- Biomedical Research (2014) Volume 25, Issue 3
Dyslipidemia and its correlation with type 2 diabetic patients at different stages of proteinuria.
Noura Al-Jamei*, Farah Aziz Khan, Sadia Arjumand, Mohammad Fareed Khan, Hajera TabassumDepartment of Clinical Laboratory Sciences, College of Applied Medical Sciences, King Saud University, Riyadh, KSA
- *Corresponding Author:
- Noura Al-Jameil
Head, Department of Clinical Laboratory Sciences
College of Applied Medical Sciences
King Saud University
P. O. 10219 Riyadh 1143,Saudi Arabia
Accepted February 06 2014
Abstract
Diabetic nephropathy is the consequence of diabetes mellitus (DM) and it is characterized by continuous albuminuria. The aim of the study was to investigate and correlate the lipid profile abnormalities in different stages of albuminuria to understand the checkpoint before progression to end stage renal disease (ESRD). A total of 154 type 2 diabetic patients were included in the study. Urine samples of all patients were subject to biochemical analysis and divided into diabetes with normoalbuminuria (n=52), diabetes with microalbuminuria (n=51), and diabetes with overt proteinuria (n=51) groups, depending upon urinary albumin creatinine ratio (ACR) of <30mg, 30-300 mg, and >300 mg/24 hours respectively. Statistical analysis was done by using One Way ANOVA and Pearson’s correlation coefficient. All lipid profile parameters, such as total cholesterol (TC), triglycerides (Tg), low density lipoprotein (LDL), very low density lipoprotein (VLDL) mean values except high density lipoprotein cholesterol (HDL-C) were increased in all the three groups i.e. diabetes with normoalbuminuria, diabetes with microalbuminuria, diabetes with overt proteinuria, were significantly increased at P<0.005 and found to be in positive correlation with ACR. Dyslipidemia concurrently with microalbuminuria should be considered as alarming signal for both cardiovascular disease (CVD) and ESRD. Therefore, detection of dyslipidemia with a corresponding microalbuminuria in the beginning of DM and accordingly therapeutic intervention could control the resulting cardiovascular or renal complications.
Keywords
Diabetes mellitus, nephropathy, albuminuria, dyslipidemia, cardiovascular disease, end stage renal disease.
Introduction
Diabetic nephropathy is the consequence of diabetes mellitus (DM) and it is characterized by continuous albuminuria, elevated blood pressure, decreased glomerular filtration rate (GFR) and high risk of CVD. The epidemiological studies have revealed that genetic susceptibility is an important factor in the development of diabetic nephropathy in patients with both type 1 and type 2 diabetes. Other contributing risk factors are glomerular hyperfiltration, smoking, dyslipidemia, levels of proteinuria, and source of protein and fat in the diet [1,2].
The incidence of DM is increasing rapidly and studies have suggested that an abnormal lipid profile in diabetes may lead to worsening the condition and direct the disease to renal impairment [3]. Glomerular and tubulointerstitial injury caused by elevated lipoproteins and lipids in DM contribute to the progression of diabetic nephropathy. However, treatment of dyslipidemia can reduce albumin excretion [4]. Abnormal lipid profile also results in cardiovascular morbidity and mortality associated with diabetic nephropathy. Early hemodynamic changes of glomerular hyperperfusion and hyperfiltration are followed by leakage of albumin from the glomerular capillaries and structural changes such as glomerular basement membrane thickening, glomerular hypertrophy, glomerulosclerosis, mesangial cell expansion, and podocyte injury [5]. Clinical manifestations of diabetic nephropathy include a decrease in the GFR and an increase in levels of urinary albumin excretion (UAE) [6].
The aim of the study is to investigate and correlate the DM-associated lipid profile abnormalities in different stages of albuminuria and to evaluate the checkpoint in controlling before the progression to ESRD.
Materials and Methods
Patients
The study was carried out at College of Applied Medical Sciences, and the subjects were recruited from King Abdul Aziz University Hospital, Riyadh. A total of 154 subjects were included in the study; 111 males and 44 fe males. An informed consent was taken from all patients and institutional ethical committee approved the study. Age of the subjects ranged between 35-80 years, and the patients were diagnosed with type 2 DM not less than 3 years. Patients having proteinuria due to other reasons like, pregnancy, hypertension, urinary tract infection, heart failure and some other chronic disorder were excluded from the study.
Sample Collection
After overnight fasting, 8 ml of venous blood samples were collected in clean glass tubes and 1 ml of sample was taken in an EDTA coated tube for the estimation of HbA1c. For further biochemical investigations serum was separated by centrifugation at 3000 rpm for 10 minutes and kept at -20° C until analysis. For urine analysis 24- hour urine sample was collected in a labeled clean plastic container.
Chemical and techniques
Albumin estimation was done by turbidimetric method, and according to the American Diabetes Association (ADA) based on 24-hour urinary albumin excretion levels [7]. Patients were categorized into three groups; diabetes with normoalbuminuria when albumin is excreted less than 30 mg/24 hrs., diabetes with microalbuminuria (30- 300 mg/24 hrs.), diabetes with macroalbuminuria (>300 mg/24 hrs.). Urinary creatinine was estimated by Jaffe’s method and taken as a cofounder for the estimation of ACR [8]. Creatinine clearance (CrCl) was estimated by Cockcroft-Gault formula [9]. Serum analysis for fasting blood glucose (FBG), post prandial blood glucose (PPBG), urea, creatinine and lipid profile parameters were analyzed by the fully automatic analyzer, ROCHE module Cobas 6000 (C-501 and C-601), and kits were procured by ROCHE. LDL-C and VLDL-C were estimated by Friedewald's equation [10]. HbA1c was estimated by direct enzymatic method. Reference ranges of various parameters according to the kits manufacturer are as follows; FBG (120 mg/dl), PPBG(200 mg/dl), HbA1c (5.5%), urea (7-20 mg/dl), creatinine (0.5-1.2 mg/dl), TC (200 mg/dl), Tg (150 mg/dl), HDL-C (≥ 40-50 mg/dl), LDL-C (120 mg/dl), VLDL-C (5-40 mg/dl), TC/HDL-C ratio (4), ACR(males 2-20 mg/mmol & females 2.8-28 mg/mmol).
Statistical Analysis
Data were represented as mean ± SD values shown in the tables. Statistical analysis was done by using one-way ANOVA to find out the difference between the groups at 0.05 level of significance, and to trace out the position of difference between each pair of means a post hoc test, "Tukeys Honest Test" was applied at 0.05 level of significance. Pearson's correlation coefficient was compassed between ACR and lipid profile parameters in all three groups to know the correlation, and the correlation coefficient (r) values were represented, at 0.05 level of significance.
Results
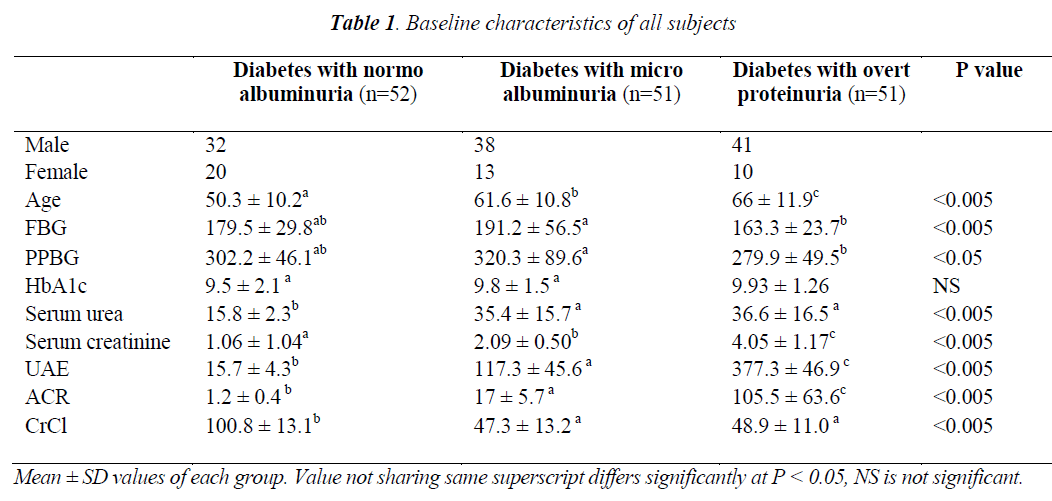
In the present study, a total of 154 type 2 diabetic subjects were included, out of them 111 were males and 43 were females. Mean age of the subjects was 55 years. Table 1 depicts the number of males and females diabetics with normoalbuminuria, diabetes with microalbuminuria and diabetes with overt proteinuria groups, with mean ± SD values of age and various biochemical parameters. The results upon comparing by one way ANOVA in the above mentioned groups were significantly different at the 0.005 level of significance, and values not sharing same superscripts differ significantly at P< 0.05.
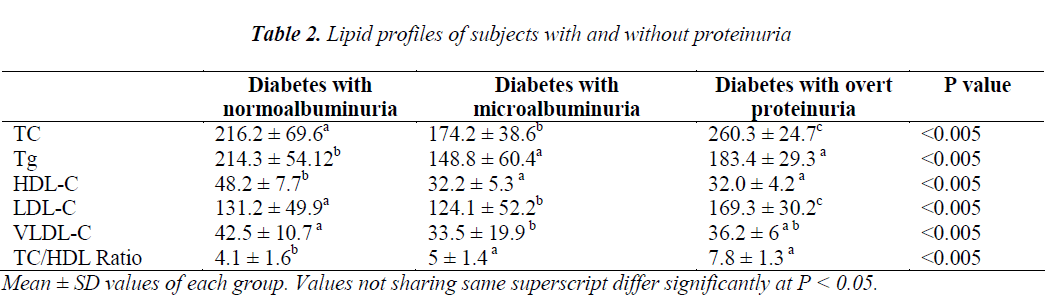
Lipid profile parameters were described and compared in Table 2. Mean ± SD values of TC, Tg, LDL, VLDL and TC/HDL ratio were significantly increased in all the three groups. Upon conducting the Post hoc test, it was observed that mean values of Tg, HDL, VLDL and TC/HDL ratio in diabetics with microalbuminuria group differ significantly from the diabetics with normoalbuminuria but not from the diabetics with overt proteinuria group. TC was significantly different between all three groups.
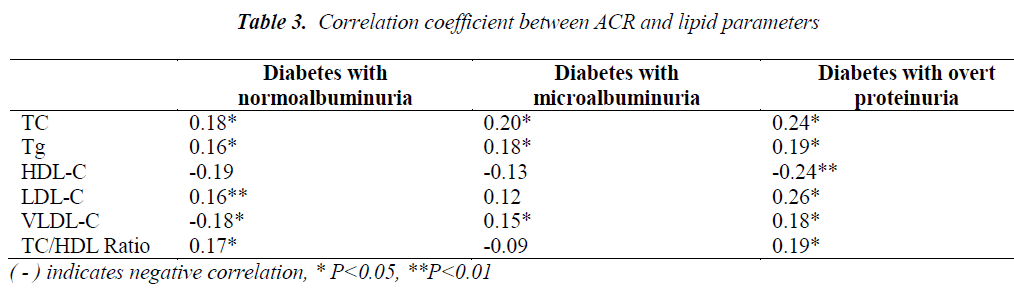
The correlation coefficient between ACR and lipid profile parameters is shown in Table 3. When analyzed in the aforementioned groups, ACR was positively correlated with TC, Tg, LDL,VLDL and found statistically significant, while HDL in all the groups and VLDL in diabetics with microalbuminuria were found to be negatively correlated at P<0.05.
Discussion
It has been predicted that worldwide the prevalence of diabetes (DM) in adults would increase to 5.4% by the year 2025, since the prevalence rate of 4.0% in 1995. It is expected that much of this increase in prevalence rate will occur in developing countries. In developing countries, most of the diabetic patients are in the age range of 45–64 years; while in developed countries most of them are ≥65 years [11]. Therefore, increasing diabetic prevalence will inevitably result in increasing proportions of death from CVD, diabetic nephropathy as well as other complications of DM [12].
In the present study according to furtherance of age, stages of diabetes can also be observed headway. FBG and PPBG were increased in DM with microalbuminuria group than DM with normoalbuminuria and DM with overt proteinuria, and both variables were significantly differ at P<0.05 with DM with overt proteinuria group.
Elevated levels of HbA1c were seen in DM with overt proteinuria with no statistically significant difference between all the groups (Table 1). This may be resulted from the long course of disease. HbA1c is referred to be an important marker for the assessment of glycemic control over a prolonged period of time, which moves in parallel with DM-induced complications [13].
In renal insufficiency, creatinine clearance and GFR decrease and inversely related to serum urea and creatinine. Although serum creatinine has great prognostic value compared to urea for anticipating inimical outcomes. Various studies showed that the serum urea level increased proportionally to the increase in serum creatinine [14,15]. Similarly in our study, serum urea and creatinine were found to be increased with the advancement of diabetes towards glomerular injury. Regardless with the increased level of serum urea and creatinine in DM with normoalbuminuric patients both the parameters differ at P<0.05 from different stages of renal insufficiency (Table 1). These results found to be similar to the findings of Pupet et.al. [16]. Therefore, continued hyperglycemia can lead to irretrievable damage to kidneys.
Rosario [17] reported that dyslipidemia is frequently noticed in patients with type 2 DM. The altered lipid profile rises from insulin resistance and defective insulin action, on lipoprotein metabolism. Thus, there is increased lipolysis with consecutively increased VLDL-C synthesis, Tg rich LDL synthesis, increased Tg's and quick breakdown of HDL-C [18].
Several studies have suggested the link between hypercholesterolemia in type 2 diabetic patients and the promotion of disease towards renal impairment [19,20]. As this variable in DM is positively linked to cardiovascular events, proteinuria in DM may also forecast for the same. Ravid et.al. [21] found that serum cholesterol levels in type 2 DM significantly related to the level of albuminuria. Similarly, Gall et.al. [22] also showed fellowship between hypercholesterolemia and urinary albumin excretion (UAE).
In the present study increased TC and LDL-C differ significantly between the three groups (Table 2) and positively associated with ACR when correlated for all ranges of albuminuria at P<0.05 and P<0.01 (Table 3). Elevated levels of LDL-C showed the requirement of lipid lowering therapy in our patients to limit the progression of disease towards CVD and ESRD. A review implies that the incidence of CVD can be controlled by the treatment of lipid lowering therapy in diabetic nephropathy. Lipid lowering therapies may prevent micro-vascular complications through pleiotropic effects like endothelial dysfunction, inflammatory pathways and vascular endothelial growth factors (VEGF) [23,24].
With the raised levels of TC, LDL-C, the levels of Tg, VLDL and TC/HDL-C ratio were also increased while HDL-C level was decreased among all the groups (Table 2). Tg, VLDL and TC/HDL-C ratio and HDL-C did not differ significantly between microalbuminuria and overt proteinuria; but DM with normoalbuminuria group. Our results agree with the findings of Rosario [17] but differ from Shoji et.al. [25] who had reported that normal TC, Tg, and LDL-C in diabetic patients with microalbuminuria did not alter from normal and microalbuminuria.
Tg, VLDL and TC/HDL-C ratio mean values were observed positively related to ACR, with respect to their correlation coefficient at P<0.05. Kim et.al. [26] reported that Tg as a key factor in the advancement of diabetic nephropathy. In a prospective study an increased TC/HDL ratio in type 2 diabetic patients was found independently associated with microalbuminuria [27].
No significant difference was observed between HDL-C mean values of diabetic microalbuminuria and overt proteinuria but the values in the groups were found with a significant difference when compared with diabetic with normoalbuminuria group (Table 2). HDL-C was found negatively correlated with ACR (Table 3). It functions in cellular cholesterol efflux and has anti-inflammatory and anti oxidative properties. The reduced HDL-C levels and increased risk of cardiovascular complication are well established [28-30].
From the present study it is evident that glucose levels, lipid profile and early proteinuria should be controlled so that further complications could be restricted. Importantly, a correlated lipid profile and albuminuria should be considered as alarming signal, so that by using therapeutic strategies, factors which are responsible for worsening the condition and dragging the disease towards CVD and ESRD could be properly managed.
Acknowledgement
The authors are thankful to be supported by the Research Center, “Center for Female Scientific and Medical Colleges”, Deanship of Scientific Research, King Saud University, Riyadh, KSA.
References
- Gross JL, Azevedo MJD, Silveiro SP, Canani LH, Ca- ramori ML, Zelmanovitz T. Diabetic Nephropathy: Di- agnosis, Prevention, and Treatment. Diabetes Care 2005; 28: 1164-1176.
- Chaturvedi N, Fuller JH, Taskinen MR. Differing asso- ciations of lipid and lipoprotein disturbances with the macrovascular and microvascular complications of type 1 diabetes. Diabetes Care 2001; 24: 2071–2077.
- Nosadini R, Tonolo G. Blood glucose and lipid control as risk factors in the progression of renal damage in type 2 diabetes. J Nephrol 2003; 16: 42-47.
- Khan FA, Khan MF, Patil SKB, Jameil N. Estimation of serum copper and magnesium levels in diabetic nephropathy patients. Asian J Biol Life Sci 2013; 2: 23-26.
- Adler AI, Stevens RJ, Manley SE, Bilous RW, Cull CA, Holman RR. Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64). Kidney Int 2003; 63: 225-232.
- Caramori ML, Fioretto P, Mauer M. Low glomerular filtration rate in normoalbuminuric type 1 diabetic pa- tients is associated with more advanced diabetic le- sions. Diabetes 2003; 52: 1036–1040.
- American Diabetes Association: Clinical Prac- tice Recommendations 2001. Diabetes Care 2001; 24:69-72.
- Hogg RJ, Portmann RJ, Milliner D, Lemley KV, Eddy A, Ingelfinger J. Evaluation and management of pro- teinuria and nephrotic syndrome in children: Recom- mendations from a pediatric nephrology panel estab-lished at the National Kidney Foundation conference on Proteinuria, Risk, Assessment, Detection, Elimina- tion (PARADE). Pediatrics 2000; 105: 1242-1249.
- Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31-41.
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein choles- terol in plasma. Clin Chem 1972; 18: 499–502.
- Wild S, Roglic G, Green A, Sicree R, King H. Global Prevalence of Diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004; 27:1047-1053.
- Amos AF, McCarty DJ, Zimmet P. The rising global burden of diabetes and its complications: estimates and projections to the year 2010. Diabetes Med 1997; 14:1–85.
- Gornik I, Brajkovic AV, Gasprovick V. Validation of HbA1c as a diagnostic marker for diabetes in the criti- cally ill. Critical Care 2010; 14: 14-24.
- Mittal A, Sathian B, Kumar A, Chandrashekharan N, Sunka A. Diabetes mellitus as a potential risk factor for renal disease among Nepalese: A hospital based con- trol study. Nepal J Epidemiol 2010; 1: 22-25.
- Kapoor S, Cohen D, Townsend R. Effects of Chronic Kidney Disease on Pulse Counter Analysis. Indian J Nephrol 2007; 17: 91-94.
- Puepet FH, Agaba EI, Chuhwak EK. Some metabolic abnormalities in type 2 diabetic patients in Jos, north central Nigeria. Nigerian J Med 2003; 12: 193-197.
- Rosario RF. Lipids and diabetic nephropathy. Current Diabetes Reports 2006; 6: 455-462.
- Trovati M, Cavalot F. Optimization of hypolipidemic and antiplatelet treatment in the diabetic patients with renal disease. J Am Soc Nephrol 2004; 15: 12-20.
- Gæde P, Vedel P, Larsen N, Jensen GVH, Parving H, Pedersen O. Multifactorial intervention and cardiovas- cular disease in patients with type 2 diabetes. N Engl J Med 2003; 348: 383-393.
- Sowers JR, Epstein M. Diabetes mellitus and associ- ated hypertension, vascular disease, and nephropathy: an update. Hypertension 1995; 26: 869-879.
- Ravid M, Neumann L, Lishner M. Plasma lipids and the progression of nephropathy in diabetes mellitus type 2: effect of ACE inhibitors. Kidney Int 1995; 47: 907-910.
- Gall MA, Hougaard P, Barch-Johnsen K. Risk factors for development of incipient and overt diabetic neph- ropathy in patients with noninsulin-dependent diabetes mellitus: prospective observational study. BMJ 1997; 314: 783-788.
- Danesh FR, Kanwar YS. Modulatory effects of HMG- CoA reductase inhibitors in diabetic microangiopa- thy. FASEB J 2004; 18: 805-815.
- Rosenson RS. Treatment of hyperlipidemia and be- yond. Expert Rev Cardiovascular Therap 2008; 6: 13:19-30.
- Shoji T, Emoto M, Kawaghishi T. Atherogenic lipo- protein changes in diabetic nephropathy. Atherosclero- sis 2011; 156: 425-433.
- Kim DM, Ahn CW, Park JS. An implication of hyper- triglyceridemia in the progression of diabetic neph- ropathy in metabolically obese, normal weight patients with type 2 diabetes mellitus in Korea. Diabetes Res Clin Pract 2004; 66: 169-172.
- Retnakaran R, Cull CA, Thorne KI. Risk factors for renal dysfunction in type 2 diabetes: UK Prospective Diabetes Study 74. Diabetes 2006; 55: 1832-1839.
- Barter P. HDL-C: role as a risk modifier. Atheroscler Suppl 2011; 12: 267-270.
- Soran H, Hama S, Yadav R, Durrington PN. HDL functionality. Curr Opin Lipidol 2012; 23: 353-366.
- Eren E, Yilmaz N, Aydin O. High density lipoprotein and its dysfunction. Open Biochem J 2012; 6: 78-93.