- Biomedical Research (2011) Volume 22, Issue 1
Biochemical markers in oral cancer
Charushila Y. Kadam*, Raghavendra V. Katkam, Adinath N. Suryakar, Kashinath M. Kumbar, Dipali P. Kadam.Department of Biochemistry, Dr. V. M. Govt. Medical College, Solapur (Maharashtra), India
- Corresponding Author:
- Charushila Y. Kadam
Department of Biochemistry
Dr. V. M. Govt. Medical College
Solapur 413003 (Maharashtra)
India
Accepted Date: july 21 2010
Abstract
β2 -Microglobulin is the invariant chain of the Major Histocompatibility Class I (MHC-I) molecule on the cell surface of all nucleated cells and it reflects the cell turn over. Altered glycosylation of glycoconjugates is among the important molecular changes that accompany malignant transformation. The objectives of this study were to validate the diagnostic value of serum β2–microglobulin, Total Sialic Acid (TSA) and Lipid-Bound Sialic Acid (LSAB) and to correlate these parameters with the stages of the malignancy. Clinically and histopathologically proven 40 oral cancer patients were selected for the study. According to TNM stage of cancer, patients studied 11, 8, 10, 11 were of stages I, II, III and IV respectively. 40 healthy controls were selected for comparison. β2 -Microglobulin was estimated by ELISA and total sialic acid and lipid-bound sialic acid determined by the spectrophotometric method of Plucinsky et al. The concentration of β2-microglobulin, total sialic acid and lipid-bound sialic acid were significantly elevated in different stages of oral cancer patients when compared with the controls. Circulating levels of these biomarkers were also elevated significantly among different clinical stages with progressive rise from stage I to stage IV of the disease and found to reflect tumor burden. These biomarkers have good sensitivity, specificity and efficiency values for oral cancer. Thus, findings of the study suggest that the evaluation of these markers would be useful in assessing early malignant change, increasing accuracy of clinical diagnosis and also in assessing the spread and invasiveness of the cancer of the oral cavity.
Keywords
Oral cancer, β2-microglobulin, Total sialic acid, Lipid- bound sialic acid, Biomarkers.
Introduction
Malignant neoplasms are major causes of fear, morbidity and mortality all over the world. Globally ‘oral cancer’ is the sixth most common cause of cancer-related death [1]. Oral cancer accounts for approximately 30-40% of all cancers in India [2]. Despite the recent advances in tumor surgery and multimodal treatment regimes, the prognosis of oral squamous cell carcinoma is still relatively poor. This may be because symptoms that indicate the presence of the carcinoma often appear when the tumor is in an advanced stage [3].
In the light of such problems, it would be very useful to find biochemical markers that allow to suspect the pres-ence of the carcinoma at early stages. During the course of tumor development, quantitative changes have been shown to occur in a variety of substances in serum. These substances are collectively referred to as biochemical markers or tumor markers [3,4].
β2-Microglobulin is a non-glycosylated peptide and is the invariant chain of the Major Histocompatibility Class (MHC) I molecules on the cell surface of all nucleated cells [2,5]. Its best characterized function is to interact with and stabilize the tertiary structure of the MHC class I α-chain. Because it is non-covalently associated with the α-chain and has no direct attachment to the cell membrane, β2-microglobulin on the cell surface can exchange with free β2-microglobulin present in serum containing medium. Free β2-microglobulin is found in body fluids under physiologic and pathological conditions as a result of shedding from cell surfaces or intracellular release [6]. Many properties of mammalian cells are expressed at, or mediated through the cell surface [7,8]. Immense increase in knowledge of the altered characteristics of malignant cells has shown that altered cell surface is the hallmark of malignant cells [9]. Glycoproteins and glycolipids are major constituents of cell membrane [10]. Altered glycosylation of glycolcon-jugates is one of the important molecular changes that accompany malignant transformation [9,11]. Studies of malignant cells have revealed alterations in cell surfaces and membranes in terms of sialic acid content of glyco-proteins and glycolipids. Sialic acid, a family of acety-lated derivatives of neuraminic acid is widely distribu ted in mammals [12]. They are the end moieties of the carbo-hydrate chains and are biologically important and essen-tial for functions of glycoconjugates [11,12]. Sialic acid is thought to be important in determining the surface properties of cells and has been implicated in cellular in-vasiveness, adhesiveness, and immunogenicity. They are released into circulation through increased turnover, se-cretion, and/or shedding from malignant cells [7,13]. In-creased levels of β2-microglobulin and glycoconjugates like total sialic acid and lipid-bound sialic acid have been reported in serum of patients of oral cancer [2,5,6,7,10,14,15,16].
In oral cancer the study of tumor markers have been limited. Several tumor makers with clinical promise need further evaluation [2,5]. Three such tumor markers are β2-microglobulin, total sialic acid and lipid-bound sialic acid.
Considering the high prevalence of oral malignancy, the present study was undertaken to validate diagnostic value of serum β2-microglobulin, total sialic acid and lipid-bound sialic acid and to correlate these parameters with the stages of malignancy.
Material and Methods
The study comprised of 80 subjects. 40 oral cancer patients admitted in the Shri Chhatrapati Shivaji Maharaj General Hospital and Shree Siddheshwar Cancer Hospital and Research Centre, Solapur (Maharashtra) proved by the clinical and histopathological evidence were selected for the study. According to TNM staging system of the UICC [17], out of 40 oral cancer patients studied, 11, 8, 10 and 11 were of stages I, II, III and IV respectively. 40 healthy controls were selected for comparison.
To avoid false positive results, care was taken to exclude subjects with renal, hepatobiliary disorders, lupus erythe- matosus, lymphoproliferative disorders, and cardio-vascular disorders as well as other malignancies [5].
Under aseptic precautions venous blood was drawn and serum was separated.
β2-microglobulin assay:
The serum was analyzed for β2-microglobulin by Enzyme linked immunosorbent assay (ELISA) (β2-microglobulin EIA Kit, Orgentec, Germany).
Measurement of Lipid-bound sialic acid (LBSA) Serum lipid-bound sialic acid was determined by the Spectropho-tometric method of Plucinsky et al. [18]. Briefly, 50 μl of serum were mixed with 150μl of distilled water and ex-tracted with 3ml cold chloroform / methanol (2:1) (v/v). The lipid extract was partitioned with 0.5ml cold distilled water. After centrifugation 1ml of aqueous layer containing the sialolipid fraction was precipitated with 50 μl phosphotungstic acid solution (1gm/ml). After cen-trifugation supernatant fluid was discarded and the pre-cipitate was resuspended in 1ml of distilled wa-ter. 1ml of resorcinol reagent was added and the tubes were boiled for 15 min and then cooled in ice bath for 10 min. The blue chromophore was extracted with 2ml butyl acetate n-butanol (85:15) (v/v) and determined spectro-pho tometrically at 580 nm. The sialic acid concentra-tion was calculated by the use of a standard curve of N-acetyl neuraminic acid (NANA).
Measurement of Total sialic acid (TSA) Serum total sialic acid level was determined by the method of Plucinsky et al. [18]. 20μl of serum was diluted with 980 μl distilled water. After treatment with resorcinol reagent the blue chromophore was extracted by butyl acetate / n-butanol (85:15) (v/v) and determined spectrophotometrically at 580nm and sialic acid was determined by the use of standard curve of N-acetyl neuraminic acid.
Statistical analysis
The data is expressed as mean ± SD. The statistical significance of the results was analyzed using a non-parametric Tukey’s test (for unequal sample size). Values of P<0.001 were considered significant.
Results
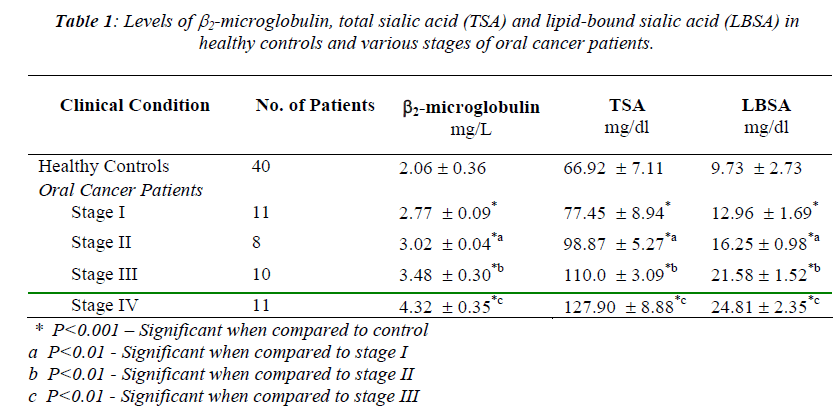
The mean values of serum β2-microglobulin, total sialic acid and lipid-bound sialic acid in various stages of oral carcinoma patients and healthy controls are given in Table 1. The levels of serum β2-microglobulin, total sialic acid and lipid-bound sialic acid elevated significantly and progressively in various stages from stage I to stage IV of oral cancer patients compared with healthy controls (P<0.001) (Table 1). The average β2-microglobulin, TSA and LBSA levels in stage II was significantly higher than that in stage I (P<0.01). Also, the average levels of these biomarkers in stage III was significantly higher than that in stage II (P<0.01) as well as stage IV patients with metastasis showed a significant rise (P<0.01) when compared with stage III patients (Table 1). The positive correlation was found between the circulating levels of lipid-bound sialic acid and total sialic acid in different stages of oral cancer patients (stage I r=0.97, stage II r=0.84, stage III r=0.82, stage IV r=0.78).
Out of 40 oral cancer patients only 33 had elevated levels of lipid-bound sialic acid and thus sensitivity of 82.5%. In case of total sialic acid and β2-microglobulin the sensitivity was 90% and 100% respectively. The test showed an abnormal result in only 1 out of 40 healthy controls for LBSA and thus the specificity was of 97.5% and only 5 out of 40 healthy controls for TSA had abnormal results and thus the specificity was of 87.5%. In case of β2-microglobulin the specificity was of 100%. For LBSA the predictive value of a positive test was 97%, predictive value of negative test was 84% and efficiency of test was 90%. For TSA, the predictive value of positive test was 87%, predictive value of negative test was 89% and efficiency of test was 88.7%. In case of β2-microglobulin predictive value of positive test was 100%, predictive value of negative test was 100% and efficiency of the test was 100%.
Discussion
The β2-microglobulin which is synthesized and secreted by lymphocytes as well as most other nucleated cells is an intrinsic part of histocompatibility antigen. It has a low molecular weight and rapid turnover [5]. In the present study the β2-microglobulin values were increased in different stages of oral cancer, when compared with healthy controls, and this is statistically significant (P<0.001) (Table 1). Also there was observed significant increase (P<0.01) among different clinical stages. The increased levels were correlated well with tumor burden. The increased levels of β2-microglobulin might be due to increased production or impaired excret- ion. However, as the patients in this present study did not have any disorder of renal function or other systemic ail- ments and other malignancies, the increase in serum β2-microglobulin appears to be a true phenomenon due to the malignant process involving oral cancer. The fact that β2-microglobulin levels were elevated in the serum of sub-jects with oral carcinoma is in agreement with previous study reports [2,5,6,14].
The mechanism of increase in β2-microglobulin levels in malignancies is not known but various possible hypotheses for the increased serum levels have been put forward. The β2-microglobulin is a cell membrane constituent along with the HLA –chain (Human leukocyte antigen), so an accelerated membrane turnover or accelerated cell division could increase the shedding of β2-microglobulin [2,5]. The ability of the carcinoma cells to produce a higher concentration of β2-microglobulin than the non-neoplastic cells may be due to either active synthesis or increased cell breakdown or both [2,5]. In many neoplasms, especially those of epithelial origin, a decrease or lack of expression of HLA-I particles is found. Despite weakened expression of HLA-I complex on the neoplastic cells, the level of β2-microglobulin in the blood serum could be increased. Recent studies explained this phenomenon as due to an imbalance of light chain and heavy chain of HLA-I complex and relative over -production of β2-microglobulin [6]. Most frequently quoted hypothesis for high levels of β2-microglobulin in neoplastic diseases explains this phenomenon with mono or polyclonal activation of lymphocytes, destruction of MHC I particles, and increased cellular transformation into neoplastic cells which could lead to higher concentration of protein β2-microglobulin. Certain studies proposed that the systemic immunosuppression observed in oral cancer patients is due to the decreased functional activity of peripheral blood monocytes which is reflected by way of decreased phagocytic process [19]. The latest studies showed that a high concentration of free β2-microglobulin may have a negative influence on the immunological system by decreasing the expression of MHC-I (Major histocompatibility complex) particles and indirectly by increasing the levels of cytokines: IL-6, IL-10, which accelerate the development of neoplasms [6,20].
Sialic acids are major constituent of the carbohydrate chains of cell membrane glycoproteins and glycolipids. Sialic acid concentrations of the tumor cell surfaces were shown to be related to malignant potential and changes in immunogenicity. The carbohydrate moi-ety may influence differentiation, growth and cell-to-cell interactions, and thus may be important in malignant transformation [7,13]. The serum total sialic acid and lipid-bound sialic acid levels were increased in oral cancer patients when compared with healthy controls and this is statistically significant (P<0.001). Also there was observed significant increase (P<0.01) among different stages of oral cancer patients. The increased concentration of total sialic acid and lipid- bound sialic acid were found to correlate with the presence and extent of malignant disease. Our results correlated well with those of others regarding alteration in sialic acid content in oral cancer [3,7,10,15,16].
Malignant transformation of oral epithelium is associated with atypical glycosylation of cell surface carbohydrates [13]. Neoplasms often have increased concentrations of sialic acid on tumor cell surface and sialoglycoproteins are shed or secreted by these cells increasing their concentration in blood [13]. The elevations found in sialic acid levels in cancer patients might also be due to selective increase in existing specific sialylated sequence or a tumor associated de novo synthesis of specific sialylated sequence [9]. A good correlation was found between circulating levels of TSA and LBSA in different stages of oral cancer patients suggesting that tissue turnover rates of both membrane glycolipid and glycoprotein were to the extent of malignant transformation. Similar findings have been reported by Rao et al. [7].
The results also revealed that the levels of β2-microglobulin TSA and LBSA elevated significantly in Stage I of oral cancer patients and thus can give early indication of malignant change and therefore malignancy can be detected at an early and treatable stage. Furthermore, because there is progressive rise in levels of these biomarkers, even the extent of malignant disease can be ascertained. In order to assess the reliability of tumor markers, we compared the sensitivity, specificity, predictive value of positive test, predictive value of negative test and efficiency of these three biomarkers. These results suggest β2-microglobulin to be a very accurate tumor marker. However it lacks specificity for oral carcinoma as an individual marker because it is elevated in other diseases also [5]. Regarding the cost of the measurements, the total sialic acid was the cheapest among the three tumor markers. All the three biomarkers tested showed good sensitivity values, specificity values, predictive values of positive test, predictive values of negative test and efficiency values in oral carcinoma.
Thus, our results suggest that the concentrations of β2-microglobulin, total sialic acid and lipid-bound sialic acid significantly increase in different stages of oral cancer patients and correlates well with the progression of clinical symptoms. These biomarkers have good sensitivity, specificity and efficiency for oral cancer. Therefore, the evaluation of these markers, with TSA being the cheapest among three in terms of cost and effectiveness, would be of immense help in assessing early malignant change, in increasing the accuracy of clinical diagnosis and also in assessing the spread and invasiveness of oral squamous cell carcinoma.
References
- Rizzolo D, Hanifin C, Chiodo TA. Oral cancer: How to find this hidden killer in two minutes. JAAPA 2007; 20(10): 42-47.
- Manzar W, Raghvan MRV, Aroor AR, et al. Evaluation of serum Ã2-microglobulin in oral cancer. Aust Dent J 1992; 37(1): 39-42.
- Ayude D, Gacio G, Cadena MP, et al. Combined use of established and novel tumor markers in the diagnosis of head and neck squamous cell carcinoma. Oncol Rep 2003; 10: 1345-1350.
- Lal H. Biochemical studies in head and neck cancer. Clin Biochem 1994; 27(4): 235-43.
- Silvia CRWD, Vasudevan DM, Prabhu KS. Alteration of serum Ã2-microglobulin in oral carcinoma. Indian J Clin Biochem 2002; 17 (2): 104-7.
- Petkowicz B, Miszczuk JW, Wojtak M, et al. The diagnostic utility of the assay of Ã2-microglobulin in the precancerous lesions and oral cancers. Ann Univ Mariae Curie Sklodowska 2006 ; XIX (1) : 243-247.
- Rao VR, Krishnamoorthy L, Kumarswamy SV, et al. Circulating levels in serum of total sialic acid, lipid associated sialic acid and fucose in precancerous lesion and cancer of the oral cavity. Cancer Detect Prev 1998; 22(3): 237-240.
- Baxi BR, Patel PS, Adhvaryu SG, et al. Usefulness of serum glycoconjugates in precancerous and cancerous diseases of the oral cavity. Cancer 1991; 67: 135-140.
- Raval GN, Parekh LH, Patel DD, et al. Clinical usefulness of alterations in sialic acid, sialyltransfe- rase and sialoproteins in breast cancer. Indian J Clin Biochem 2004; 19(2): 60-71.
- Chitra CS,. Devi S. Effect of vitamin E on protein bound carbohydrate complexes in radiation treated oral squamous cell carcinoma patients. Indian J Clin Biochem 2008; 23(1): 92-94.
- Patel PS, Raval GN, Rawal RM, et al. Importance of glycoproteins in human cancer. Indian J Biochem Biophys 1997; 34: 226-233.
- Akcay F, Taysi S, Uslu C, et al. Levels of soluble intercellular adhesion molecule-1 and total sialic acid in serum of patients with laryngeal cancer. Jpn J Clin Oncol 2001; 31 (12): 584-588.
- Manoharan S, Padmanabhan M, Kolanjiappan K, et al. Analysis of glycoconjugates in patients with oral squamous cell carcinoma. Clin Chim Acta 2004; 339: 91-96.
- Vaishali N, Tupkari JV. An estimation of serum Ã2-microglobulin level in premalignant lesions / conditions and oral squamous cell carcinoma: A clinicopathological study. J Oral Maxillofac Pathol 2005; 9 (1): 16-19.
- Silvia CRWD, Vasudevan DM, Prabhu KS. Evalua-tion of serum glycoproteins in oral carcinoma. Indian J Clin Biochem 2001; 16 (1): 113-115.
- Kimura Y, Fujieda S, Bayashi TT, et al. Conventional tumor markers are prognostic indicators in patients with head and neck squamous cell carcinoma. Cancer lett 2000; 155: 163-168.
- Neville BW, Day TA. Oral cancer and precancerous lesions. CA Cancer J Clin 2002; 52:195-215.
- Plucinsky MC, Riley WM, Prorok JJ, et al. Total and lipid associated serum sialic acid levels in cancer patients with different primary sites and differing degrees of metastatic involvement. Cancer 1986; 58: 2680-2685.
- Reshma K, Bharathi B, Rao AV et al. Phagocytosis: A marker of decreased immune response in radiation treated oral cancers. Biomed Res 2009; 20(1): 75-77.
- Xie J, Wang Y, Muta E, et al. Ã2-microglobulin as a negative regulator of the immune system: high concentration of the protein inhibits in vitro generation of functional dendritic cells. Blood 2003; 101: 4005-4012.