Research Article - Biomedical Research (2017) Volume 28, Issue 1
Bone marrow stromal cells with quercetin show a synergistic effect on permeability reduction of blood-spinal cord barrier in rat model with spinal cord injury
Jing-Xin Nie#, Qing-Tao Meng#, Ren-Bo Li, Guang Yang, Wei Zhou, Hong-De Yu, Bo Chen, Li Jiang, Jing-Bo Shang*Department of Spine Surgery, the Third Peoples’s Hospital of Dalian, Non-Directly Affiliated Hospital of Dalian Medical University, Dalian116033, Liaoning province, PR China
#These authors contributed equally to this work
- *Corresponding Author:
- Jing-Bo Shang
Department of Spine Surgery
The Third Peoples’s Hospital of Dalian
Dalian Medical University
PR China
Accepted date: June 04, 2016
Abstract
The effect of combined treatment of Bone Marrow Stromal Cells (BMSC) along with long-term quercetin pre-treatment was studied. SCI was induced in rats by compression. Animals were randomly divided into five groups including sham group, groups representing individual treatments and group representing combined treatment. The effects of various treatment groups were estimated using the Blood Spinal Cord Barrier (BSCB) permeability test, estimation of TJ-associated proteins and NF-?B, and motor function test. The spinal cord water content and BSCB permeability were significantly reduced by treatment with BMSC as well as quercetin. Their combined treatment showed a synergistic effect. Moreover, the animals also showed marked improvement in the hind-limb motor-function after the combination treatment. TJ-associated proteins showed a significant increase in expression upon combination treatment. This increase was greater than the individual treatments, indicating a synergistic effect. A significant decrease of NF-?B expression and a significant attenuation of its activity was found to be due to the combination treatment. The combination treatment of BMSC and quercetin caused a reduction in the permeability of BSCB by the up-regulation of proteins associated with TJ. The decreased BSCB permeability causes overall improvement in the SCI symptoms including edema and motor function. It was also concluded that the alleviation of SCI symptoms is mediated by the NF-?B pathway.
Keywords
Spinal cord injury, Bone marrow stromal cells, Blood-spinal cord barrier
Introduction
The traumatic symptoms of spinal cord are collectively called as the Spinal Cord Injury (SCI). The SCI is known to happen frequently in the accidental cases worldwide [1]. A majority of the SCI cases are known to affect young people [2]. The elaborate vascular system of the spinal cord is known to form its micro-circulatory system. Similar to the blood-brain barrier (BBB), the existence of a blood-spinal cord barrier (BSCB) has been reported [3]. The disruption of BSCB has been recently reported in SCI. This breakdown of BSCB is known to be associated with the pathology of the spinal cord such as edema and sensory-motor disturbances [4]. Moreover, the restoration of BSCB is known to result into a significant neuro-protection.
The Bone Marrow Stromal Cells (BMSC) transplantation is reported to show significant improvement in the recovery of tissue damage [5]. Further, it has been shown that the transplantations of BMSC also have a significant effect on the attenuation of SCI [6,7]. However, it is still not known whether such improvements are caused due to the reduction in the disruption of BSCB. The treatment with BMSC have been reported to show significant improvements in the decreased BBB leakage in rats [8], attenuation of BSCB permeability rabbits [9], restoration of BBB in stroke rats [10].
Flavonoids are a group of natural substances known for their several anti-inflammatory, anti-oxidative, and neuroprotective properties [11]. Quercetin (3,3′,4′,5,7-Pentahydroxyflavone, MW=338.27) is a flavonoid which is known to be present in several fruits, vegetables and tea [12]. Several pharmacological properties of quercetin have been reported viz., anti-proliferative [13], and anti-oxidative [14] and anti-inflammatory [12] properties. Further, quercetin has also been reported to help in maintaining the integrity of the BBB by scavenging the ROS [15].
The findings of previous studies led us to hypothesize that combining the BMSC therapy with long-term pre-treatment with quercetin may provide prevention and cure for SCI. Further, the combined effect of these two therapies may have an additive effect on the attenuation of SCI. In this work, the role of combining BMSC with quercetin was studied for their effects on BSCB permeability in rat model. Further, we identified whether BMSC and quercetin could exert a cumulative attenuation of SCI.
Materials and Methods
Study animals, establishment of the SCI model and design of experiment
Adult male Sprague-Dawley rats (230 to 260 g) were kept in a 12/12 h dark/light circadian cycle under controlled conditions and fed with the standard laboratory water and diet (ad libitum). The animal experiment was approved by animal ethics committee of the Dalian Medical University. All the animals up keeping procedures were carried out according to World Medical Association (WMA) animal ethics guidelines (Declaration of Helsinki). The animals were anesthetized with a 10% solution of Chloral Hydrate corresponding to a dose of 3.5 ml per Kg. The spinal column of the animals was surgically exposed followed by the laminectomy of T9 level. Dura was left intact during the laminectomy. Subsequently, the T9 of spinal cord was impressed with a weight of 50 g to induce SCI.
Quercetin was procured from Sigma Aldrich (USA). The animals were assigned to five groups (six animals each) at random viz. (1) The SCI group (2) the SCI + BMSC group (3) the SCI + quercetin group (4) the SCI + BMSC + quercetin group (5) Sham group on which anesthesia, skin incision and laminectomy were performed without the dural compression. However in the SCI group, the animals were administered with the respective blank solutions. Whereas in the SCI + BMSC group, the animals were administered with BMSC suspensions (0.75 × 105) around the lesion area of with within 1 hour of SCI in the form of four injections. This led to an overall dose of 3 × 105. In the SCI + quercetin group, Quercetin was administered intragastrically (10 mg/kg/day in 0.3 ml solution by gavage) for 15 days before the induction of SCI. Finally, in the SCI + BMSC + quercetin group, a combination of administration of quercetin for 15 days and injection of BMSC post-SCI was given.
BMSC isolation and culture
Isolation of BMSC was performed from rats aging 4 to 6 weeks (weighing 80 to 100 g). The isolation of BMSC was performed from the bone marrow. Subsequently, the BMSC were cultured following the standard protocol [16]. Briefly, the tibia and femur were isolated, followed by cutting of both ends. The extracted whole bone marrow was suspended in the DMEM (Sigma-Aldrich) along with 10% solution of fetal bovine serum (Sigma-Aldrich). The cells were cultured in 25 cm2 plastic flasks. The cultured were digested and passaged after attainment of 80-90% confluency, with 0.25% trypsin. After expansion for three passages the expanded cultures had a fibroblast-like appearance. These were identified using the CD34 and CD44 antibodies.
Estimation of the permeability of BSCB
The BSCB permeability was estimated using the Evan's blue (EB) extravasation, this test functioned as an indicator of albumin exudation. 1% EB solution in PBS was injected into the femoral vein for 2 hours. Subsequently, 10% chloral hydrate was used to anesthetize the animals. Then, transcardial perfusion of heparin (in PBS) was performed. This perfusion was performed until the detection of fluid in the right atrium. The weighting of tissues was performed after scarifying the animals. The extracted tissues were subsequently stored in formamide solution (10 ml/g) at 60°C temperature for 18 hours. The dye extracted from brain was measured for its concentration spectrophotometrically at 620 nm wavelength. The dye content of spinal cords was estimated using a standard curve of an external standard.
Test of motor function
The hind limb motor function was measured using the scale of Basso, Beattie, and Bresnahan (BBB) [17] for the scoring of hind limb motor function [18]. Briefly, the animals were individually placed in an open field. Thereafter, two observes continuously observed the animal locomotion, independently. This assessment of motor function was performed on a daily basis for 21 days.
Estimation of protein concentrations by western blot densitometry
The spinal cord tissues (20 ml containing 18% dextran) were centrifuged for 15 minutes at 13000 g at 4°C temperature to extract the micro-vessel fractions. These extracted proteins were homogenized in lysis buffer (Pierce, USA). The total protein estimation was performed using Bradford's method and using the Protein Assay Dye (Bio-Rad, USA). 12 μg of the estimated protein was loaded per well on 12% sodium dodecyl sulfate polyacrylamide (SDS-PAGE). Thereafter, the proteins were electrophoretically transferred on a polyvinylidene difluoride (PVDF) membrane after completion of gel run. Blocking of the membranes was performed for 1.5 hours in Tris-buffered saline which contained 0.2% Tween 20 (TBST) and 2% non-fat dry milk (NFDM). Binding of primary antibodies onto the membranes was performed by incubating it with primary antibody solution in TBST and 2% NFDM for 8 hours at 4°C temperature. Then the membrane washing was performed thrice with TBST. Finally, the membranes incubated for 1 hour with Horse Radish Peroxidase (HRP) conjugated secondary antibodies and developed using an ECL detection system. The band density on the membranes was scanned and then analyzed using the ImageJ (NIH, USA) software program. The antibodies for claudin-5 (Cldn-5), occludin (Ocln), ZO-1 (Tjp) and NF-кB-p65 (Santa Cruz Biotechnology, USA) were used for this assay.
Estimation of NF-кB-p65 using ELISA
To determine the effect of BMSC and quercetin on the concentration of NF-кB-p65, Enzyme-linked immunosorbent assay (ELISA) was performed.NF-кB-p65. The ELISA was carried out using the ActivELISA (Imgenex) following the manufacturer's protocol. The estimation of protein concentration was done using a standard curve.
Statistical evaluation
The quantitative results were depicted as means ± SEM. As there were two independent variables (BMSC and quercetin), two-way analysis of variance (ANOVA) was used for the analysis of quantitative results. The statistical analysis for motor-function tests was performed by repeated measures ANOVA. The analysis for multiple tests correction was performed by Bonferroni's method. All the statistical calculations were performed in STATISTICA 8 software program. The maximum threshold for statistically significant P-values was set at 0.05.
Results
Effects of BMSC, quercetin and the combination treatment on the tissue water content
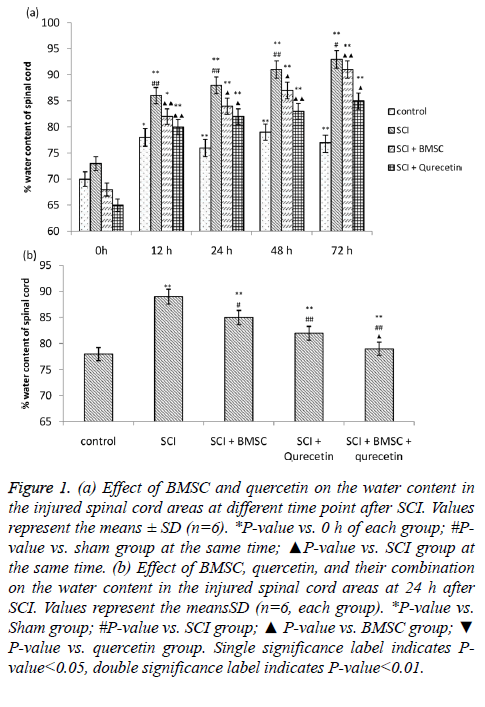
The water content of the sham, SCI, SCI+BMSC and SCI +BMSC+quercetin was found to be significantly increased in the at 12 h, 24 h, 48 h and 72 h as compared to 0 h. Further, the water content of SCI group was found to be significantly larger in comparison to the sham group (Figure 1A). However, the water content showed a marked decrease in the in the BMSC and quercetin groups in comparison to that of sham group. Moreover, the cord water content also showed a marked decrease in BMSC+quercetin group as compared to the individual treatment groups of BMSC and quercetin (Figure 1B).
Figure 1: (a) Effect of BMSC and quercetin on the water content in the injured spinal cord areas at different time point after SCI. Values represent the means ± SD (n=6). *P-value vs. 0 h of each group; #Pvalue vs. sham group at the same time; ▲P-value vs. SCI group at the same time. (b) Effect of BMSC, quercetin, and their combination on the water content in the injured spinal cord areas at 24 h after SCI. Values represent the meansSD (n=6, each group). *P-value vs. Sham group; #P-value vs. SCI group; ▲ P-value vs. BMSC group; ▼ P-value vs. quercetin group. Single significance label indicates P-value< 0.05, double significance label indicates P-value<0.01.
Effect of BMSC, quercetin and the combination treatment on the permeability of BSCP
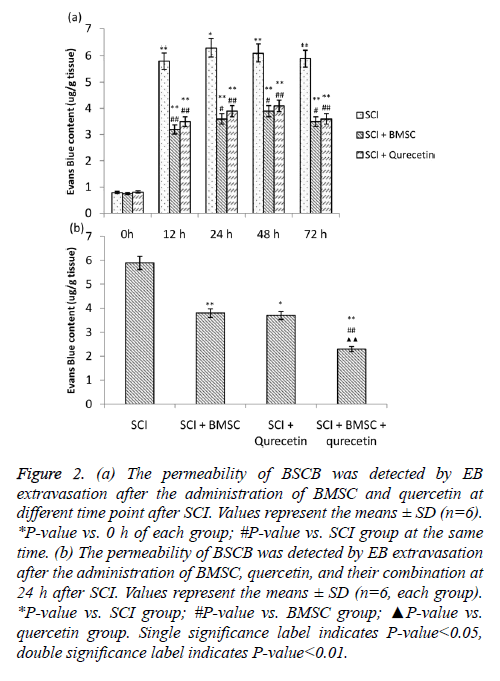
The extravasation of EB was used as a marker for BSCP permeability. The EB quantity showed a significant increase in induction of SCI at all time points studied as compared to the 0h measurement (Figure 2A). The EB quantity peaked at 24 h time point in the SCI group. The EB extravasation was significantly decreased in both the individual treatment groups and at all time points in both the individual treatment groups. As the highest amount of EB was detected at the 24 h time point, it was considered as a base for estimation of the attenuation of BSCB in the combination treatment. The combination treatment group showed a marked reduction in the EB activity in comparison to the BMSC and quercitin treatment groups individually (Figure 2B).
Figure 2: (a) The permeability of BSCB was detected by EB extravasation after the administration of BMSC and quercetin at different time point after SCI. Values represent the means ± SD (n=6). *P-value vs. 0 h of each group; #P-value vs. SCI group at the same time. (b) The permeability of BSCB was detected by EB extravasation after the administration of BMSC, quercetin, and their combination at 24 h after SCI. Values represent the means ± SD (n=6, each group). *P-value vs. SCI group; #P-value vs. BMSC group; ▲P-value vs. quercetin group. Single significance label indicates P-value<0.05, double significance label indicates P-value<0.01.
Effects of BMSC, quercetin and combination treatment on the motor function of SCI rats
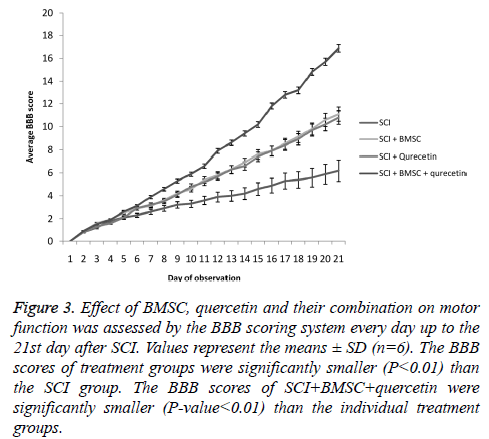
A significant disruption of the hind limb motor function was observed in the SCI animals. However, the motor function was significantly improved in the BMSC and quercetin treatment groups (Figure 3). Moreover, the combination therapy of BMSC and quercetin showed an even greater improvement in the motor function.
Figure 3: Effect of BMSC, quercetin and their combination on motor function was assessed by the BBB scoring system every day up to the 21st day after SCI. Values represent the means ± SD (n=6). The BBB scores of treatment groups were significantly smaller (P<0.01) than the SCI group. The BBB scores of SCI+BMSC+quercetin were significantly smaller (P-value<0.01) than the individual treatment groups.
Effects of BMSC, quercetin and combination treatment on the activity on TJ-associated proteins
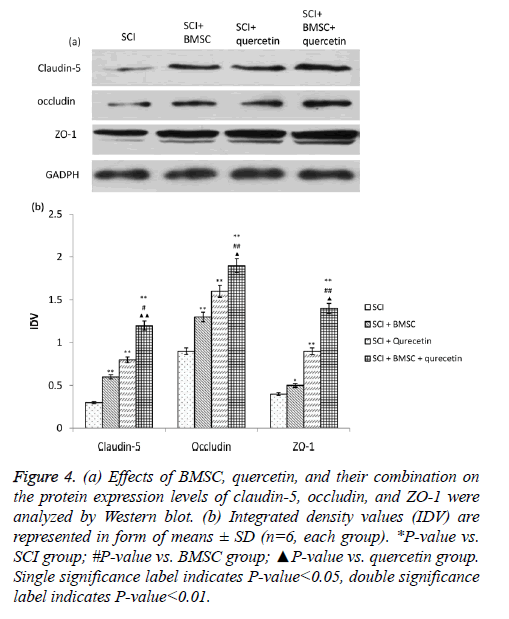
The activity of Cldn5, Ocln and Tjp1 showed a significant increase upon treatment with BMSC and quercetin individually in comparison to the SCI group (Figure 4). Moreover, the combination of both therapies showed a synergistic effect, where the activity of the three proteins showed an even greater and significant increase as compared to the individual treatment groups.
Figure 4: (a) Effects of BMSC, quercetin, and their combination on the protein expression levels of claudin-5, occludin, and ZO-1 were analyzed by Western blot. (b) Integrated density values (IDV) are represented in form of means ± SD (n=6, each group). *P-value vs. SCI group; #P-value vs. BMSC group; ▲P-value vs. quercetin group. Single significance label indicates P-value<0.05, double significance label indicates P-value<0.01.
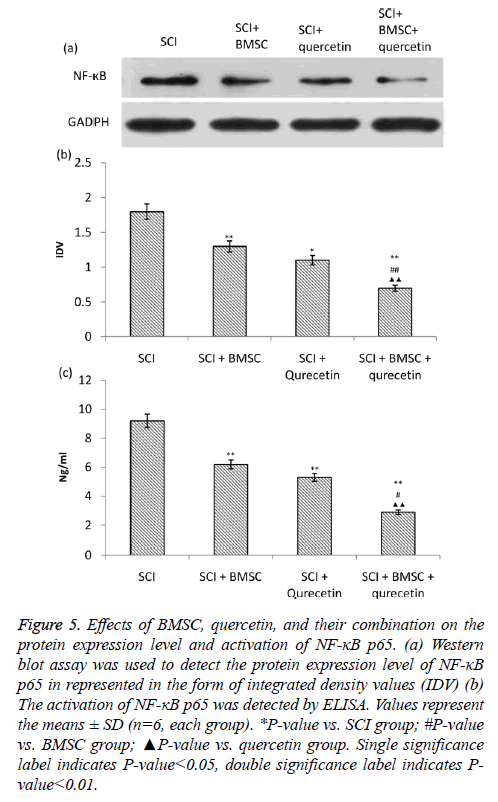
Effects of BMSC, quercetin and combination treatment on NF-кB-p65 activity and expression NF- кB-p65
The NF-кB-p65 expression NF-кB-p65was estimated using immunoblotting followed by its densitometry. Whereas, ELISA was employed for the estimation of NF-кB-p65 activation. The SCI group showed highest NF-кB-p65 expression and activity (Figure 5). However, the BMSC and quercetin treatment caused significant attenuation of NF-кB-p65 as compared to SCI group. Moreover, the combination of the two treatment groups i.e. BMSC+quercetin caused even greater attenuation of NF-кB-p65 activity as compared to when these treatments were given individually.
Figure 5: Effects of BMSC, quercetin, and their combination on the protein expression level and activation of NF-κB p65. (a) Western blot assay was used to detect the protein expression level of NF-κB p65 in represented in the form of integrated density values (IDV) (b) The activation of NF-кB p65 was detected by ELISA. Values represent the means ± SD (n=6, each group). *P-value vs. SCI group; #P-value vs. BMSC group; ▲P-value vs. quercetin group. Single significance label indicates P-value<0.05, double significance label indicates Pvalue< 0.01.
Discussion
We identified the attenuation ability of BMSC and the flavonoid, quercetin. We also identified the combination therapy of the two treatments and compared with the individual treatments. The water content and the extravasation of EB were significantly decreased upon treatment with BMSC and quercetin. This reduction was found to be even greater when BMSC treatment was combined with long term quercetin pre-treatment. The Tight Junction (TJ) associated proteins were evaluated for their expression. The expression of Cldn5, Ocln and Tjp1 was markedly increased upon treatment with BMSC and quercetin. Moreover, the combination therapy showed even greater increased in the TJ-associated proteins' expression. These results indicated that BMSC and quercetin can decrease the permeability of BSCB. The results also clearly indicated a synergistic effect of combining the treatment of BMSC with long term quercetin pre-treatment. These results were corroborated by the hind-limb motor function test where the individual treatments, as well as the combination treatment, showed marked improvement.
The normal function of the spinal cord is maintained by its microenvironment. SCI is known to cause disruption of BSCB and vasculature damage [3]. This damage to BSCB allows inflammatory immune cells to enter the spinal cord and cause an inflammatory response and damage to nerve cells. This causes swelling of the spinal cord [2]. Improvement in BSCB-pathophysiology is known to decrease the inflammatory damage to the nerve cells due to SCI. Moreover, the recovery of motor function has also been associated with improvement in BSCB [4]. Although, decrease in permeability of BSCB due to BMSC treatment has been shown in rabbits [9]. But, this effect of BMSC in SCI rats remains elusive. The SCI includes two stages of injury, first; the mechanical injury, second; the pathological changes caused due to the oxidative stress [19]. Quercetin is known for its anti-inflammatory and anti oxidative properties [15,20]. Moreover, quercetin has been reported to possess neuroprotective properties in ischemic/reperfusion (I/R) injury [20]. This study showed that quercetin improved the water content in spinal cord and BSCB permeability. The synergistic effect of quercetin with BMSC showed marked improvement in all the parameters of SCI.
Like BBB, BSCB provides optimal conditions for the spinal cord cells. The opening of TJ-associated proteins and elevated vascular transport associated with the capillary endothelium are primary markers of the disruption of BSCB [3]. Transmembrane proteins such as cludin-5, occludin and ZO-1contribute a primary role in the maintenance of tight junctions [21]. Therefore, low expression of proteins associated with has a major role in the disruption of BSCB [22]. Therefore, the low expression of Cldn5, Ocln and Tjp1 in SCI animals was as expected. The combination of BMSC and quercetin showed marked up-regulation of Cldn5, Ocln and Tjp1. Moreover, the closure of TJ due to high expression of these proteins was further corroborated by the marked reduction of EB extravasation.
The transcription factor NF-κB is closely associated with the nervous system and known to have diverse functions [23]. Further, NF-κB is known to be involved in the regulation of the permeability of BBB and BSCB [24]. Moreover, the heightened expression and permeability of NF-κB is known to be closely associated with the permeability of BBB and BSCB [25,26]. This mediation of permeability is primarily caused by regulating the expression of TJ-associated proteins by NF-κB [27,28]. Moreover, BMSC the inhibition of the pathway of NF- κB [29] is well known. In addition to BMSC, Quercetin is also known to inhibit the activation of NF-κB in several studies [30,31]. The over expression of Cldn5, Ocln and Tjp1 (TJ proteins) and attenuation of NF-κB p65 corroborated our hypothesis that BMSC and quercetin can have a positive effect on the permeability of BSCB in rats by inhibiting NF-κB pathway.
Conclusion
The Plant flavonoid quercetin could function as a therapeutic agent for SCI. The combination of long-term pre-treatment with quercetin along with BMSC treatment post-SCI can prove to be a potentially effective treatment regimen against SCI. This treatment can not only improve the physiological symptoms of SCI such as edema but also can show marked improvement in the loss of motor function caused by SCI. However, here we would like to emphasise that because SCI are caused by accidents and thus cannot be predicted beforehand. Therefore, it would be difficult to implement a preventive treatment for SCI.
Acknowledgement
This work was supported by grants from the Research Project of Dalian Medical University (Liaoning province, China).
References
- Ryan TZ, Sharma A, Nozari A, Subramaniam R, Lundstedt T, Shanker Sharma H. Nanowired drug delivery to enhance neuroprotection in spinal cord injury. CNS Neurol Disord Drug Targets 2012; 11: 86-95.
- Kumaran MP, Fior MD, Sharma A, Mossler H, Shanker SH. Cerebrolysin, a mixture of neurotrophic factors induces marked neuroprotection in spinal cord injury following intoxication of engineered nanoparticles from metals. CNS Neurol Disord Drug Targets 2012; 11: 40-49.
- Bartanusz V, Jezova D, Alajajian B, Digicaylioglu M. The blood-spinal cord barrier: morphology and clinical implications. Annal Neurol 2011; 70: 194-206.
- Sharma HS. Early microvascular reactions and blood-spinal cord barrier disruption are instrumental in pathophysiology of spinal cord injury and repair: novel therapeutic strategies including nanowired drug delivery to enhance neuroprotection. J Neural Trans 2011; 118: 155-176.
- Kitada M. Mesenchymal cell populations: development of the induction systems for Schwann cells and neuronal cells and finding the unique stem cell population. Anatomi Sci Int 2012; 87: 24-44.
- Dasari VR, Veeravalli KK, Dinh DH. Mesenchymal stem cells in the treatment of spinal cord injuries: a review. World J Stem Cells 2014; 6: 120-133.
- Mitsuhara T, Takeda M, Yamaguchi S, Manabe T, Matsumoto M, Kawahara Y, Yuge L, Kurisu K. Simulated microgravity facilitates cell migration and neuroprotection after bone marrow stromal cell transplantation in spinal cord injury. Stem Cell Res Ther 2013; 4: 35.
- Zacharek A, Chen J, Cui X, Li A, Li Y, Roberts C, Feng Y, Gao Q, Chopp M. Angiopoietin1/Tie2 and VEGF/Flk1 induced by MSC treatment amplifies angiogenesis and vascular stabilization after stroke. J Cereb Blood Flow Metabol 2007; 27: 1684-1691.
- Fang B, Wang H, Sun X-J, Li X-Q, Ai C-Y, Tan W-F, White PF, Ma H. Intrathecal transplantation of bone marrow stromal cells attenuates blood-spinal cord barrier disruption induced by spinal cord ischemia-reperfusion injury in rabbits. J Vascular Surgery 2013; 58: 1043-1052.
- Borlongan CV, Lind JG, Dillon-Carter O, Yu G, Hadman M, Cheng C, Carroll J, Hess DC. Bone marrow grafts restore cerebral blood flow and blood brain barrier in stroke rats. Brain research 2004; 1010: 108-116.
- Nijveldt RJ, Van Nood E, Van Hoorn DE, Boelens PG, Van Norren K, Van Leeuwen PA. Flavonoids: a review of probable mechanisms of action and potential applications. Am Journal Clin Nutri 2001; 74: 418-425.
- Boots AW, Haenen GR, Bast A. Health effects of quercetin: from antioxidant to nutraceutical. European J Pharmacol 2008; 585: 325-337.
- Borska S, Gebarowska E, Wysocka T, Drag-Zalesinska M, Zabel M. The effects of quercetin vs cisplatin on proliferation and the apoptotic process in A549 and SW1271 cell lines in in vitro conditions. FOLIA MORPHOLOGICA-WARSZAWA-ENGLISH EDITION 2004; 63: 103-106.
- Saito A, Sugisawa A, Umegaki K, Sunagawa H. Protective effects of quercetin and its metabolites on H2O2-induced chromosomal damage to WIL2-NS cells. Biosci Biotechnol Biochem 2004; 68: 271-276.
- Lapi D, Vagnani S, Pignataro G, Esposito E, Paterni M, Colantuoni A. Protective effects of quercetin on rat pial microvascular changes during transient bilateral common carotid artery occlusion and reperfusion. Front Physiol 2012; 3: 10.3389.
- Tohill M, Mantovani C, Wiberg M, Terenghi G. Rat bone marrow mesenchymal stem cells express glial markers and stimulate nerve regeneration. Neurosci Lett 2004; 362: 200-203.
- Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 1995; 12: 1-21.
- Joshi M, Fehlings MG. Development and characterization of a novel, graded model of clip compressive spinal cord injury in the mouse: Part 1. Clip design, behavioral outcomes, and histopathology. J Neurotrauma 2002; 19: 175-190.
- Beattie MS. Inflammation and apoptosis: linked therapeutic targets in spinal cord injury. Trend Mol Med 2004; 10: 580-583.
- Lei X, Chao H, Zhang Z, Lv J, Li S, Wei H, Xue R, Li FLi Z. Neuroprotective effects of quercetin in a mouse model of brain ischemic/reperfusion injury via antiapoptotic mechanisms based on the Akt pathway. Mol Med Report 2015; 12: 3688-3696.
- >Luissint A-C, Artus C, Glacial F, Ganeshamoorthy K, Couraud PO. Tight junctions at the blood brain barrier: physiological architecture and disease-associated dysregulation. Fluid Barrier CNS 2012; 9: 1-12.
- Liebner S, Czupalla CJ, Wolburg H. Current concepts of blood-brain barrier development. Int J Development Biol 2011; 55: 467.
- Kaltschmidt B, Kaltschmidt C. NF-kappaB in the nervous system. Cold Spring Harb Perspect Biol 2009; 1: a001271.
- Coelho-Santos V, Leitão RA, Cardoso FL, Palmela I, Rito M, Barbosa M, Brito MA, Fontes-Ribeiro CA, Silva AP. The TNF-α/NF-κB signaling pathway has a key role in methamphetamine-induced blood-brain barrier dysfunction. J Cerebral Blood Flow Metabol 2015; 35: 1260-1271.
- Lee JY, Choi HY, Ahn H-J, Ju BG, Yune TY. Matrix Metalloproteinase-3 Promotes Early Blood-Spinal Cord Barrier Disruption and Hemorrhage and Impairs Long-Term Neurological Recovery after Spinal Cord Injury. Am J Pathol 2014; 184: 2985-3000.
- Li X-Q, Wang J, Fang B, Tan W-F, Ma H. Intrathecal antagonism of microglial TLR 4 reduces inflammatory damage to blood-spinal cord barrier following ischemia/reperfusion injury in rats. Mol Brain 2014; 7: 1.
- He F. Mechanisms of tumor necrosis factor-alpha-induced leaks in intestine epithelial barrier. Cytokine 2012; 59: 264-272.
- Burek M, Förster CY. Cloning and characterization of the murine claudin-5 promoter. Mol Cell Endocrinol 2009; 298: 19-24.
- Cao C. Bone marrow mesenchymal stem cells slow intervertebral disc degeneration through the NF-κB pathway. Spine J 2015; 15: 530-538.
- Shi Y, Liang X-c, Zhang H, Wu Q, Qu L, Sun Q. Quercetin protects rat dorsal root ganglion neurons against high glucose-induced injury in vitro through Nrf-2/HO-1 activation and NF-κB inhibition. Acta Pharmacologica Sinica 2013; 34: 1140-1148.
- Wang C-P, Li J-L, Zhang L-Z, Zhang X-C, Yu S, Liang X-M, Ding F, Wang Z-W. Isoquercetin protects cortical neurons from oxygen-glucose deprivation-reperfusion induced injury via suppression of TLR4-NF-кB signal pathway. Neurochem Int 2013; 63: 741-749.