Research Article - Biomedical Research (2017) Health Science and Bio Convergence Technology: Edition-II
Clinical significance of serum hyaluronic acid level alteration in patient with nasopharyngeal carcinoma
Li Chen1, Jianhua Yu2, Zhihui Lu1, Jiaxuan Qiu3*1Department of Oncology, the First Affiliated Hospital of Nanchang University, Nanchang, PR China
2Department of Oncology, Jiangxi Provincial People's Hospital, Nanchang, PR China
3Department of Oral and Maxillofacial Surgery, the First Affiliated Hospital of Nanchang University, Nanchang, PR China
- *Corresponding Author:
- Jiaxuan Qiu
Department of Oral and Maxillofacial Surgery
The First Affiliated Hospital of Nanchang University
Nanchang, PR China
Accepted date: March 9, 2017
Abstract
To determine the significance of serum Hyaluronic Acid (HA) levels in prognosis of patients with Nasopharyngeal Carcinoma (NPC) by observing the variations of serum HA levels in patients before and after the radiotherapies.116 serum samples, including 40 from NPC patients prior to the treatment, 36 from NPC patients after the treatment, and 40 from healthy individuals were collected. Radioimmunology assays were performed to determine the serum HA levels. The results showed that Serum HA levels are 3.75-fold elevated in the NPC patients (205.8 ± 62.7 μg/L) compared with healthy individuals (54.9 ± 19.2 μg/L, P<0.001). The threshold of the serum HA level was set as 74.1 μg/L, which exhibits 85.0% of sensitivity, 87.5% of specificity, and 86.3% of accuracy for positive NPC determination. Serum HA levels in the patients treated with radiotherapies (117.5 ± 46.3 μg/L) were significantly lower than those in the patients without treatments (194.4 ± 62.4 μg/L, P<0.01). Statistical analysis revealed that the serum HA level is positively correlated with clinical stages and local invasion (P<0.01). These results suggested that the serum HA level may be served as a sensitive and specific tumor marker for the detection, monitoring, and prognosis of NPC.
Keywords
Nasopharyngeal carcinoma, Hyaluronic acid.
Introduction
Nasopharyngeal Carcinoma (NPC) is one of the most common head and neck malignant tumors in China, which can be treated by combination of drug delivery, as well as radio and chemotherapies. Although the cancerous cells can be attacked by the immune system, immune escape could lead to metastasis of the cancerous cells, reducing the effect of the treatments of the radio and chemotherapies. Many studies have demonstrated the circulating Hyaluronic Acid (HA) levels are significantly related to the metastasis of tumors [1].
HA is a linear polysaccharide consisting of repeated units of nonsulfated glycosaminoglycans, widely distributed in connective tissues and Extracellular Matrix (ECM) [2-4]. As one of the major components of ECM, HA significantly contributes to the maintenance of cell hydration, osmotic pressure, and the resilience of cartilage [3,5]. Also, HA can interact with various cellular receptors, including CD44, RHAMM, and LYVE-1, to promote angiogenesis [6], as well as the growth, invasion and metastasis of tumors [7-9]. The circulating HA has also been found to facilitate cancerous cells to escape the immune surveillance [7], and reduce the delivery of chemotherapeutic agents, including doxorubicin, and mitomycin C [10].
Clinical studies have revealed elevated serum HA levels in the patients with various solid tumors, such as gastric cancer, bladder carcinoma, prostate cancer, head and neck squamous cell carcinoma, ovarian cancer, colon cancer, breast cancer, and lung cancer [11,12]. Lokeshwar et al. used urine HA levels to diagnose the bladder cancer, exhibiting 83.1% of sensitivity and 90.1% of specificity, indicating the urinary HA is a sensitive and specific diagnostic marker for the bladder cancer. In addition, the urinary HA levels also indicated the probabilities of recurrence of the bladder cancer.
Local fibrosis and chronic inflammation of nasopharynx after radiotherapies is challenging the diagnosis of the recurrence of NPC, which is an essential factor for NPC prognosis. The diagnosis of the recurrence of NPC, as well as the prognosis of NPC after radio or chemotherapies can significantly increase survival time and rate [13,14]. A significant element for NPC prognosis is the clinical TNM stages of NPC, which involves the circulating HA levels [11,15,16]. Studies on the gastric cancer, colon cancer and breast cancer suggested the serum HA levels were related with the survival time and the prognosis of the patients. Therefore, in this study, we observed the changes of serum HA levels in the patients before and after the treatments to determine the significance of the HA in the prognosis of patients with NPC.
Materials and Methods
Patients
40 patients accepting their first treatment from May to July of 2005, at the age of 47.7 ± 12.7, were collected in this study, including 28 males and 12 females, and identified as poorlydifferentiated squamous cell carcinoma by pathological histology. In according to the 92 Fuzhou staging standard, T represents local invasive area of NPC with 1-4 indicating increased areas and N represents the clinical stages of NPC. The 40 patients were classified as one case of phase I (one case of T1N0), seven cases of phase II (three T1N1, one case of T2N0, three cases of T2N1), 16 cases of phase III (two of T1N2, six of T2N2, three of T3N0, three of T3N1, two of T3N2), 16 cases of phase IVA (one of T1N3, one of T2N3, two of T3N3, two of T4N0, five of T4N1, three of T4N2, and two of T4N3). Radiotherapies, chemotherapies or the combination of radio and chemotherapies were performed to each patient dependent upon the NPC stages. 40 serum samples were obtained from healthy individuals at the age of 47.2 ± 12.4, including 26 males and 14 females. The informed consent was acquired before the sampling procedure, and the experiments were approved by a local ethics committee.
Serum collection and storage
2 ml peripheral venous blood was collected from the healthy individuals and the patients with empty stomach before and after their radio- and/or chemotherapies. Blood samples were centrifuged immediately after collection at 2500 rpm. The supernatant was collected and stored at -20°C. Competitive radioimmunoassays (Haiyan Medical and Biological Technologies, Ltd., Shanghai, China) were performed to determine the serum HA levels, as per the protocol provided by the manufacturer.
Statistical analysis
Continuous variables were presented as mean ± SD, categorical variables were presented as percentages (%). Mean ± SD of the control group indicates the positive detection. Test of normality and homogeneity test of variance were performed prior to any statistical analysis. Variance analysis and pairedsamples t-test were performed to compare means. All the statistical analysis was performed with SPSS 10.0, with α=0.05, P<0.05 indicating significant differences.
Results
Serum HA levels from NPC patients compared with that from healthy controls
To evaluate the significance of serum HA levels in the diagnosis of NPC, the serum HA levels from the patients and controls were determined. The NPC patients showed 205.8 ± 62.7 μg/L serum HA, approximately 3.75-fold higher than 54.9 ± 19.2 μg/L of the healthy controls (P<0.001). The threshold for positive detection of NPC was set as 74.1 μg/L (54.9 + 19.2 μg/L), discovering 6 NPC cases lower than the threshold and 5 healthy cases higher than the threshold. These results suggested the use of serum HA levels in NPC diagnosis exhibited 85.00% (34/40) of sensitivity, 87.50% (35/40) of specificity, and 86.25% (69/80) of accuracy.
Serum HA levels from NPC patients before treatments compared with that after treatments
4 of the NPC patients did not complete the treatments. Therefore, only 36 serum samples were analysed to study the effect of treatments on the changes of HA levels. Before treatments, the serum HA levels were 194.4 ± 62.4 μg/L and were significantly decreased due to the treatments, reaching 117.5 ± 46.3 μg/L (P<0.01).
The involvement of serum HA levels in the NPC stages
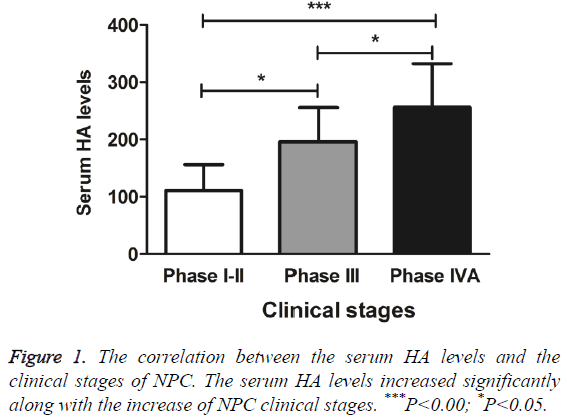
To further investigate the sensitivity, specificity and accuracy of the serum HA levels in diagnosing NPC, the correlation between serum HA levels and the stages, local invasive areas, and lymphatic metastasis was analysed. The only case of phase I was merged with phase II cases for analysis. As summarized in Figure 1, the serum HA levels increased significantly along with the increase of NPC clinical stages (F=13.48, P<0.0001). Phase IVA showed significantly higher level of serum HA than phase I-II did (q=7.30, P<0.001). Also, phase I-II and phase III, phase III and phase IVA patients exhibit significant different serum HA levels (q=4.27 and 3.71 respectively, P<0.05).
The correlation between serum HA levels and local invasive area of NPC
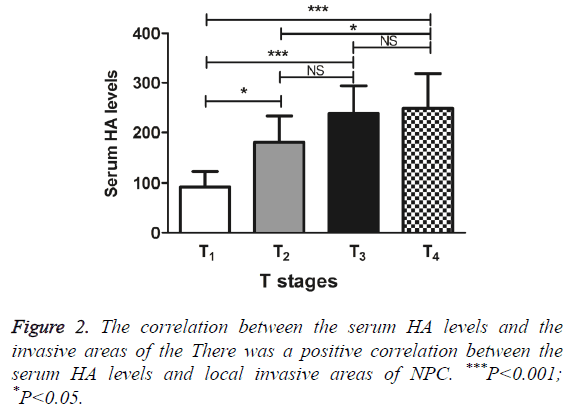
The serum HA levels increased, as local invasive areas of NPC increased from T1 to T4. Compared with T1, the serum HA levels of T2, T3 and T4 were significantly increased (q=4.55, 7.41 and 8.21, P<0.05, 0.001 and 0.001 respectively). The serum HA levels of T2 and T4 were also showed significant difference (q=4.08, P<0.05). However, the serum HA levels of T2 and T3, as well as the serum HA levels of T3 and T4 had no significant difference, with q=3.32 and 0.59, respectively (P>0.05) (Figure 2). The results indicated the positive correlation between the serum HA levels and local invasive areas of NPC.
The correlation between serum HA levels and NPC neck lymphatic metastasis
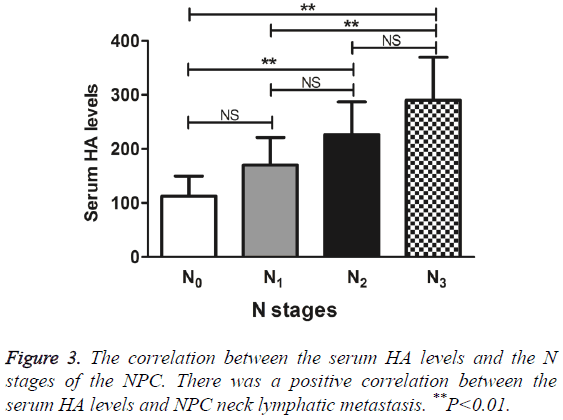
The serum HA levels increased, as the clinical stages of NPC increased from N0 to N3. Compared with N0, N2 and N3 had significantly higher levels of serum HA, N3 also exhibited a significantly higher level of serum HA than N1 did (q=6.00, 7.85, and 6.03, respectively, P<0.01). Nevertheless, the serum HA levels between N0 and N1, N1 and N2, and N2 and N3 had no significant differences (q=3.08, 3.59, and 3.16, respectively, P>0.05) (Figure 3). The results suggested the positive correlation between the serum HA levels and NPC neck lymphatic metastasis.
Discussion
As one of the most common head and neck malignant tumors in China, the diagnosis and prognosis of the NPC have been suggested to be essential to influence the survival time and rate of the NPC patients. Therefore, in this study, we aim to evaluate the significance of the serum HA levels for NPC diagnosis and prognosis. By comparing the serum HA levels between the NPC patients and the healthy controls, we found the patients showed 3.75-fold higher levels of serum HA prior to the treatments than the healthy controls (P<0.001). With the threshold for positive NPC detection at 74.1 μg/L, serum HA levels exhibited 85% of sensitivity, 87.5% of specificity, and 86.25% of accuracy as an NPC diagnostic marker. Studies on various types of cancers have discovered that the elevated HA levels were produced by the cancerous cells or/and tumor interstitial cells [11,12,17,18]. Studies on the head and neck tumors by Franzmann et al. [18] have shown the expression of hyaluronic acid and hyaluronidase by RT-PCR, as well as considerable amount of HA in the supernatant of the cell culture [18]. Although the elevated serum HA levels were detected in our study, the causes of the increase remains unknown, and will be investigated in future studies
This study also compared the serum HA levels in the patients before and after the radio- or chemotherapies. The results showed that the treatments significantly reduced the serum HA levels, suggesting the serum HA levels might be used as one of the indicators for the efficacy of the treatments. However, the patients after the treatments also exhibited higher levels of serum HA than the healthy controls did, this might be due to the immediate detection of the HA levels after the treatment, as the tumors would be depleted in three months after the treatment [19].
The statistical analysis of this study revealed the serum HA levels were positively correlated with the clinical stages, local invasion, and lymphatic metastasis of NPC. This can be explained as the HA has been found to promote the formation of lymphatic capillaries of the NPC tissues [20]. In addition, the increased amount of LYVE-1 expressed lymphatic capillaries in the tumors could lead to lymphatic metastasis, local recurrence and poor prognosis of the tumors [21]. The interaction of HA and LYVE-1 facilitates the formation of lymphatic vessel tumor thrombus during tumor invasion [22]. Although this study did not discover the correlation between the serum HA levels and the distant metastasis of NPC, some studies indicated the elevated HA levels can be used as an independent factor to predict the metastasis of NPC [16,23].
References
- Huang JH, Mu ZL, Zhou XJ, Huang QL, Gao F. Effect of Yupingfeng granules on HA and Foxp3(+) Treg expression in patients with nasopharyngeal carcinoma. Asian Pac J Trop Med 2015; 8: 674-676.
- Orasan OH, Orasan RA, Patiu IM, Dumitrascu DL. Hyaluronic acid in end-stage renal disease treated by hemodialysis. Ren Fail 2015; 37: 1531-1532.
- Selyanin MA, Boykov PY, Khabarov VN. Hyaluronic acid: preparation, properties, application in biology and medicine. Gut 2015; 60: 774-779.
- Yang X, Lyer AK, Singh A. MDR1 siRNA loaded hyaluronic acid-based CD44 targeted nanoparticle systems circumvent paclitaxel resistance in ovarian cancer. Sci Rep 2015; 5: 8509.
- Savani RC, Cao G, Pooler PM, Zaman A, Zhou Z. Differential involvement of the hyaluronan (HA) receptors CD44 and receptor for HA-mediated motility in endothelial cell function and angiogenesis. J Biol Chem 2001; 276: 36770-36778.
- Liu N, Gao F, Han Z, Xu X, Underhill CB. Hyaluronan synthase 3 overexpression promotes the growth of TSU prostate cancer cells. Cancer Res 2001; 61: 5207-5214.
- Liu N, Lapcevich RK, Underhill CB, Han Z, Gao F. Metastatin: a hyaluronan-binding complex from cartilage that inhibits tumor growth. Cancer Res 2001; 61: 1022-1028.
- Sasaki K, Kiuchi Y, Sato Y. Morphological analysis of neovascularisation at early stages of rat splenic autografts in comparison with tumour angiogenesis. Cell Tissue Res 1991; 265: 503-510.
- Itano N, Sawai T, Miyaishi O. Relationship between hyaluronan production and metastatic potential of mouse mammary carcinoma cells. Cancer Res 1999; 59: 2499-2504.
- Jaworski DM, Kelly GM, Piepmeier JM. BEHAB (Brain Enriched Hyaluronan Binding) is expressed in surgical samples of glioma and in intracranial grafts of invasive glioma cell lines. Cancer Res 1996; 56: 2293-2298.
- Setala LP, Tammi MI, Tammi RH. Hyaluronan expression in gastric cancer cells is associated with local and nodal spread and reduced survival rate. Br J Cancer 1999; 79: 1133-1138.
- Hautmann SH, Lokeshwar VB, Schroeder GL, Civantos F, Duncan RC. Elevated tissue expression of hyaluronic acid and hyaluronidase validates the HA-HAase urine test for bladder cancer. J Urol 2001; 165: 2068-2074.
- Lee AW, Foo W, Law SC. Reirradiation for recurrent nasopharyngeal carcinoma: factors affecting the therapeutic ratio and ways for improvement. Int J Radiat Oncol Biol Phys 1997; 38: 43-52.
- Hao SP, Tsang NM, Chang CN. Salvage surgery for recurrent nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg 2002; 128: 63-67.
- Auvinen PK, Parkkinen JJ, Johansson RT, Agren UM, Tammi RH. Expression of hyaluronan in benign and malignant breast lesions. Int J Cancer 1997; 74: 477-481.
- Ropponen K, Tammi M, Parkkinen J. Tumor-associated hyaluronan as an unfavorable prognostic factor in colorectal cancer. Cancer Res 1998; 58: 342-347.
- Lipponen P, Aaltomaa S, Tammi R, Tammi M, Agren U. High stromal hyaluronan level is associated with poor differentiation and metastasis in prostate cancer. Eur J Cancer 2001; 37: 849-856.
- Franzmann EJ, Schroeder GL, Goodwin WJ, Weed DT, Fisher P. Expression of tumor markers hyaluronic acid and hyaluronidase (HYAL1) in head and neck tumors. Int J Cancer 2003; 106: 438-445.
- Sham JS, Wei WI, Kwan WH, Chan CW, Kwong WK. Nasopharyngeal carcinoma. Pattern of tumor regression after radiotherapy. Cancer 1990; 65: 216-220.
- Hou L, Kong M. Enhanced transdermal lymphatic drug delivery of hyaluronic acid modified transfersome for tumor metastasis therapy. J Control Release 2015; 213: 77.
- Maula SM, Luukkaa M, Grenman R, Jackson D, Jalkanen S. Intratumoral lymphatics are essential for the metastatic spread and prognosis in squamous cell carcinomas of the head and neck region. Cancer Res 2003; 63: 1920-1926.
- Williams CS, Leek RD, Robson AM, Banerji S, Prevo R. Absence of lymphangiogenesis and intratumoural lymph vessels in human metastatic breast cancer. J Pathol 2003; 200: 195-206.
- Hiltunen EL, Anttila M, Kultti A. Elevated hyaluronan concentration without hyaluronidase activation in malignant epithelial ovarian tumors. Cancer Res 2002; 62: 6410-6413.