- Biomedical Research (2006) Volume 17, Issue 3
Antioxidant treatment a new therapeutic approach to reversible male infertility
Sunil B. Yadav1*, Adinath N. Suryakar2, Anil D. Huddedar3, Puspha P. Durgawale1, Pramod S. Shukla11Department of Biochemistry, Krishna Institute of Medical Sciences Deemed University, Karad (MS), India
2Department of Biochemistry, Dr. V.M.Medical College, Solapur (MS), India
3Department of Surgery, Krishna Institute of Medical Sciences Deemed University, Karad (MS), India
- Corresponding Author:
- Sunil Baburao Yadav
Department of Biochemistry
Krishna Institute of Medical Sciences Deemed University Karad (MS), India
Phone: 9823389234
Residence: (02164) 281219
Fax: (02164) 242170
Office: (02164) 214555, ext 305
E-mail: ombamalu@uwc.ac.za
Accepted Date: August 07 2006
Abstract
The imbalance between reactive oxygen species (ROS) production and total antioxidant capacity in seminal fluid indicates oxidative stress and is correlated with male infertility. One of the rational strategies to counteract the oxidative stress is to increase the scavenging capacity of seminal plasma. In such cases a combination of antioxidants may e more useful treatment of male infertility. We therefore undertook this study the usefulness of antioxidant supplementation on seminal oxidative stress and its relation with sperm function test. In present study total Sixty one oligozoospermia patients and forty four fertility proved healthy donors with normal sperm analysis were included in the study. Seminal lipid peroxide, nitric oxide and total antioxidant power and various parameters were compared including sperm function test among the fertile control and before & after treatment of oligozoospermic patients. Of the 61 untreated oligoozoospermia patients the mean lipid peroxide and nitric oxide levels were found to be increased along with decreased total antioxidant power and sperm function ability as compared to fertile control subjects. However significant reduction in lipid peroxide and nitric oxide and improvement in seminal total antioxidant power, sperm function ability were observed after supplementary treatment of combined antioxidant.
Keywords
Antioxidants, Male infertility, Oxidative stress, Sperm function ability
Introduction
Infertility is defined as the failure of conception at least 12 month’s of unprotected intercourse [1]. Infertility is a worldwide problem and approximately 8-10% of couples within reproductive age group are infertile [2]. It is estimated that globally 60-80 million couples suffer from infertility every year, of which probably between 15-20 million are in India alone [3]. In the era of evidencebased medicine specific management of infertility should be based on identifying reversible causes of infertility and treating them with suitable medications. However this may constitute a challenge. Since inspite of extensive research no identifiable cause can be found in 25% of infertile males [4]. Recently, oxidative stress has become the focus of interest as a potential cause of male infertility [5,6]. Normally, equilibrium exists between reactive oxygen species (ROS) production and antioxidant scavenging activities in the male reproductive tract. Under physiological conditions, spermatozoa produces small amount of ROS, which are needed for capacitation acrosome reaction and fertilization 7. However excessive amount of ROS produced by leukocytes and immature spermatozoa can cause damage to the normal spermatozoa by inducing lipid peroxidation and DNA damage [8,9,10]. High concentration of ROS was detected in the semen of 30-80% infertile men [11]. In view of this rational strategies with the goal of reducing concentration of oxidative stress may be effective in the treatment of male infertility. Initially Clinician can identify and treat the cause for increased ROS production like reproductive tract infection, smoking, varicocele etc.[12]. Following this, augmentation of the scavenging capacity of the seminal plasma can be an effective treatment for the male infertility.
Material and Methods
Semen Samples
A total of sixty One oligozoospermic patients (sperm count is below 20 X 106) visiting the Infertility Clinic, at Krishna Hospital and Medical Research Centre, Karad and forty four healthy normozoospermic matched non patients controls were included in the study. Institutional ethical committee clearance was taken. All subjects were in the age group between 21-45 years. They were advised to observe an abstinence of 3 days and to deliver semen sample on 4th day. After due orientation about nature of study a written informed consent was taken from healthy individuals and infertile male subjects. After liquefaction, ejaculates were analyzed for volume, pH, sperm count, percentage motility, morphology and sperm function test according to the guidelines prescribed by World Health Organization [13]. As per the WHO criteria semen specimens were divided into two group of normozoospermic having >20 X 106 sperms/ ml. with normal morphology and motility and oligozoospermic with <20 X 106 sperms/ ml.
After centrifugation seminal plasma was used for measurement of free radical production by estimating malondialdehyde[14] and assays of nitric oxide by Griess reaction[15]. In kinetic method in which nitrate is reduced to nitrite by copper coated cadmium granules. Total anti oxidant power in seminal plasma was measured as ferric reducing ability of plasma (FRAP) assay of Benzie I.F.F. [16].
Sperm function test was carried out hypoosmotic swelling (HOS) test [17]. Healthy viable sperms having good fertilizing potential have the property of curling of tails when exposed to the stress of hypoosmotic pressure. The unhealthy poor quality sperm do not have curling property under the hypoosmotic pressure. Oligozoospermic patients were followed after three months of supplementary multiple antioxidant treatment with commercial available Tab.A-Z antioxidant – 1 Tab /Day (Vit. C 100 mg., Niacinamide 50 mg., Vit. B1 10 mg., Vit. B2 10 mg., Vit. B6 3 mg., Calcium Pantothenate 12.5 mg., Folic acid 1 mg., Vit. B12 5.0 mcg., Vit. A (as acetate) 5000 i.u., Vit. D3 500 i.u. Vit.E acetate 25 i.u., zinc oxide 15 mg., cupric oxide 2.5 mg., sodium selenate 60 mcg., mangnese chloride 1.4 mg., chromium chloride 60 mcg.) in addition to with Clomiphene citrate 25mg treatment was given for period of three months.
Statistical Analysis
Mean values were compared using the unpaired student’s ‘t’ test.
Results
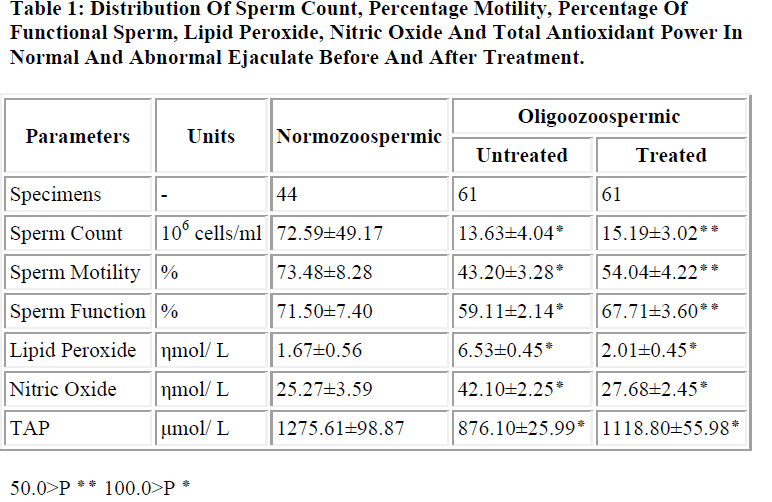
Table 1 shows the distribution of sperm count, percentage sperm motility, percentage of functional sperm, lipid peroxide in term of MDA, nitric oxide and total antioxidant power in Normozoospermic and Oligozoospermic patients before and after treatment.
In the present study, it was observed that, lipid peroxide and nitric oxide concentration were significantly higher (P<0.001) in untreated Oligozoospermic male patients as compared to Normozoospermic. Further significant reduction (P<0.05) in lipid peroxide and nitric oxide were observed in Oligozoospermic group after treatment.
Significant lower values of (P<0.001) sperm count, motile sperm and number of functional sperms was observed in Oligozoospermic cases before treatment correlating well with increased free radical.
Non-enzymatic total antioxidant power was found to be significantly reduced (P<0.001) in untreated Oligozoo-spermic patients as compared to normal fertile control. Further after combined treatment of antioxidant along with conventional therapy to such individuals, significantly elevated (P<0.005) level of total antioxidant power was observed and it positively correlated with sperm function ability.
Discusstion
The identification of adequate molecular markers to diagnose a semen sample, in conjugation with the design of appropriate therapeutic strategies to combat defects and deficiencies, is decisive for resolving male factor problem. Among these, oxidative stress markers are relevant factors in male infertility, since they play a significant role in sperm physiology. A fine balance of these markers could determine the reproductive success [18]. Although the importance of seminal plasma in the protection of spermatozoa against ROS is well established as other few studies [6,7,10] have investigated its antioxidative properties, and the possible relationship between infertility and plasma antioxidant defence system.
The lipid composition of plasma membrane of mammalian spermatozoa is markedly different from mammalian somatic cells. They have very high levels of phospholipids, sterols, saturated and polyunsaturated fatty acids. Therefore sperm cells are particularly susceptible to the damage induced by excessive ROS release [19-20]. The most significant product of lipid peroxidation is malondialdehyde (MDA). Lipid peroxidation plays a major role in the aetiology of defective sperm function. This may lead to the onset of male infertility via the mechanism involving the induction of peroxidative damage to plasma membrane [20-21].
Nitric oxide (NO) is another oxygen free radical which is apparently generated by defective population of spermatozoa. NO is normally generated from L-arginine by three isoforms of nitric oxide synthase (NOs) [22] .The fact that iNOs (inducible nitric oxide synthase) deficient spermatozoa exhibited significantly higher in vitro fertilization rates than the wild type control, clearly suggests that Nitric oxide may be involved in the aetiology of defective sperm function. Nitric oxide clearly has a detrimental effect on normal sperm function inhibiting both motility and the competence of these cells for sperm – zona binding [23] . If the sperm lipid peroxidation and nitric oxide inhibits sperm maturation, the therapeutic intervention would be beneficial [22]. In the present study, increased levels of lipid peroxide and nitric oxide along with decreased total antioxidant power in seminal plasma of infertile male is an indicative of increased oxidative stress. A negative correlation was observed between the oxidative stress markers (Lipid peroxide and nitric oxide) and sperm function ability and sperm motility. This suggests that lipid peroxidation and nitric oxide were responsible for significant reduction in sperm function ability and sperm motility. A negative correlation between total ferric reducing ability of plasma and lipid peroxide and nitric oxide were observed respectively. Further after treatment with multiple antioxidants as supplementary to such individuals, significant elevated total antioxidant power along with decreased lipid peroxide and nitric oxide were observed. This change in status of seminal plasma positively correlated with impaired sperm function. Thus the present study suggests that multiple antioxidant treatment may be helpful to maintain the balance between prooxidant and antioxidant, which may prevent damage of sperm membrane by oxidative stress and help in improvement of the sperm function ability. Further studies are warranted to determine the structure and functional changes in sperm membrane lipid during the process of peroxidation and this may be useful in understanding the role of lipid metabolism in spermatozoa physiology and may help to develop novel therapeutic strategies for male infertility.
References
- Rowe PJ, Compaire FH, Hargereave TB, Mellows HJ. WHO manual for standard investigation and diagnosis of the infertile couple. Cambridge University Press, Cambridge1993
- World Health Organization. Recent advances in medically assisted conception. WHO Tech Res Ser 1992; 820: 2.
- World Health Organization. The world health report. WHO, Geneva 1996.
- March MR, Isidori A. New frontiers in the treatment of male infertility. Contraception 2002; 65:279-281.
- Agarwal A, Said TM. Role of sperm chromatin abnormalities and DNA damage in male infertility. Human reproduction Update 2003; 9: 331-345.
- Aitken RJ, Baker MA, Sawyer D. Oxidative stress in the male germ line and its role in the aetiology of male infertility and genetic diseases. Reproduction Medicine Online 2003; 7: 65-70.
- Griveau JF, Le Lannou D. Reactive oxygen species and human spermatozoa: Physiology and Pathology. International Journal of Andrology 1997; 17: 225-231.
- Wang X, Sharma RK, Sikka SC, et al. Oxidative stress is associated with increased apoptosis leading to spermatozoa DNA damage in patients with male factor infertility. Fertility and Sterility 2003; 80: 531-535.
- Moustafa MH, Sharma RK, Thornton J, et al. Relationship between the ROS production, apoptosis and DNA denaturation in spermatozoa from patients examined for infertility. Human Reproduction 2004; 19: 129-138.
- Sikka SC. Role of oxidative stress and antioxidants in andrology and assisted reproduction technology. Journal of Andrology 2004; 25:5-18.
- Padron O, Brackett N, Sharma R, et al. Seminal reactive oxygen species , sperm motility and morphology in men with spinal cord injury. Fertility and Sterility 1997; 67: 1115-1120.
- Sharma RK, Pasqualotto AE, Nelson DR, et al. Relationship between seminal while blood cell count and oxidative stress in men treated at an infertility clinic. Journal of Andrology 2001; 22: 575-583.
- World Health Organization: WHO laboratory manual for the examination of human semen and semen cervical mucus interaction. 2nd Edition Cambridge: The Press Syndicate of The University of Cambridge 1987; 59: 1-59.
- Satoh K. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chem Acta 1978; 90: 37-43.
- Najwa KC and Nabil WW. Determination of inorganic nitrate in serum and urine by kinetic cadmium reduction method. Clinical Chemistry 1990;36(8):1440-1443.
- Benzie Iris FF, and Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of antioxidants power; the FRAP assay. Analytical Biochemistry 1996; 239: 70-76.
- Khadilkar SS, Parathe, Tayade SM. Role of sperm function tests in unexplained infertility. Journal of Obst & Gynecology of India 2001; 51: 146-149.
- Nicoles G, Marcos M, Carlos S, Antonio P, Jos R. Pro oxidative and antioxidative imbalance in human semen and its relation with male infertility. Asian J. Andrology 2004; 6: 59-65.
- Alvarez JG, Storey BT. Differential incorporation of fatty acids into and peroxidative loss of fatty acids from phospholipids of human spermatozoa. Male Reprod Dev 1995; 42: 334-346.
- Jones R, Mann T, Sherine RJ. Peroxidative breakdown of phospholipids in human spermatozoa, spermicidal properties of fatty acid peroxides and protective action of seminal plasma. Fertile and Sterile 1979; 31: 531-537.
- Yadav SB, Suryakar AN, Huddedar AD, Shukla PS. Effect of antibiotics and antioxidants on levels of seminal oxidative stress in leukocytospermic infertile men. Indian Journal of Clinical Biochemistry 2006;21: 152-156.
- Mark AB, Aitken RJ. Review: Reactive oxygen species in spermatozoa: methods for monitoring and significance for the origins of genetic diseases and infertility. Reproductive Biology and Endocrinology 2005; 3: 67.
- Yang JZ, Ajonuma LC, Rowlands DK, Tsang LL, HO Ls, Lam SY, et al. The role of inducible nitric oxide synthase in gamete interaction and fertilization: a comparative study on knockout mice of three NOS isoforms. Cell Biol Int 2005; 29: 785-791.