Research Article - Biomedical Research (2017) Volume 28, Issue 5
Aqueous extract from Radix Astragali ameliorates osteoporosis subsequent to spinal cord injury
Yaonan Zhang1, Yunru Li2, Chi Liu1, Xuesong Gao2, Lin Wang1, Xuefei Duan2*
1Department of Orthopaedic, Beijing Hospital, National Center of Gerontology, No.1 Dahua Road, Dong Dan, Beijing, PR China
2General Medicine Department, Beijing Ditan Hospital, Capital Medical University, Beijing, PR China
- *Corresponding Author:
- Xuefei Duan
General Medicine Department
Beijing Ditan Hospital Capital Medical
University Beijing, PR China
Accepted on October 06, 2016
Abstract
Introduction: Radix Astragali (RA) has been used for herbal formulations in the treatment of bone degradation in traditional Chinese medicine.
Methods: The effects of aqueous extract from Radix Astragali (RA) on bone loss in spinal cord-injured rats were investigated in the present study. Forty male Wistar rats were divided into four groups: control group (rats with sham operation); osteoporosis group (Op group, rats with spinal cord sectioned); L group (spinal cord-injured rats treated with low-dose extract); and H group (spinal cordinjured rats treated with high-dose extract).
Results: Op group showed a significant decrease in bone mineral density and biomechanical properties versus control group. While RA extract could significantly reverse this bone degeneration trend, and the effects of high dose was better than low dose, which means such effects maybe dose dependent.
Conclusion: We conclude that RA decoction was able to initiate a positive effect on bone disorder after spinal cord injury, and RA is a hopefully source of new drugs for osteoporosis.
Keywords
Spine cord injury, Osteoporosis, Traditional chinese medicine, Radix astragali.
Introduction
Less than 1% people who suffered significant injury of spinal cord could fully recover, and such injuries would result in partial or complete paralysis [1-3]. The number of people who currently living with spinal cord injury (SCI) in China is estimated to be about 953,000 with 41,000 new cases occurring each year [4]. SCI is responsible for several physiological modifications, including a great decline in bone mineral density (BMD) and an increased risk of fractures [4-6], individuals with functionally-complete SCI have a 20- to 100- fold greater fracture risk than age-matched ambulatory individuals [7], with nearly 50% of those with SCI experiencing one or more low-trauma fractures at some point following injury [8].
Currently, there is a critical need to develop technologies to treat bone loss after SCI. Functional exercise and pharmacological intervention are two major methods to prevent the progress of osteoporosis. Pharmacologic intervention on osteoporosis after SCI includes intake of calcium, phosphate, vitamin D, calcitonin and biphosphonates. To some extent, supplementation of calcium and vitamin D may contribute to calcium homeostasis, but can’t prevent osteoporosis after SCI [9,10]. Similarly, phosphate supplementation can reduce urinary calcium excretion, but does not prevent bone loss after SCI [11]. Tiludronate, pamidronate [12], alendronate and some other drugs were reported to have beneficial effects on osteoporosis after SCI. However, most of such pharmacological treatments didn’t show long-term effects in clinical practice [13].
The puzzle made us to look for other ways and traditional Chinese medicine (TCM) naturally become the alternative with the long history in China. TCM is a systemic and complex science based on a theory different from western medicine. Though the mechanisms of most TCM are unclear, herbal medicines have been used for about two thousand years with apparent efficacy in treating and alleviating various complicated diseases in Asia, including China, Korea and Japan. In fact, there are a few famous and well used herbal formulations in the treatment of bone degradation, such as Danggui Buxue Tang [14], Bushen Huayu Tang, Xianling Gubao capsule [15], Longmu Zhuanggu Granule, etc. Radix Astragali (RA) is one of the herbals constituting Danggui Buxue Tang, Bushen Huayu Tang, Longmu Zhuanggu Granule, and this encouraged us to study the effects of RA alone on bone loss.
Materials and Methods
Preparation of RA decoction
Radix Astragali (root of Astragalus membranaceus (Fisch.) Bge. var. mongholicus (Bge.) Hsiao, RA) prepared pieces were purchased from ShiYiTang Prepared Herbal Medicine Co. Ltd. (Harbin, Heilongjiang, China). The extraction was performed twice in a 10-fold volume of water at 100˚C for 2 h. Decoctions were put together, filtered through 5 layers’ medical gauze, centrifuged for 0.5 h at 15,000 rpm, and then the supernatant was concentrated by low pressure recovery system to a concentration of 1mL decoction equal 1.2 g crude drug.
Experimental design
Forty male Wistar rats (aged 8 weeks and weighing 260-280 g) were used in this study. The rats were maintained under controlled temperature (22-28°C), light–dark periods of 12 h, and with free access to water and a commercial diet. All animal-handling and surgical procedures were strictly conducted according to the Guiding Principles for the Care and Use of Laboratory Animals. This study was approved by the Institute of Orthopedics and Traumatology of Chinese PLA hospital Animal Care and Use Committee guidelines (number 20110289).
Rats were randomly distributed into four groups: Control group, rats with sham operation on spinal cord (n=10); osteoporosis group (Op group), rats with spinal cord sectioned, without any treatment (n=10); L group, rats with spinal cord sectioned and administration of low-dose RA decoction (n=10); H group, rats with spinal cord sectioned and administration of high dose RA decoction (n=10).
Construction of spinal cord-injured rat model
Rats were anesthetized with an intraperitoneal injection of ketamine (90 mg/kg) and xylazine (10 mg/kg), and a laminectomy was performed at Th9-10. In injured rats, the dura mater was exposed, and the spinal cord was completely sectioned with microscissors. In sham-operated animals, the same laminectomy was performed without spinal cord section. During the surgical procedure, body temperature was kept at 37°C to 38°C. After surgery, cefazolin (25 mg/kg; Bristol- Myers Squibb, New York, NY, USA) was subcutaneously administered for 7 consecutive days to all groups to reduce the possibility of bacterial infections. Bladders were manually emptied 3 times daily until sufficient bladder-emptying function returned (about 7 to 10 days after surgery).
Treatments
Treatments started 4 h after surgery, and were performed daily for a consecutive 6 weeks. Upon confirmation of SCI, rats were orally given normal saline 10 mL/kg (control and Op group), RA decoction 10 mL/kg (L group) and 20 mL/kg (H group) respectively.
Behavioral study
Hindlimb movement of the rats was assessed using the Basso- Beattie-Bresnahan locomotor (BBB) rating scale as reported previously [16]. Briefly, the score range from 0 to 21, each number has a description of movement improvement. All rats were previously tested (each one reflected a score of 21). After surgery, all rats were tested one time per week. Two investigators observed the rats for 4 minutes without knowing which treatment was administered (double blinded), and assigned a score depending on the movement of the rat hindlimbs.
Densitometry
Dual energy X-ray absorptiometry (DEXA, Hologic Inc. Discovery model, Belford, MA) was used to measure the BMD (g/cm2) of the right femur, analysis was carried out by using specific software for small animals.
Tissue preparation
The rats were sacrificed by cervical dislocation. Tibiae and femur were collected with soft tissues removed and immersed in normal saline, kept at -20.
Mechanical test
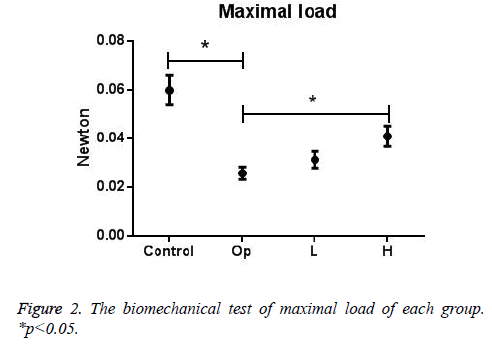
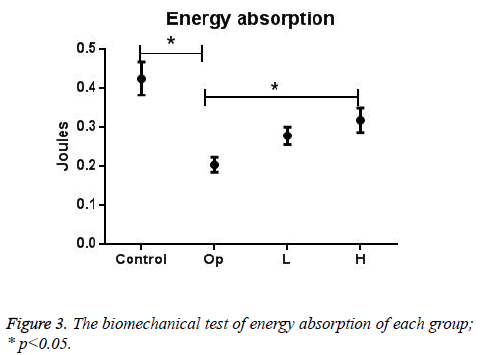
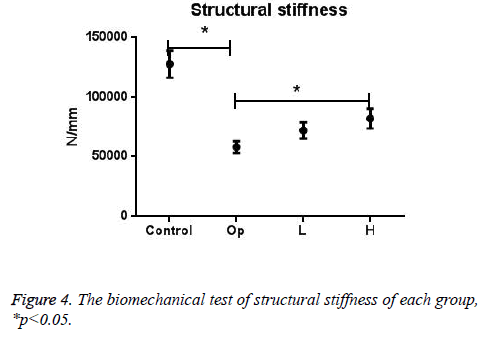
The tibiae were thawed at room temperature overnight before analysis. The specimens were kept in saline to prevent dehydration throughout the evaluation period. Biomechanical properties of the right tibia were determined by a 3-point bending test in Instron Universal Testing Machine (San Francisco, CA, 4444 model, 1 KN load cell). Tibiae were placed on a 3.8 cm long/metal device, which provides a 1.8 cm distant double support on the bone diaphysis. The load cell was perpendicularly positioned at the middle point of the bone. A 5 N preload was applied to avoid specimen sliding. Finally, the bending force was applied at a constant deformation rate of 0.5 cm/min until a fracture occurred. From the load-deformation curve, the maximum load at failure (N), structural stiffness (N/ mm), and energy absorption (J) were obtained.
Statistical analysis
Data were presented as the mean ± SD (n=10). Comparisons among the groups were made using one-way analysis of variance (ANOVA) with SPASS 17.0 and p<0.05 was considered statistically significant.
Results
General findings
The lesion procedure caused severe degradation in behavioral performance, as measured by the BBB rating scale. Rats with spinal cord sectioned showed no plantar placement of the paw with weight support, and the BBB scores were below 7 after the surgery, which means paraplegia was successfully constructed. During the experimental period, spinal cord sectioned rats did not show any recovery in their general motor behavior. Though BBB scores were a little bit higher in L group and H group than Op group, this change was not statistically significant.
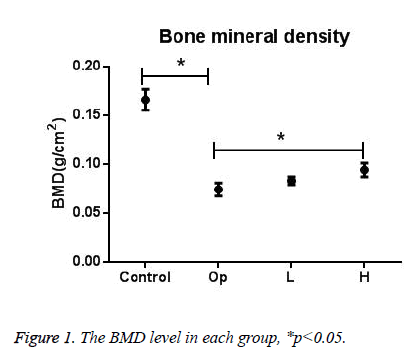
Densitometry
The BMD level of all groups was shown in Figure 1. From the data, we can see that BMD index of Op group was significantly lower than control group, and T<2.5SD (analyzed by software special for small animal. Data didn’t show in the figure), which means osteoporosis happened in femur 6 weeks after spinal cord section. BMD index of H group was significantly higher than Op group, while BMD index of L group was higher than Op group, but without statistical significance, which suggesting that RA extract could reverse bone loss after SCI with a dose dependent manner.
Biomechanical analysis
Data in Figures 2-4 displayed the maximal load, energy absorption, and structural stiffness from biomechanical test of all groups (means ± SD). Statistical analysis showed a high consistency in these three properties: in control group all significantly lower than Op group, and in H group all significantly higher than Op group. The data proved that spinal cord injury could significantly decrease the mechanical strength of tibia, according to publications such decrease could happen to tibia, femur, hipbone etc. Biomechanical properties of H group were significantly higher than Op group, while these properties of L group was higher than Op group, but without statistical significance, which suggesting that RA extract could increase bone strength after SCI with a dose dependent manner.
Discussion
SCI model was obtained by spinal cord section, and to evaluate the model, firstly, BBB rating scale was used to evaluate spinal cord injury, and results showed paraplegia was done. Secondly, BMD were detected to evaluate the bone loss. Normally, BMD<2.5SD was considered osteoporosis happened. Thirdly, to better understand the bone loss condition and evaluate the effects of RA on bone degradation, biomechanical test with tibiae (including maximal load, energy absorption, and structural stiffness test) was acquired besides BMD. The maximum load (N), structural stiffness (N/mm), and energy absorption (J) are widely used in the measurement of bone mechanical strength, especially in several bone disorders [17]. Results in the present study showed a significant decrease in BMD and biomechanical properties in spinal cord injured rats, which are in accordance with other publications. In TCM, herbs that tonify the kidney and stimulate blood circulation are greatly used in the treatment of fracture, arthritis, osteoporosis, etc. RA is an extensively used herbal in TCM. RA is widely used for its liver protective, immunoenhancing, antiinflammatory, antiperspirant, antidiuretic, and antidiabetic properties and as a tonic drug. RA is one of the herbals of Danggui Buxue Tang and Bushen Huayu Tang, which both has been verified could alleviate osteoporosis.
Danggui Buxue Tang (DBT), a Chinese herbal decoction consisting of RA and Radix Angelicae Sinensis with a weight ratio of 5:1, is widely used for menopausal women to nourish and stimulate blood circulation. DBT was proved could significantly increase the number of osteoblasts, intracellular ALP levels, and nodule numbers, while inhibit osteoclast activity. Additionally, DBT could stimulate the proliferation and differentiation of osteoblast by stimulating the downstream signaling of p-ERK and p-JNK [18]. Besides, DBT was verified exhibiting significant estrogenic properties both in vitro and in vivo. The application of DBT did not alter the weight of uterus and liver, as well as the estrogen receptors α and β. However, DBT stimulated the transcript expression of the estrogen responsive genes. In addition, the inductive role of DBT on the expression of members of the aryl hydrocarbon receptor family in uterus and liver of ovariectomized rats was confirmed [19]. It is well know that the deficiency of ovarian hormones, especially estrogens, is the main reason of postmenopausal osteoporosis. No wonder DBT could antagonize menopausal syndrome. And the efficacy of DBT is greater than RA or RAS used alone, which means there are synergistic effects when RA and RAS used together [20].
Though RA is the main part of DBT, there is a deficiency of studies on the efficacy of RA alone on osteoporosis. We hope the present study could be an impactful complement. Beyond that, to adapt to the modernization of herbal medicine, studies on active components and mechanisms are very important. Commonly, there are various components in one herbal medicine. When speaking of RA, active components including astragalosides, isoflavonoids, isoflavones, isoflavan, and pterocarpan glycosides, among which astragaloside IV has been proved having osteogenetic effects. Astragaloside IV was proved could significantly induce osteogenesis related proteins and genes expression of preosteoblasts (OCT-1 cells) when cocultured in 3D-culture, including alkaline phosphatases (ALP), type I collagen (Col I), Osteocalcin (OC), and runt-related transcription factor-2 (Runx2) [21]. In summary, decrease in BMD and biomechanical properties were found in spinal cord sectioned rats, and water extract of RA could antagonize such degradations. From the positive effects of RA on bone degeneration in this study, RA is proved to be a potential source of new drugs for osteoporosis. The next step we should do is digging out the active components in RA extracts and the exactly mechanisms of RA on bone disorders. Although RA extract shows beneficial potential for osteoporosis, it is still a complementary and alternative medicine, it had better be used with other beneficial treatment.
Acknowledgement
We appreciate the funding from Construction of high level health technical personnel in Beijing health system, the funding number is 2014-3-093.
References
- Jiang SD, Jiang LS, Dai LY. Changes in bone mass, bone structure, bone biomechanical properties, and bone metabolism after spinal cord injury: a 6-month longitudinal study in growing rats. Calcif Tissue Int 2007; 80: 167-175.
- Jiang SD, Jiang LS, Dai LY. Spinal cord injury causes more damage to bone mass, bone structure, biomechanical properties and bone metabolism than sciatic neurectomy in young rats. Osteoporosis Int 2006; 17: 1552-1561.
- Medalha CC, Amorim BO, Ferreira JM, Oliveira P, Pereira RM, Tim C. Comparison of the effects of electrical field stimulation and low-level laser therapy on bone loss in spinal cord-injured rats. Photomed Laser Surg 2010; 28: 669-674.
- Wang H, Xiang Q, Li C, Zhou Y. Epidemiology of Traumatic Cervical Spinal Fractures and Risk Factors for Traumatic Cervical Spinal Cord Injury in China. J Spinal Disord Techniques 2013.
- Pluskiewicz W. Quantitative ultrasound assessment of acute bone loss following spinal cord injury: a longitudinal pilot study-comment. Osteoporosis Int 2003; 14: 785.
- Warden SJ, Bennell KL, Matthews B, Brown DJ, McMeeken JM, Wark JD. Quantitative ultrasound assessment of acute bone loss following spinal cord injury: a longitudinal pilot study. Osteoporosis Int 2002; 13: 586-592.
- Frisbie JH. Fractures after myelopathy: the risk quantified. J Spinal Cord Med 1997; 20: 66-69.
- Szollar SM, Martin EM, Sartoris DJ, Parthemore JG, Deftos LJ. Bone mineral density and indexes of bone metabolism in spinal cord injury. Am J Physical Med Rehab 1998; 77: 28-35.
- Schneider VS, McDonald J. Skeletal calcium homeostasis and countermeasures to prevent disuse osteoporosis. Calcif Tissue Int 1984; 36:S151-244.
- Jiang SD, Dai LY, Jiang LS. Osteoporosis after spinal cord injury. Osteoporosis Int 2006;17:180-192.
- Hulley SB, Vogel JM, Donaldson CL, Bayers JH, Friedman RJ, Rosen SN. The effect of supplemental oral phosphate on the bone mineral changes during prolonged bed rest. J Clin Invest 1971;50:2506-2518.
- Zhang S, Xin H, Li Y, Zhang D, Shi J, Yang J. Skimmin, a Coumarin from Hydrangea paniculata, Slows down the Progression of Membranous Glomerulonephritis by Anti-Inflammatory Effects and Inhibiting Immune Complex Deposition. Evidence-based complementary and alternative medicine:eCAM. 2013;2013:819296.
- Bauman WA, Schnitzer TJ, Chen D. Management of osteoporosis after spinal cord injury: what can be done? Point/counterpoint. PMR 2010;2:566-572.
- Wang WL, Sheu SY, Chen YS, Kao ST, Fu YT, Kuo TF. Enhanced Bone Tissue Regeneration by Porous Gelatin Composites Loaded with the Chinese Herbal Decoction Danggui Buxue Tang. PloSone 2015;10:e0131999.
- Jiang M, Han ZY, Yue HX, Li FP, Lin L, She WJ. Xianling Gubao Capsule improves semen quality. Zhonghua nan ke xue 2008;14:1146-1148.
- Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 1995;12:1-21.
- Peng Z, Tuukkanen J, Zhang H, Jamsa T, Vaananen HK. The mechanical strength of bone in different rat models of experimental osteoporosis. Bone 1994;15:523-532.
- Wang WL, Sheu SY, Chen YS, Kao ST, Fu YT, Kuo TF. Evaluating the bone tissue regeneration capability of the Chinese herbal decoction Danggui Buxue Tang from a molecular biology perspective. BioMed Res Int 2014;2014:853234.
- Zierau O, Zheng KY, Papke A, Dong TT, Tsim KW, Vollmer G. Functions of Danggui Buxue Tang, a Chinese Herbal Decoction Containing Astragali Radix and Angelicae Sinensis Radix, in Uterus and Liver are Both Estrogen Receptor-Dependent and -Independent. Evidence-based complementary and alternative medicine: eCAM. 2014;2014:438531.
- Choi RC, Gao QT, Cheung AW, Zhu JT, Lau FT, Li J. A chinese herbal decoction, danggui buxue tang, stimulates proliferation, differentiation and gene expression of cultured osteosarcoma cells: genomic approach to reveal specific gene activation. Evidence-based complementary and alternative medicine: eCAM. 2011;2011:307548.
- Bian Q, Huang JH, Liang QQ, Shu B, Hou W, Xu H. The osteogenetic effect of astragaloside IV with centrifugating pressure on the OCT-1 cells. Die Pharmazie 2011;66:63-68.