Research Article - Biomedical Research (2017) Volume 28, Issue 21
Comparison of the effect of two methods of intraductal injection of the nipple catheter and modified up-the-teat intraductal injection in construction of tree shrew breast cancer model
Nuo Yang1,2#, Siwen Wu1#, Rong Zheng1#, Panpan Huang1, Xiaoxue Li1, Sufang Zhou1* and Yongxiang Zhao1,2*
1National Center for International Research of Biological Targeting Diagnosis and Therapy, Guangxi Key Laboratory of Biological Targeting Diagnosis and Therapy Research, Collaborative Innovation Center for Targeting Tumor Diagnosis and Therapy, Guangxi Medical University, Nanning, Guangxi 530021, PR China
2Department of Cardio-Thoracic Surgery, the First Affiliated Hospital of Guang Xi Medical University, Nanning 530021, PR China
#These authors contributed equally
- *Corresponding Authors:
- Sufang Zhou
National Center for International Research of Biological
Targeting Diagnosis and Therapy
Guangxi Key Laboratory of Biological Targeting Diagnosis
and Therapy Research
Collaborative Innovation Center for Targeting Tumor
Diagnosis and Therapy
Guangxi Medical University, PR China
Yongxiang Zhao
National Center for International Research of Biological
Targeting Diagnosis and Therapy
Guangxi Key Laboratory of Biological Targeting Diagnosis
and Therapy Research
Collaborative Innovation Center for Targeting Tumor
Diagnosis and Therapy
Guangxi Medical University, PR China
Accepted on November 16, 2017
Abstract
Background: Topical administration through the mammary duct is an important way to study breast cancer treatment. However, there is a lack of suitable primate animal model of breast cancer for studying topical administration treatment through the mammary duct.
Methods: 12 female healthy tree shrews at the age of 6 months were selected and randomly divided into experimental group (n=6) and control group (n=6). The experimental and control groups were respectively treated using protecting the intraductal injection of the nipple catheter (PIINC) method and modified up-the-teat intraductal injection (MUTII) method to establish the tree shrew breast cancer model. The degree of drug dispersion in mammary duct system, morphological changes of nipples after injection, efficiency and time of tumor formation were compared between the two groups.
Results: The drug injected by PIINC method can successfully enter the mammary gland along the ductal-alveolar tree just like by MUTII method. However, edema, bleeding and injury of nipples and surrounding tissues were not observed after the PIINC injection but can be observed after MUTII injection. More importantly, intraductal injection by PIINC not only can successfully construct the tree shrew breast cancer model but also can be repeated in the same nipple after the model construction which could not be realized by MUTII. There was no significant difference in the efficiency and time of tumor formation between the two methods (P<0.05).
Conclusions: PIINC is a more suitable method than MUTII to construct the tree shrew breast cancer model for studying topical administration treatment through the mammary duct.
Keywords
Intraductal injection, Breast cancer, Tree shrew.
Introduction
Breast cancer is the most common malignancy in women around the world. Each year about 1.7 million women are suffering from breast cancer and about 50 million people die from breast cancer [1,2] which is largely due to lack of economical and effective treatment. The current common treatment of breast cancer includes intravenous chemotherapy, radiotherapy and surgical resection, but these treatment methods exist systemic drug toxicity, damage to normal tissue, high cost, high recurrence rate and so on [3,4]. It has been reported that topical administration through the mammary duct can not only achieve effective outcomes for rodent breast cancer, but also significantly reduce drug toxicity, side effects and cost [5-7]. In order to further study the treatment of topical administration of breast cancer through the mammary duct, it is particularly important to construct a suitable animal model.
Tree shrew (Tupaia belangeri chinensis) belongs to primates in species category and is similar to human in genetic background [8,9]. Comparing with rodents, the study of tree shrew is more likely to be transformed from experimental to clinical practice. It has been reported that the breast cancer model can be successfully constructed by injecting PyMT lentivirus into the mammary gland of tree shrew, and the model was very similar to human breast cancer [10]. But during the construction of tree shrew breast cancer model, the intraductal injection method used in the above literature was modified up-the-teat intraductal injection (MUTII) [11]. The MUTII process involves cutting the top of the nipple to fully expose the breast duct and then injecting the PyMT overexpression lentivirus into the mammary duct to complete model construction. Although this kind of injection method is beneficial for fully exposure of breast duct opening to facilitate the intraductal injection, it leads to nipple injury and formation of scars to block the catheter opening and make repeated intraductal injection in the same nipple after the model construction impossible. So it is not conducive to the research on the topical administration treatment of breast cancer through the mammary duct.
In this study, we established a noninvasive tree shrew breast intraductal injection method, that is protecting the intraductal injection of the nipple catheter (PIINC), to construct tree shrew breast cancer model. This method not only provides a successful method for construction of the tree shrew breast cancer model, but also protects the nipple during the modeling process so that the same nipple can be repeatedly treated by intraductal injection after model construction. Therefore, it provides a suitable primate animal modeling method that is beneficial for studying topical administration treatment of breast cancer through the mammary duct. It is of great significance for human breast cancer research.
Materials and Methods
Tree shrew (Tupaia belangeri chinensis)
Experimental female tree shrews were purchased from Kunming Institute of Zoology, Chinese Academy of Sciences and Kunming Medical University. All tree shrews were healthy at the age between 6 months and weighed 150 ± 17 g (Table 1). All animal experimental programs are approved by the Guangxi Medical University Animal Ethics Committee, in line with experimental animal ethical requirements.
| Groups | N | Weight | Number of adverse effects after injection | ||
|---|---|---|---|---|---|
| edema | bleeding | injury | |||
| experimental group | 6 | 150.9 ± 16.5 | 0 | 0 | 0 |
| control group | 6 | 151.7 ± 17.8 | 6 | 5 | 5 |
| P | <0.05 | <0.05 | <0.05 | <0.05 | |
Table 1. Comparison of weight and number of adverse effects after injection between 2 group.
In this study, 12 tree shrews were averagely and randomly divided into experimental group and control group. The experimental group was treated by PIINC method and control group was treated by MUTII method to construct the tree shrew breast cancer model.
Lentivirus
The PyMT overexpression lentivirus used for constructing the model was from Kunming Institute of Zoology, Chinese Academy of Sciences and Kunming Medical University.
Laboratory equipment and reagents
Microliter syringe (50 μl) with a 33 G or 25 G metal hub needle was purchased from Swiss company Hamilton. Microsurgical tweezers were purchased from Ningbo Chenghe Microscope Factory. 4.5 lacrimal probe was purchased from Suzhou Concord Medical Equipment Factory. Ophthalmology scissors were from Guangdong Jieyang City Kang Huajia Medical Devices Co., Ltd. Inverted phase contrast microscope (TS100-F) was purchased from Nikon Co., Ltd., Japan. Trypan blue purchased from Shanghai Health Lei Chemical Co., Ltd. Ketamine hydrochloride injection (2 mL, 0.1 g) was purchased from Fujian Gutian Pharmaceutical Co., Ltd.
Preoperative preparation
Initially, each tree shrew was weighted and intramuscularly injected with ketamine hydrochloride (40 mgkg-1) [12]. Depilatory cream was then evenly applied around the second pair of nipples. Hair was then removed about 5 min later using wet cotton swab, and cream around the nipples was cleaned using warm water. The shaving device was avoided during hair removal because it may cause nipple injury. Finally, the nipple area was sterilized with alcohol cotton swabs.
Intraductal injection
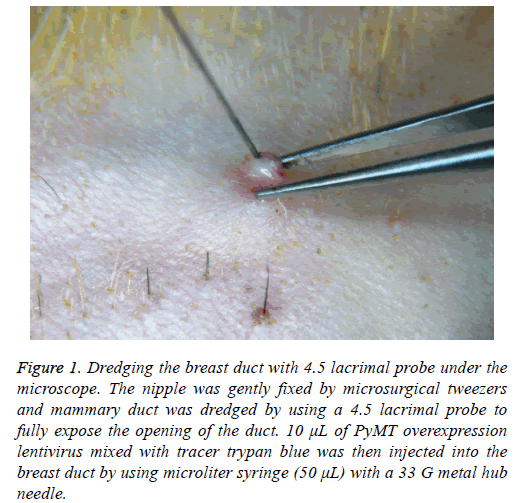
PIINC: The dead skin, secretions and other impurities covering the nipples were initially removed by microsurgical tweezers to fully expose the nipples under the microscope. And then, as shown in Figure 1, the nipple was gently fixed by microsurgical tweezers and mammary duct was dredged by using a 4.5 lacrimal probe to fully expose the opening of the duct. 10 μl of PyMT overexpression lentivirus mixed with tracer trypan blue was then injected into the breast duct by using microliter syringe (50 μL) with a 33 G metal hub needle. The drug injection rate was maintained at about 50 μLmin-1 because too fast injection may damage the breast duct.
Figure 1: Dredging the breast duct with 4.5 lacrimal probe under the microscope. The nipple was gently fixed by microsurgical tweezers and mammary duct was dredged by using a 4.5 lacrimal probe to fully expose the opening of the duct. 10 μL of PyMT overexpression lentivirus mixed with tracer trypan blue was then injected into the breast duct by using microliter syringe (50 μL) with a 33 G metal hub needle.
MUTII: The nipples were gently fixed by microsurgical tweezers in the MUTII process as in the PIINC process, followed by cutting the top of nipple with ophthalmic scissors to fully expose the opening of the duct. 10 μL of PyMT overexpression lentivirus mixed with tracer trypan blue was then injected into the breast duct by using microliter syringe (50 μL) with a 25 G metal hub needle. The drug injection rate was maintained at about 50 μL min-1 as well.
Postoperative observation
The injection site and surrounding tissue were then observed overtime. The swollen skin around the nipple indicated that the drug was not successfully injected into the breast duct but into the subcutaneous tissues around the nipple. The tree shrew was then taken back into a separate and warm cage and close observation was needed until the tree shrew recovered consciousness and activity.
Statistical analysis
All data were analyzed using GraphPad Prism 5.0 software (CA, USA) and results were expressed as mean ± SD. Comparisons between two groups were performed using an unpaired two-sided t-test. P<0.05 indicated that the difference was statistically significant.
Results
Drugs injected by PIINC successfully entered into breast tissue along the breast duct system
Same as in human, the mammary gland of tree shrews consists of a complex ductal-alveolar tree that converges into a central duct at the nipple. During the intraductal injection, trypan blue was used as a tracer. The dendritic dispersion of drug indicated that the drug was successfully injected into the breast duct. On the contrary, when the drug was observed flaky, it demonstrated that the drug failed to enter into the breast duct but into the subcutaneous tissue. Results indicated that the injected drug through the PIINC method can successfully enter the breast duct, which was like the MUTII method.
Nipples were effectively protected during the PIINC process
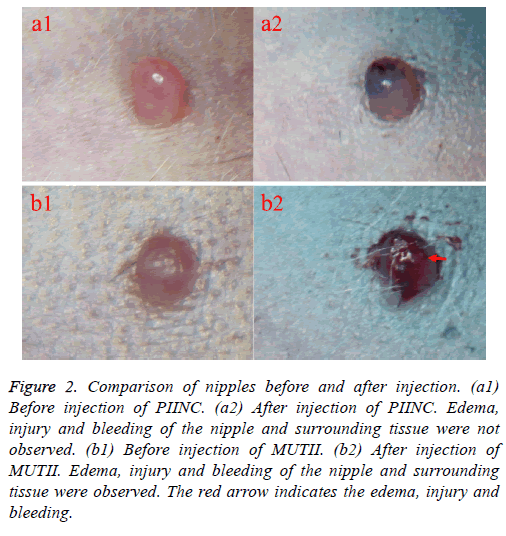
During the PIINC process, we used a 4.5 lacrimal probe to dredge mammary duct rather than cutting the top of the nipple to fully expose the breast duct opening. Results showed that edema, injury and bleeding of the nipple and surrounding tissue were not observed after injection by PIINC, contrary to what were observed after injection by MUTII (Figure 2). Besides, drugs were easily refluxed and dispersed from the opening of the duct to the subcutaneous tissue around the nipple after injection by MUTII.
Figure 2: Comparison of nipples before and after injection. (a1) Before injection of PIINC. (a2) After injection of PIINC. Edema, injury and bleeding of the nipple and surrounding tissue were not observed. (b1) Before injection of MUTII. (b2) After injection of MUTII. Edema, injury and bleeding of the nipple and surrounding tissue were observed. The red arrow indicates the edema, injury and bleeding.
PIINC can be repeated many times in the same nipple after model construction
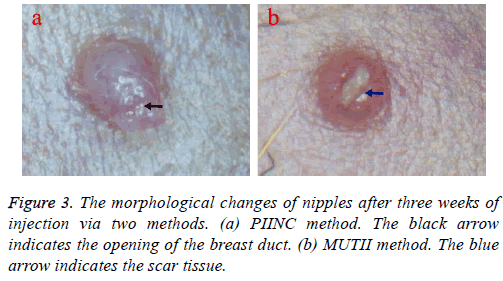
The morphological changes of nipples injected by the two methods respectively were also observed under the microscope three weeks after injections. Result showed that the opening of the mammary duct injected via the PIINC method was still clearly visible and the intraductal injection could be repeated at the same nipple weekly or biweekly over several months.
Oppositely, the opening of breast duct injected via the MUTII method was blocked by scar formed in the previous injury site which, making it impossible to repeat intraductal injection in the same nipple (Figure 3).
No significant difference in tumorigenic efficiency and tumor formation time between PIINC and MUTII methods
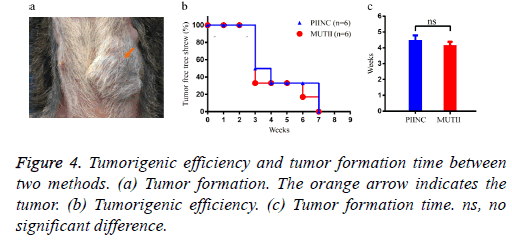
After 3-7 weeks, the breast of all the 12 tree shrews were cut for biopsy which showed all tree shrews were succeeded in growing the tumors (Figures 4a and 4b), meant the tumorigenic efficiency was 100%. In addition, there was no significant difference in tumor formation time between two methods (P<0.05) (Figure 4c).
Pathologically confirmed: the tumor induced by PIINC is indeed tree shrew breast cancer
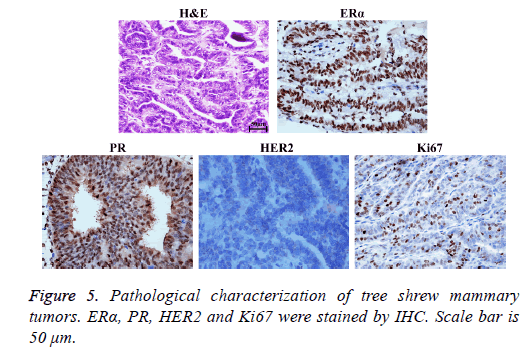
ERα, PR, Ki67 were positive and Her2 was negative in Tree shrew breast cancer. H&E staining and immunohistochemical method (IHC) confirmed these results in the tree shrew mannary tumors induced by PIINC, which meant the tumors were Tree shrew breast cancer (Figure 5).
Discussion
Breast cancer is the most common malignancy in women and seriously threatens human health [13]. So its diagnosis and treatment have drawn great attention [14,15]. The current common treatment of breast cancer includes intravenous chemotherapy, radiotherapy and surgical resection, but these treatment methods exist systemic drug toxicity, damage to normal tissue, big expense, high recurrence rate and so on [3,4]. It has been reported that the topical administration of chemotherapy drugs through intraductal injection has a better therapeutic effect and less systemic toxicity in both patients and mouse breast cancer models [5-7,16], which provides a new direction for breast cancer treatment. It is therefore of urgent demand to construct valuable animal models to further study on topical administration treatment through the mammary duct. At present, rodents, especially transgenic mice and rats, are used as animal models commonly used in breast cancer studies [17,18]. However, due to big differences between rodents and human species, many treatment methods which are effective in the rodent models have poor performance in treatment of human diseases, leading to barriers in their transformation from experimental to clinical practice [19]. Tree shrews are primates whose species characteristics are closer to human than rodents. Mammary gland development process in tree shrews is also similar to that of human, which is divided into four stages (virgin period, pregnancy, lactation and degeneration). In addition, the mammary gland structure of the tree shrew is similar to that of human as well, consisting of a complex ductal-alveolar tree that converges into a central duct at the nipple [20]. Compared with the rodents, research on tree shrews are more significant to clinical transformation [21,22].
Topical administration treatment through the mammary duct is an important way to study breast cancer treatment [5-7]. Ge et al. have successfully constructed the tree shrew breast cancer model by intraductally injecting the overexpression PyMT lentivirus which provided a more suitable animal model for human breast cancer research than rodents [10], but the modeling process involved cutting part of the nipple and the modeling method was invasive. Scar tissues were then formed in the site of injured nipple after model construction, leading to the blockage of opening of breast duct and failure of repeated intraductal injections in the same nipple. This was thus not conducive in studying the topical administration treatment of breast cancer through the mammary duct. Accordingly, we established a new method called PIINC to construct tree shrew breast cancer model. During PIINC process, we used a 4.5 lacrimal probe to dredge mammary duct instead of cutting the part of the nipple to fully expose the breast duct opening, which can protect the integrity of the nipple and its duct effectively. This can keep the breast duct open without obstruction by scar tissues and achieve the repeated intraductal injection effect in the same nipple successfully after the model construction.
Therefore, the PIINC method is more suitable for the tree shrew breast cancer model construction than MUTII method because PIINC method is more conducive for follow-up topical administration treatment research of breast cancer through mammary duct. In addition, the reported results herein indicated that there was no significant difference in the dispersion of drug in the mammary ducts, tumor formation efficiency and tumor formation time between the two methods (P<0.05). At last, we also confirmed through the pathology that the tumors induced by PIINC method were indeed mammary tumors since ERα, PR, HER2 and Ki67 were biomarkers routinely used in clinical diagnosis of breast cancers. However, the PIINC method has some shortcomings, too. For example, microscope is a necessary instrument in this method and the 33 G needle used during the operation is not easy to control because it is fine and soft. The technical requirement for this method is therefore relatively high. These shortcomings need to be further improved.
Conclusion
In this study, we successfully established a noninvasive intraductal injection method to construct the tree shrew breast cancer models, which can protect integrity of the nipple and its duct effectively, and provide technical support for repeated intraductal injection in the same nipple after the tree shrew breast cancer model construction. So this is an effective method for tree shrew breast cacer model construction and more conducive for the research of topical administration of breast cancer through mammary duct than conventional intraductal injection method. The establishment of this method is of great significance to human breast cancer research.
Acknowledgement
Funding: This work was supported, in part, by grants from National Major Science and Technology Major Project- National Major New Drug Creation (No.2015GKS-462); Programs for Changjiang Scholars and Innovative Research Team in University (No.IRT_15R13); National Natural Scientific Foundation of China (Nos. 81430055 and 81372452); Guangxi Science Research and Technology Development Project (No.guikehe 1599005-2-10); Interprovincial cooperation projects (No.guikehe 14251001).
Ethical Statement
All experiments were carried out according to the guidelines of the Federation of European Laboratory Animal Science Associations, and all protocols were approved by the Institutional Animal Care and Use Committee of Guangxi Medical University.
References
- Speers C, Zhao SG, Kothari V. Maternal embryonic leucine zipper kinase (MELK) as a novel mediator and biomarker of radioresistance in human breast cancer. Clin Cancer Res 2016; 22: 5864-5875.
- Cloughgorr K, Silliman RA, Moser A. Motivation and mortality in geriatric patients with early stage breast cancer. J Clin Oncol 2015; 33: 105.
- Nixon NA, Khan OF, Imam H. Drug development for breast, colorectal, and non-small cell lung cancers from 1979 to 2014. Cancer 2017.
- Mamounas EP, Kuehn T, Rutgers EJ. Current approach of the axilla in patients with early-stage breast cancer. World 2017.
- Zhang B, Love SM, Chen G. The safety parameters of the study on intraductal cytotoxic agent delivery to the breast before mastectomy. Chin J Cancer Res 2014; 26: 579-587.
- Mahoney ME, Gordon EJ, Rao JY. Intraductal therapy of ductal carcinoma in situ: a presurgery study. Clin Breast Cancer 2013; 13: 280-286.
- Stearns V, Mori T, Jacobs LK. Preclinical and clinical evaluation of intraductally administered agents in early breast cancer. Sci Transl Med 2011; 3: 106ra08.
- Fan Y, Huang ZY, Cao CC. Genome of the Chinese tree shrew. Nat Commun 2013; 4: 1426.
- Xiao J, Liu R, Chen CS. Tree shrew (Tupaia belangeri) as a novel non-human primate laboratory disease animal model. Zool Res 2017; 38: 127.
- Ge GZ, Xia HJ, He BL. Generation and characterization of a breast carcinoma model by PyMT overexpression in mammary epithelial cells of tree shrew, an animal close to primates in evolution. Int J Cancer 2016; 138: 642-651.
- Siwko SK, Bu W, Gutierrez C. Lentivirus-mediated oncogene introduction into mammary cells in vivo induces tumors. Neoplasia 2008; 10: 653-662.
- Ge GZ, Xia HJ, He BL. Generation and characterization of a breast carcinoma model by PyMT overexpression in mammary epithelial cells of tree shrew, an animal close to primates in evolution. Int J Cancer 2016; 138: 642-651.
- Zheng R, Zeng H, Zhang S. Estimates of cancer incidence and mortality in China, 2013. Chinese J Cancer 2017; 36: 66.
- Silwal-Pandit L, Nord S, von der Lippe Gythfeldt H. The longitudinal transcriptional response to neoadjuvant chemotherapy with and without bevacizumab in breast cancer. Clin Cancer Res 2017.
- Petrelli F, Ghidini M, Lonati V. The efficacy of lapatinib and capecitabine in HER-2 positive breast cancer with brain metastases: A systematic review and pooled analysis. Euro J Cancer 2017; 84: 141-148.
- Brock A, Krause S, Li H. Silencing HoxA1 by intraductal injection of siRNA lipidoid nanoparticles prevents mammary tumor progression in mice. Sci Transl Med 2014; 6: 217ra2.
- Al Faraj A, Shaik AS, Ratemi E. Combination of drug-conjugated SWCNT nanocarriers for efficient therapy of cancer stem cells in a breast cancer animal model. J Control Release 2016; 225: 240-251.
- Pyter LM, Suarezkelly LP, Rd CW. Novel rodent model of breast cancer survival with persistent anxiety-like behavior and inflammation. Behavioural Brain Res 2017.
- Knop J, Joëls M, Veen RVD. The added value of rodent models in studying parental influence on offspring development: opportunities, limitations and future perspectives. Curr Opinion Psychol 2017.
- Xu L, Chen SY, Nie WH, Jiang XL, Yao YG. Evaluating the phylogenetic position of Chinese tree shrew (Tupaia belangeri chinensis) based on complete mitochondrial genome: implication for using tree shrew as an alternative experimental animal to primates in biomedical research. J Genet Genomics 2012; 39: 131-137.
- Xia HJ, Wang CY, Zhang HL. Characterization of spontaneous breast tumor in tree shrews (Tupaia belangeri chinenesis). Dongwuxue Yanjiu 2012; 33: 55-59.
- Xia HJ, He BL, Wang CY. PTEN/PIK3CA genes are frequently mutated in spontaneous and medroxyprogesterone acetate-accelerated 7,12-dimethylbenz(a)anthracene-induced mammary tumours of tree shrews. Eur J Cancer 2014; 50: 3230-3242.