- Biomedical Research (2009) Volume 20, Issue 1
Evaluation of the anti-ulcer activities of Morus alba extracts in experimen-tally-induced gastric ulcer in rats.
Mahmood Ameen Abdulla1*, Hapipah Mohd AliM2, Khaled Abdul-Aziz Ahmed1,3, Suzita Mohd Noor1, Salmah Ismail1
1Department of Molecular Medicine, Faculty of Medicine, University Malaya 50603 Kuala Lumpur, Malaysia
2Department of Chemistry, Faculty of Science, University Malaya 50603 Kuala Lumpur, Malaysia
3Faculty of Dentistry, Ibb University, Ibb, Yemen
- *Corresponding Author:
- Mahmood Ameen Abdulla
Department of Molecular Medicine, Faculty of Medicine, University of Malaya
50603 Kuala Lumpur, Malaysia
Phone: +603 7697 6604
Fax: +603 79676600
e-mail: khaaah@gmail.com
Accepted date: January 02 2009
Abstract
This study was aimed to elucidate the anti-ulcerogenic effects of Morus alba ethanolic ex-tracts on ethanol induced gastric mucosal injury in animals. Four groups of adult male Sprague Dawley rats were included in the study. 10% Tween 80 was given orally to Group 1 rats as a negative control group. Group 2 and 3 animals received alcoholic ex-tract of Morus alba orally at 250 mg/kg and 500 mg/kg doses, respectively. Omeprazole (20 mg/kg body weight) were given orally to Group 4 rats as a positive control. 60 min-utes after their pretreatment, all rats were administered with 5 ml/kg absolute ethanol orally. One hour later, all animals were sacrificed. Grossly, Group 1 animals showed sever mucosal injury of gastric mucosa. However, a statistically significant reduction of gastric mucosal damage (p<0.05) was seen in animals pretreated with either plant ex-tracts (250 mg/kg or 500 mg/kg body weight) or Omeprazole compared to negative con-trol animals. Rats pretreated with 500mg/kg plant extract or omeprazole have signifi-cantly (p<0.05) protected gastric mucosa compared to animals pretreated with 250mg/kg plant extract. Microscopically, animals (Group 1) pretreated with 10% Tween 80 and administered absolute alcohol showed severe gastric mucosal damages, submucosal edema, and leucocytes infiltration. Animals pretreated with plant extracts or omeprazole showed marked reduction of gastric mucosal damage, reduction of edema and leucocytes infiltration of the submucosal layer. These results indicate that M. alba extract exhibits significant anti-ulcerogenic activity in rats.
Keywords
Mourus alba, anti-ulcer, omeprazole, histology
Introduction
Morus alba L. (mulberry) as a non-toxic natural therapeu-tic agent, belongs to the family of Moraaceae. The whole plant of M. alba L. possesses multiple medicinal values [1]. The root bark, leaves and fruits of Morus alba have been traditionally used in Asian countries for medicinal purposes due to its anti-inflammatory [2,3], hypoglycemic [4], hypolipidemic and antioxidant roles [5,6]. Several studies have shown that Morus alba has neuroprotective, antiphlogistic, liver and kidney protective, hypotensive, diuretic, anticough, and analgesic effect [7-9]. Further-more, it shows antiviral [10] and antimicrobial effects [11].
Pharmacological studies reveal that flavonoids, phenols, cumarins and terperols are the main bioactive constituents in M.alba [1]. Recent studies have revealed that flavon-oids and phenolic acid show speeding cruor, antioxidant, radioprotective, anti-mutagenic and anticarcinogenic ef-fects and that cumarins possess antibacterial, hypotensive, calmative and spasmolytic functions [12,13]. To our knowledge, there were no reports regarding the anti-ulcerogenic effect of M. alba leaves. The present study aimed to elucidate the anti-ulcerogenic effect of M. alba leaves in experimental rats with macroscopical and histo-logical investigation.
Materials and Methods
Omeprazole, as a reference anti-ulcer drug, belongs to a class of drugs called proton pump inhibitor which block the production of acid by the stomach. The drug was sus-pended in 10% Tween 80 and administered orally to animals at a concentration of 20 mg/kg body weight (5 ml/kg body weight). The leaves of Morus alba were collected from the Animal Department of the Faculty of Agricul-ture, University Kebangsaan Malaysia and identified by comparison with specimens available at the Herbarium of Rimba Ilmu, Institute of Biological Sciences, University of Malaya. The leaves were washed with distilled water and dried in incubator at 50°C for 5-7 days. The dried leaves were ground to powder using a grinder.
Preparation of Morus alba alcoholic extract
The dried powdered leaves were extracted by maceration in ethanol (100g/1500 ml, w/v) in a conical flask for 5 days at room temperature. Afterwards, the solvent was filtered followed by distillation under reduced pressure in EYELA rotary evaporator until excess solvent was evapo-rated. The extract was suspended in 10% Tween 80 (w/v) and was administered to rats at doses of 250 mg/kg and 500 mg/kg body weight.
Experimental animals
Animals weighing 200-250 g were deprived of food for 48 h, but they were allowed free access to tap water until 2 h before the experiment. During the fasting period, the animals were placed individually in cages with wide-mesh wire bottoms to prevent coprophagy. On the day of experiment, the rats were randomly divided into 4 groups with 6 animals each.
Acute toxicity test
The acute toxic class method of the Organization for Eco-nomic Cooperation and Development (OECD) was used to determine a safe dose for the extracts [14]. Thirty six animals (18 males and 18 females) were assigned equally into 3 groups labeled as vehicle (distilled water); low (2 g/kg body weight) and high (5 g/kg body weight) doses of leaf extract preparations. The animals were fasted over-night (food but not water) prior dosing. Food was with-held for a further 3 to 4 hours after dosing. Observations were done on mortality and behavioral changes of the rats following treatment for 24 hours. The acute toxicity LD50 was calculated at the geometric mean of the dose that re-sulted in 100% lethality and that which cause no lethality at all.
Animals' treatment
Control rats (Group 1) received 10% Tween 80 (5 ml/kg) by orogastric intubations; whereas Group 2 and 3 rats received alcoholic extract of M. alba at concentrations of 250 mg/kg and 500 mg/kg suspended in 10% Tween 80 (5 ml/kg body weight of rats) by the same route, respec-tively. Group 4 animals received 20 mg/kg of Omeprazole suspended in 10% Tween 80 by orogastric intubations.
After 60 minutes, all animals were administered orally with 5 ml/kg absolute alcohol to induce gastric ulcer in all rats groups. One hour after their administration, all ani-mals were sacrificed by over-dose of diethyl ether and their stomach were rapidly removed and immersed in 10% buffered formalin solution.
Gross gastric lesions evaluation
Each stomach was opened along the greater curvature, rinsed with tap water to remove gastric contents and the mucosa was examined under dissecting microscope grossly (1.8x) with a square-grid eyepiece (big square: length X width = 10 X 10 mm2 = ulcer area) to assess the formation of ulcers area (hemorrhagic lesions). The length and width of each lesion was determined and the sum of the area of all lesions for each stomach was expressed as the ulcer area (mm2). The ulcer area (UA) was calculated as described by [15] with few modifications. The inhibi-tion percentage was calculated by the following formula of Njar et al. [16]:
(%I) = [(UAcontrol − UAtreated) ÷ UAcontrol] X 100.
Histological lesions evaluation
The gastric wall specimen were fixed in 10% buffered formalin and processed by paraffin tissue processing ma-chine. The stomach was assessed for damage by taking a 5 μm section, stained with hematoxylin and eosin.
Ethics
The experimental protocol for animal work was approved by the local ethics committee for animal experimentation in the Faculty of Medicine, University of Malaya, Ethic No. PM 28/-9/2007 MAA (R).
Statistical analysis
All values are reported as mean + S.E.M. and the statisti-cal significance of differences among groups were as-sessed using one-way analysis of variance (ANOVA). A value of P<0.05 was considered significant. Statistical computations were calculated using SPSS 11.5 for Win-dows software (SPSS Inc, Chicago, IL, USA).
Results
Acute toxicity study
Animals treated with M. alba extract (low and high dose) did not produce any major clinical signs of toxicity (respi-ratory distress, haematuria, uncoordinated muscle move-ment) during 24 hours observation period. No lethality was observed upon oral administration at the end of 24 hours of observation. It is indicated that the extract is safe and the oral lethal dose (LD50) for male and female rats is above 5 g/kg body weight of rats.
Macroscopical examination
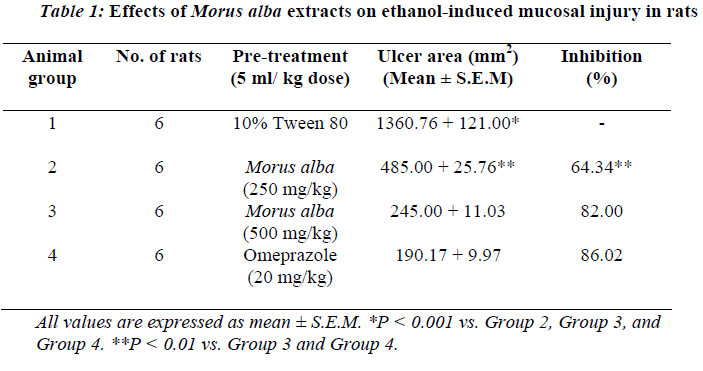
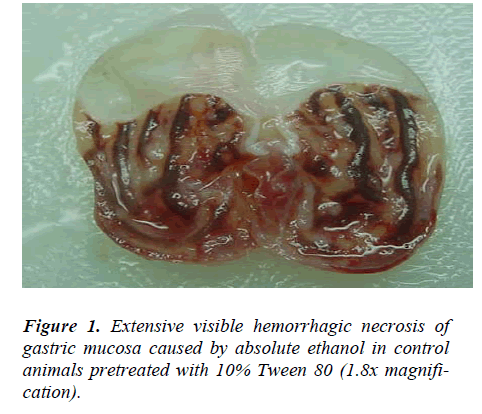
Animals pre-treated with 10% Tween 80 followed by ab-solute ethanol showed extensive macroscopic lesions (Table.1 and Fig.1). Severe hemorrhagic lineage was ob-served in variable sizes with the hemorrhagic lesions dis-tributed all over the glandular part of the stomachs indi-cating that gastric ulcer was completely formed. Pre-treatment with ethanolic extracts of M. alba or Omepra-zole significantly reduced (P<0.05) the formation of ethanol-induced gastric lesions compared to Group 2 animals (Table.1 and Fig.2).
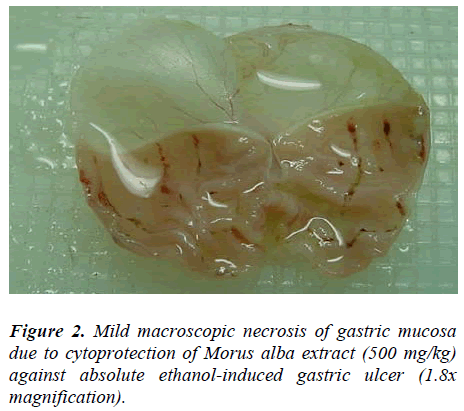
Rats pretreated with M. alba (500 mg/kg body weight) or Omeprazole showed flattening of gastric mucosal folds and significant reduction in hemorrhagic lines compared with the animals administered Morus alba extract at a dose of 250 mg/kg body weight. There were no signifi-cant differences between rats pretreated with M. alba ex-tract at a dose of 500 mg/kg body weight or rats pre-treated with Ompreazole in term of cytoprotection (Table.1).
Microscopical examination
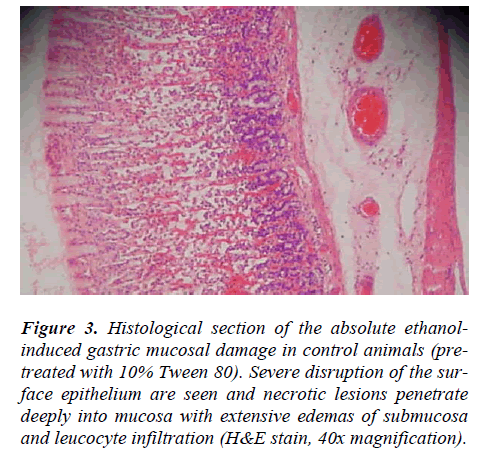
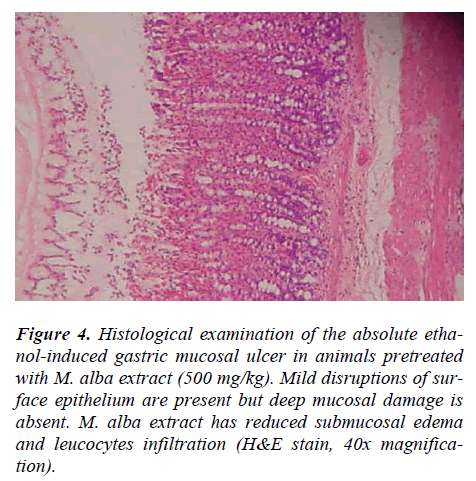
Rats pre-treated with 10% Tween 80 (Group 2) for 60 minutes followed by administration of absolute ethanol showed severe mucosal damage and submucosa was markedly thickened by edema (Fig.3). Deep hemorrhagic lesions of mucosal layer and infiltration of leucocytes were observed in the submucosal layer. As shown in Figure 4, animals pre-treated with plant extracts or Omepra-zole, showed mild mucosal damage, submucosal edema, and leucocytes infiltration.
Figure 3: Histological section of the absolute ethanol-induced gastric mucosal damage in control animals (pre-treated with 10% Tween 80). Severe disruption of the sur-face epithelium are seen and necrotic lesions penetrate deeply into mucosa with extensive edemas of submucosa and leucocyte infiltration (H&E stain, 40x magnification).
Figure 4: Histological examination of the absolute etha-nol-induced gastric mucosal ulcer in animals pretreated with M. alba extract (500 mg/kg). Mild disruptions of sur-face epithelium are present but deep mucosal damage is absent. M. alba extract has reduced submucosal edema and leucocytes infiltration (H&E stain, 40x magnifica-tion).
Discussion
The results of the present study showed that rats pre-treated with M. alba extract significantly reduced the gas-tric mucosal injury with marked reduction in the leuco-cytes infiltration of submucosal layer. Cytoprotection was assessed by reduction in the macroscopically and micro-scopically visible lesions. Similarly, Cheng and Koo [17] have showed that oral administration of plant extract be-fore ethanol administration significantly decreases neu-trophil infiltratinn of gastric mucosa. Absolute alcohol extensively damaged the gastric mucosa leading to in-creased neutrophil infiltration into the gastric mucosa. Neutrophils are a major source of inflammatory mediators and can release potent reactive oxygen species such as superoxide, hydrogen peroxide and myeloperoxidase de-rived oxidants. These reactive oxygen species are highly cytotoxic and can induce tissue damage [17]. Further-more, neutrophil accumulation in gastric mucosa has been shown to induce microcirculatory abnormalities [18]. Suppression of neutrophil infiltration during inflammation was found to enhance gastric ulcer healing [19]. The dis-turbances in gastric secretion, damage to gastric mucosa, alterations in permeability, gastric mucus depletion and free-radical production have been reported as the patho-genic mechanism of ethanol [20].
The anti-ulcer mechanisms of gastric mucosa may be due to strengthening action on gastric mucosal lining and the suppression of damaging effects of free radicals [17]. Other studies have shown the potent inhibitory effect other types of plants on inflammation and the anti-inflammatory property of these plants may be attributed to inhibition of nitric oxide synthesis leading to enhance-ment of ulcer healing [21,22]. In addition, M. alba ex-tract possesses a potential anti-inflammatory activity and may prevent organ or tissue injury during acute en-dotoxemia or sepsis by suppressing inflammatory media-tors and thus facilitates ulcer healing [2,3]. M. alba leaves contain anti-inflammatory and antioxidant properties [23]. The anti-inflammatory and antioxidant properties of tur-meric may exert antiulcer activity through scavenging reactive oxygen species [24]. The acute toxicity profile of M. alba leaf extract could be considered favorable judg-ing from the high oral LD50 value obtained and the ab-sence of adverse clinical manifestations in experimental animals after 24 hours of observation. In conclusion, the results of the present study suggest a direct protective effect of Morus alba extracts on gastric mucosal damage and that the gastroprotective action of this plant may be due to its anti-inflammateroy and antioxidant properties. However, further studies are required to explore the mechanism of action in the gastric mucosa and submu-cosa.
Acknowledgements
This study was financially supported by the University of Malaya through the Science Fund grant 12-02-03-2051. The authors are grateful to the Head of the Department of Molecular Medicine, Faculty of Medicine, University of Malaya, Malaysia.
References
- Committee of Jiangsu New Medicine College, Ency- clopedia of Traditional Chineses Medicine, Shanghai Science and Technology Press, Shanghai, 1995, p. 1963.
- Sohn HY, Son KH, Kwon CS, Kwon GS, Kang SS. Antimicrobial and cytotoxic activity of 18 prenylated flavonoids isolated from medicinal plants: Morus alba L., Morus mongolica schneider, Broussnetia papyrifera (L.) vent, Sophora flavescens ait and Echinosophora koreensis nakai. Phytomedicine 2004; 11: 666-672.
- Choi EM, Hwang JK. Effects of Morus alba leaf ex- tract on the production of nitric oxide, prostaglandin E2 and cytokines in RAW 264.7 macrophages. Fitoterapia 2005; 76: 608-613.
- Singab A, El-Beshbishy H, Yonekawa M, Nomura T, Fukai T. Hypoglycemic effect of Egyptian Morus alba extract: effect on diabetes and lipid peroxidation of streptozotocin-induced diabetic rats. J Ethnopharmacol 2005; 100: 333-338.
- El-Beshbishy H, Singab AB, Sinkkonen J, Pihlaja K. Hypolipidemic and antioxidant effects of Morus alba L. (Egyptian mulberry) root bark fractions supplementa- tion in cholesterol-fed rats. Life Science 2006; 78: 2724-2733.
- Katsube T, Imawaka N, Kawano Y, Yamazaki Y, Shi- waku K, Yamane Y. Antioxidant flavonol glycosides in mulberry (Morus alba L.) leaves isolated based on LDL antioxidant activity. Food Chem 2006; 97: 25-31.
- Fukai T, Satoh K, Nomura T, Sakagami H. Antinephri- tis and radical scavenging activity of prenylflavonoids. Fitoterapia 2003; 74: 720-724.
- Kim H, Jeong T, Lee M, Park Y, Choi M. Lipid lower- ing efficacy of hesperetin metabolites in high- cholesterol fed rats. Clin Chim Acta 2003; 237: 129-137.
- Yu D, Duan Y, Bao Y, Wei C, An L. Isoflavonoids from Astagalus monghilocus protect PC12 cells from toxicity induced by L-glutamine. J Ethnopharmacol 2005; 98: 89-94.
- Du J, He Z, Jiang R, Ye W, Xu H, But P. Antiviral fla- vonoids from the root bark of Morus alba L., Phyto- chemistry 2003; 62: 135-138.
- Fukai T, Kaitou K, Terada S. Antimicrobial activity of 2-arylbenzofurans from Morus species against methi- cillin-resistant Staphylococcus aureus. Fitoterapia 2005; 76: 708-711.
- Kosmider B, Osiecka R. Flavonoid compounds: a re- view of anticancer properties and interactions with cis- diamminedichloroplatinum (II). Drug Dev Res 2004; 63: 200-211.
- Manach C, Mazur A, Scalbert A. Polyphenols and pre- vention of cardio-. vascular diseases. Curr Opin Lipidol 2005; 16: 77–84.
- Organization for Economic Cooperation and Develop- ment (OECD) Guideline for the testing of chemicals (425). Acute oral toxicity-up-and-down procedure, 2001.
- Kauffman GL, Grossman MI. Prostaglandin and cime- tidine inhibit the formation of ulcers produced by pare- nteral salicylates. Gastroenterology 1978; 75: 1099-1102.
- Njar VCO, Adesanwo JK, Raji Y. Methyl Angolensate: The antiulcer agent of the stem bark of Etandro- phragma angolense. Planta Medica 1995; 16: 91-92.
- Cheng CL, Koo MWL. Effect of Centella asiatica on ethanol induced gastric mucosal lesions in rats. Life Sciences 2000; 67: 2647-2653.
- Bou-Abboud CF, Wayland H, Panlsen G, Guth PH. Microcirculatory stasis precedes tissue necrosis in ethanol-induced gastric mucosal injury in rat. Dig Dis Sci 1988; 33: 872-877.
- Tsukimi Y, Nozue C, Okabe S. Effects of teminopra- zole, omeprazole and sucralfate on indomethacin-indu- ced delayed healing of kissing gastric ulcers in rats. J Gastroenterol Hepatol 1996; 11: 335-340.
- Salim AS. Removing oxygen-derived free radicals stimulates healing of ethanol induced erosive gastritis in the rat. Digestion 1990; 47: 24-28.
- Cheng CL, Guo JS, Luk J, Koo MWL. The healing
- Guo JS, Cheng CL, Koo M.WL. Inhibitory effects of Centella asiatica water extract and asiaticoside on in- ducible nitric oxide synthase during gastric ulcer heal- ing in rats. Planta Medica 2004; 70: 1150-1154.
- Chung KO, Kim BY, Lee MH, Kim YR, Chung HY, Park JH, Moon JO. In vitro and in vivo anti- inflammatory effect of oxyresveratrol from Morus alba L. J Pharm Pharmacol 2003; 55: 1695-700.
- Swarnakar S, Ganguly K, Kundu P, Banerjee A, Maity P, Sharma AV. Curcumin regulates expression and ac- tivity of matrix metalloprotinases 9 and 2 during pre- vention and healing of indomethacin-induced gastric ulcer. J Biol Chem 2005; 280: 9409-9415.