal mesothelioma have a history of asbestos exposure [3]. The disease mostly occurs in male and shows a trend of younger [4]. We report an extremely rare case of malignant peritoneal mesothelioma with chronic hepatitis B cirrhosis in a 58-yearold male patient.
Case Presentation
A 58-year-old patient who had no history of asbestos exposure was admitted to our hospital with lower extremity oedema more than a month. The patient who had been diagnosed as chronic hepatitis 10 years prior to admission has been taking oral medicine ‘lamivudine’ and ‘adefovir dipivoxil’ for antiretroviral therapy. Four years ago, the patient received splenectomy due to hypersplenism.
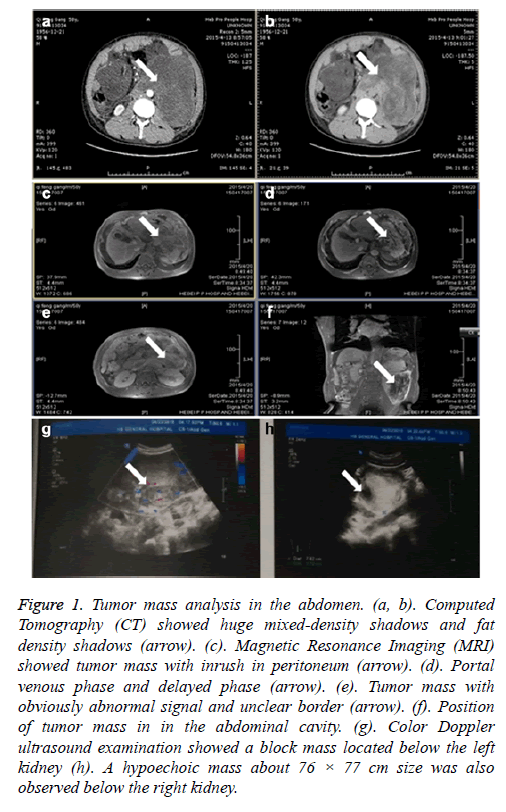
On admission, physical examination revealed Child-Pugh score 9, blood pressure 140/80 mmHg, consciousness, a little slow reaction, acceptable discrimination, 10 cm scar on the left side abdomen. No murmur, no abdominal varicose veins, no tenderness, no rebound tenderness and no muscle tension and no palpable mass were found. The levels of RBC (Red blood cell count), haemoglobin, albumin, lactate dehydrogenase, total bilirubin, adenosine deaminase, aspartate aminotransferase, hydroxybutyrate dehydrogenase, cholinesterase, total bile acid, glucose and ESR (erythrocyte sedimentation rate) were 3.47 × 1012/L, 117.00 g/L, 27.2 g/L, 294 IU/L, and 30 U/L, 34.6 umol/L, 19.00 U/L, 43 IU/L, 238 IU/L, 2496.0 U/L, 162.5 umol/L, 5.9 mmol/L, 2 m /h, respectively. CT scans showed cirrhosis, ascites, closure of the mesenteric gap, low-density liver lesions, left pleural effusion, oesophageal wall thickening. Moreover, two huge mixed-density shadows and many fat density shadows were found in the abdominal cavity with the left border unclear (Figures 1a and 1b). No lesions were observed in spleen. Similar to the CT findings, MRI showed intra-abdominal fluid, liver with a smaller size, left kidney with larger size, the abdominal cavity with obviously abnormal signal and unclear border around two huge mixed density shadows, in which portal venous phase and delayed phase were heterogeneously contrast-enhanced (Figures 1c-1f). Color Doppler ultrasound examination showed a block mass located below the left kidney which is about 163 × 115 mm size with irregular shape and blur margin. A hypoechoic mass about 76 × 77 cm size was also observed below the right kidney, in which short rod-like and star-shaped blood flow signals showed (Figures 1g and 1h).
Figure 1: Tumor mass analysis in the abdomen. (a, b). Computed Tomography (CT) showed huge mixed-density shadows and fat density shadows (arrow). (c). Magnetic Resonance Imaging (MRI) showed tumor mass with inrush in peritoneum (arrow). (d). Portal venous phase and delayed phase (arrow). (e). Tumor mass with obviously abnormal signal and unclear border (arrow). (f). Position of tumor mass in in the abdominal cavity. (g). Color Doppler ultrasound examination showed a block mass located below the left kidney (h). A hypoechoic mass about 76 × 77 cm size was also observed below the right kidney.
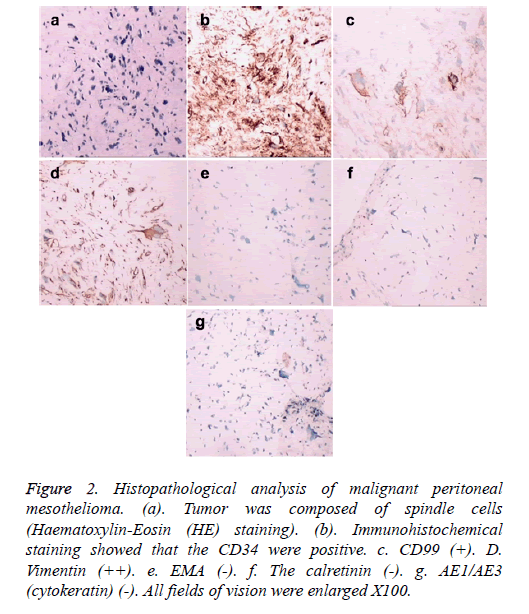
A sarcomatoid type fibrous mesothelioma was strongly suspected, and ultrasound biopsy was carried out. The surgical findings showed nodular or patchy tumor tissue located parietal or visceral peritoneum with hard texture, clear margin, and partial haemorrhage and necrosis. Histopathological analysis by Haematoxylin-Eosin (HE) staining showed that the tumor was composed of spindle cells with collagen fibers (Figures 2a and 2b). Immunohistochemical staining showed that vimentin (++), CD34 (++), CD99 (+), S100 (-), myoglobin (-), α-SMA (-), CD68 (-), the calretinin (-), AE1/AE3 (cytokeratin) (-), and Ki-67 antibody labelling index was high (Figures 2c-2j). To date, peritoneal mesothelioma was mostly diagnosed by immunohistochemistry, which exhibited different expression of such antibodies as AE1/AE3, EMA, CK, vimentin and no highly sensitive and specific antibodies. The patient refused surgery and chemotherapy. Currently, the patient has been taking oral anticancer drug ‘Gleevec’ and has a relatively stable condition.
Figure 2: Histopathological analysis of malignant peritoneal mesothelioma. (a). Tumor was composed of spindle cells (Haematoxylin-Eosin (HE) staining). (b). Immunohistochemical staining showed that the CD34 were positive. c. CD99 (+). D. Vimentin (++). e. EMA (-). f. The calretinin (-). g. AE1/AE3 (cytokeratin) (-). All fields of vision were enlarged X100.
Discussion
As a rare type of malignant mesothelioma, malignant peritoneal mesothelioma accounts for approximately 5 to 10% in all cases of malignant mesothelioma [5]. Malignant pleural mesothelioma accounts for 80 to 90% of all malignant mesothelioma cases, which has 0.2% in all malignant tumors [5,6]. Besides the pleura and peritoneum, malignant mesothelioma may involve in a small percentage of cases, the tunica vaginalis, testis, or pericardium [7]. Histologically, malignant mesotheliomas are classified as the epithelioid, biphasic, or sarcomatoid type [8]. And the sarcomatoid type is the rarest and has a poor prognosis. Macroscopically, malignant mesotheliomas are classified as diffuse and localized malignant mesotheliomas. Diffuse malignant mesotheliomas account for 85% of such cases, and localized cases are very rare [9]. Mostly, liver cirrhosis, secondary peritoneal carcinomatosis, and tuberculous peritonitis can cause ascites. Diffuse malignant peritoneal mesothelioma has been reported a rare cause of ascites [10]. This article describes a 58-year-old man inflicted with a sarcomatoid type fibrous malignant peritoneal mesothelioma and chronic hepatitis B cirrhosis. Less study has been reported.
In recent 20 years, peritoneal mesotheliomas have an increasing tendency. One of the reasons may be the rapid development of asbestos-related industries [11]. Accordingly to previous studies, asbestos exposure is the etiology of mesotheliomas and 20 to 50% of patients have peritoneal mesotheliomas in 2005 [8,12]. Some study also showed that the disease correlated with penetrating trauma and direct transmission in the peritoneal cavity of fiber vaccination [13]. Moreover, a history of radiation therapy, thorium dioxide exposure or fluorspar, chronic serositis, viral infections, and genetic predisposition may contribute to the disease development. In the present case report, patients with chronic liver cirrhosis had no direct or indirect contact with a history of asbestos and the disease may be caused by constantly stimulus from peritoneal effusion, ascites and chronic inflammation.
Due to lack of specific subjective symptoms, malignant peritoneal mesothelioma is easily misdiagnosed. Diagnosis can be achieved by tumor resection, ascitic fluid cytology, MRI, CT scan, PET and laparoscopy. Cytological examination of ascitic fluid removed by paracentesis rarely results in a positive finding and the sensitivity of ascitic fluid cytology was 54% [10]. Imaging positioning, for example, CT scan is helpful in the diagnosis of peritoneal mesothelioma, but still difficult for the diagnosis of early peritoneal lesions. The definite diagnosis of malignant peritoneal mesothelioma often depends on laparoscopy or open surgery with biopsy, as in the case described in our report. The gross appearance of mesothelioma is characterized by soft, greyish-white, papillary nodules or masses extending over peritoneal surfaces, which is consistent with previous study [10]. In pathology, malignant mesothelioma is characterized by staining for vimentin, CD34, CD99, S100, myoglobin, α-SMA, CD68, the calretinin, AE1/AE3 (cytokeratin) and Ki-67. Our findings illustrated that the calretinin and AE1/AE3 (cytokeratin) showed a negative result, respectively, which is inconsistent with previous study [8].
In addition, it is essential to explore a normative treatment for malignant peritoneal mesothelioma. Most cases are treated with surgical resection, postoperative radiotherapy or chemotherapy comprehensive treatment, especially the chemotherapy [14]. The prognosis of malignant peritoneal mesothelioma is poor, with the mean survival period<1 year. In some cases, cytoreduction surgery and hyperthermic intraperitoneal chemotherapy have been reported to prolong survival considerably 50 to 60 months [15].
Malignant peritoneal mesothelioma associated with chronic hepatitis B cirrhosis is a rare disease, which is characterized by various causes, variable courses, poor prognosis, and difficult diagnosis. It is challenging of diagnosis and treatment for malignant peritoneal mesothelioma. It should be considered in patients with peritoneal mesothelioma with recurrent ascites or abdominal mass, especially when many symptoms associated with the tumor appeared with no particular incentives.
References
- Schijf LJ, Adriaensen MEAPM, de Klerk JMH, Baarslag HJ, Heggelman BGF, Schaefer-Prokop CM. Pleural and peritoneal mesothelioma: Imaging findings on CT and FDG PET/CT. Eur J Radiol Extra 2009; 69: e89-e92.
- Haber SE, Haber JM. Malignant mesothelioma: a clinical study of 238 cases. Industr Health 2011; 49: 166-172.
- Fasola G, Puglisi F, Follador A, Aita M, Di Terlizzi S, Belvedere O. Dramatic tumour response to pemetrexed single-agent in an elderly patient with malignant peritoneal mesothelioma: a case report. BMC cancer 2006; 6: 289.
- Thomas A, Chen Y, Yu T, Gill A, Prasad V. Distinctive clinical characteristics of malignant mesothelioma in young patients. Oncotarget 2015; 6: 16766.
- Gemba K, Fujimoto N, Kato K, Aoe K, Takeshima Y, Inai K. National survey of malignant mesothelioma and asbestos exposure in Japan. Cancer Sci 2012; 103: 483-490.
- Attanoos R, Gibbs A. Pathology of malignant mesothelioma. Histopathol 1997; 30: 403-418.
- Robinson BW, Lake RA. Advances in malignant mesothelioma. N Engl J Med 2005; 353: 1591-1603.
- Takehara Y, Endo S, Mori Y, Nakahara K, Takayanagi D, Shimada S. Malignant peritoneal mesothelioma with lymph node metastasis that originated in the transverse colon. W J SurgOncol 2014; 12: 1-4.
- Allen TC, Cagle PT, Churg AM, Colby TV, Gibbs AR, Hammar SP. Localized malignant mesothelioma. Am J SurgPathol 2005; 29: 866-873.
- Shih CA, Ho SP, Tsay FW, Lai KH, Hsu PI. Diffuse malignant peritoneal mesothelioma. Kaohsiung J Med Sci 2013; 29): 642-645.
- Reid A, De Klerk N, Magnani C, Ferrante D, Berry G, Musk A. Mesothelioma risk after 40 years since first exposure to asbestos: a pooled analysis. Thorax 2014;204161.
- Welch LS, Acherman YI, Haile E, Sokas RK, Sugarbaker PH. Asbestos and peritoneal mesothelioma among college-educated men.Int J OccupEnvir Health 2005;11:254-258.
- Humphrys N, Downing A, Evans L, Sinclair M. Traumatic implantation: A novel aetiology in the development of peritoneal mesothelioma. Case Rep Emerg Med 2013;2013.
- Thomas A, Chen Y, Yu T, Gill A, Prasad V. Distinctive clinical characteristics of malignant mesothelioma in young patients. Oncotarget 2015;6.
- Manzini VP, Lcafferata R. Malignant peritoneal mesothelioma: a multicenter study on 81 cases. Ann Oncol 2010;21:348-353.