Case Report - Journal of Bacteriology and Infectious Diseases (2018) Journal of Bacteriology and Infectious Diseases (Special issue 2-2018)
A case of post-vaccination tubercular abscess in thigh of an infant.
Sumit Chavan1*, P Vishalakshi1, DVPR Vittal1, Mohammed RasheedUddin1, Neha1, Deeraj Kondagari2
1Department of Microbiology, Vijaya Diagnostic Centre Pvt Ltd, Hyderabad, India
2Department of Medicine, Yashoda Hospitals, Hyderabad, India
- *Corresponding Author:
- Sumit Chavan
Department of Microbiology
Vijaya Diagnostic Centre Pvt Ltd
3-6-16 &17, Street No # 19, Himayat Nagar
Hyderabad 500029, India
Tel: 919850194433
E-mail: sumitchavan28@yahoo.co.in
Accepted Date: June 08, 2018
Citation: Chavan S, Vishalakshi P, Vittal DVPR, et al. A case of post-vaccination tubercular abscess in thigh of an infant. J Bacteriol Infec Dis. 2018;2(2):16-18
Abstract
We report a rare case of post vaccination tubercular soft tissue abscess on a lateral part of thigh in an immunocompetent infant without underlying source of infection. A soft tissue cold abscess usually results from an underlying bone infection; however, a positive case due to intramuscular hexavalent vaccination was presented in our case.
Keywords
Abscess, tuberculosis, injection, immunocompetent.
Introduction
To determine causal relationship of adverse event following vaccination with specific vaccine is often challenging. Most of the abscess formations at the site of vaccination are pyogenic in nature caused by bacterial pathogen; common causative agents include Staphylococcus aureus, Escherichia coli, Klebsiella pneumonia, Pseudomonas spp. Rarely rapid growing nontuberculous mycobacteria (NTM) and M. bovis are implicated as a cause due to contaminated needles or probable use of tap water, the common reservoir of M. chelonae and M. fortuitum. Mycobacterium tuberculosis causing abscess formation at vaccination site is very rare complication and has been reported by few authors [1-4].
Intramuscular (IM) administration of vaccine contributes up to 21.5%; a significant proportion of post vaccination abscess cases [5]. Most of reported cases are attributed to use of a contaminated needle/ syringe and lack of aseptic measures while administration [6]. Even though IM injections are a minor procedure, needs asepsis and appropriate administration technique to prevent rare but serious complications.
We herein report a rare case of tubercular soft tissue abscess formation post intramuscular vaccination. This work was approved by institutional ethical review committee and written informed consent was obtained from the child’s parents.
Case Report
A healthy four months old male child developed swelling and erythema at the site of an injection on anterolateral part of right thigh. The lesion was appeared four days following his third dose of intramuscular Hexavalent vaccine (combination of Diphtheria toxoid-tetanus toxoid-acellular pertusis, inactivated Poliomyelitis, Hepatitis B and H influenza type b vaccines) administered at some private clinic. The previous vaccinations including Hexavalent vaccine first, second dose and BCG at birth were well tolerated without any post-vaccination complications. There was no past history of trauma and/or any disease. The child was thriving well with adequate weight gain.
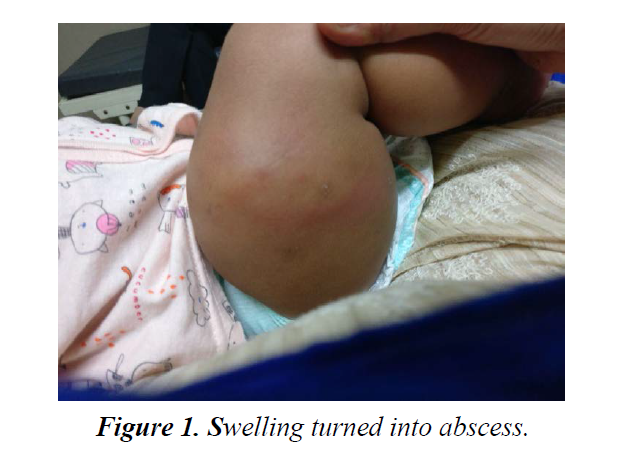
On clinical examination done one week post vaccination showed erythematous swelling of 2 × 1 cm size, over anterolateral aspect of right thigh with mild to no tenderness. Movement of legs were not restricted. Vital signs remained normal and afebrile. Child showed no toxic features except slight irritability and discomfort. Ultrasound of right thigh showed intramuscular liquefied hematoma. The lesion progressively enlarged over next one and half months to form an abscess (around 40-50 cc) measuring approximately 5.0 × 3.0 × 5.3 cm. Local inguinal lymph nodes were enlarged. No underneath bone involvement noted.
Around same time needle aspiration was done as the lesion did not respond to empirical antibiotic therapy with Intravenous ampicillin-sulbactum for one week followed by oral amoxyclav for another week.
Aspirated fluid was found positive for AFB (acid fast bacilli) on Ziehl Neelsen (ZN) staining and aerobic bacterial culture was sterile.
GeneXpert MTB/RIF assay (Real time nested PCR, Cepheid) corroborated to AFB staining by detecting MTB complex sensitive to Rifampicin.
On AFB culture (BD MGIT 320, liquid culture), Mycobacterium growth was seen after 15 days and was also positive on solid media (Lowenstein Jensen medium) after 23 days of incubation. The isolate was sensitive to first line antitubercular drugs (BD MGIT 320) which includes streptomycin, isoniazid, rifampicin and ethambutol (SIRE).
There was no evidence of primary focus of tuberculosis on clinical and radiographic examination. Haematological parameters were normal except mild elevation of lymphocyte count. Immunodeficiency disorders were ruled out by normal haemoglobin; complements and Immunoglobulin profile.
The RNTCP (Revised national tuberculosis control program) antituberculos (ATT) regimen for new cases of tuberculosis (Isoniazid 5 mg/kg, rifampicin 10 mg/kg, pyrazinamide 25 mg/ kg and ethambutol 15 mg/kg daily for first 2 months followed by Isoniazid, rifampicin and ethambutol daily for 4 months) was commenced two months post abscess formation. Follow up was taken every weekly for first two months followed by every two weeks for any adverse effects, weight gain and dimensions of lesion.
Localised recollection of fluid was started requiring Incision and drainage, 3 wks. after commencement of ATT. Fluid sample was negative for AFB on staining and culture was not done. Antibiotic course was given to cover procedural risk of bacterial infection. Swelling turned into abscess (Figure 1).
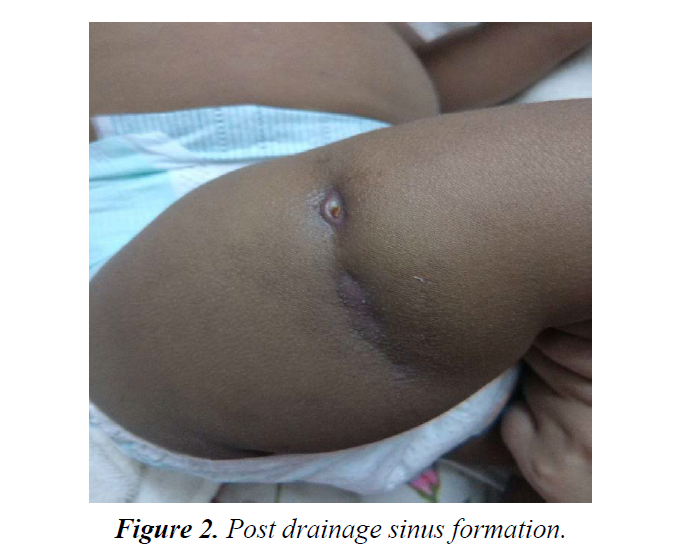
Post procedure, lesion formed active sinus tract formation draining subcutaneous plane to skin (Figure 2). At the entry point on skin, lesion was developed, diagnosed by dermatologist as tubercular lesion- scrofuloderma.
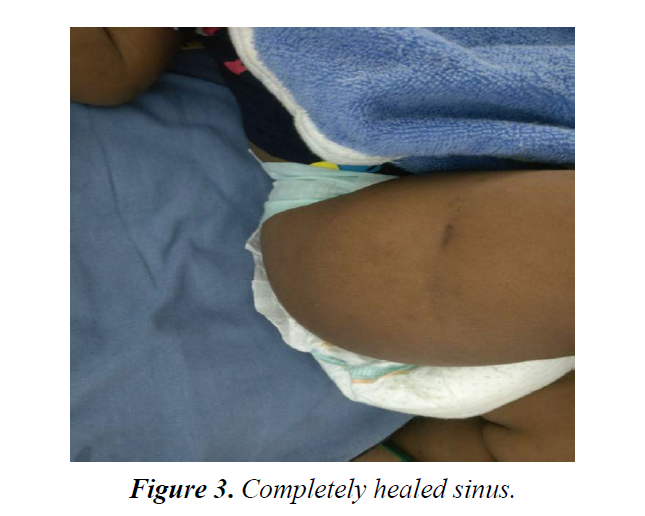
A child tolerated ATT well. Sinus slowly started healing after 4 months of ATT and showed complete healing by 5th month (Figure 3). ATT was given for total period of 6 months and by this time child was completely recovered with no residual lesion.
Discussion
In our case, tubercular origin of abscess was microbiologically confirmed by positive AFB microscopy, MGIT broth based culture, positive Gene Xpert MTB RIF (nested real time PCR) supplemented by clinical findings such as normal systemic parameters, clinical response to ATT and development of tubercular skin lesion-scrofuloderma at sinus entry point.
Hexavalent vaccine has shown good efficacy and is one of the safest vaccines available. As first two doses of hexavalent vaccines were well tolerated, vaccine cannot be completely attributed as a cause for complication.
Mishra et al. (2013), Agrawal et al. (2007) and Dixit et al. (2014) have reported cases of tuberculosis gluteal abscess caused by Mycobacterium tuberculosis. All three cases were of immunocompetent female infant and gave previous history of DTP vaccination. A variable time period observed between vaccination and appearance of swelling of 5 days in Mishra et al, 15 days in Agrawal et al. and 21 days in Dixit et al. [2-4].
Most of such complications are secondary to direct spread from underlying bone or tissue infection and contaminated vaccine use with improper administration technique. Few authors have suggested rare possibilities such as inadvertent intramuscular injection of the BCG vaccine, direct inoculation of injection site with coughed up tuberculosis bacilli by infected nursing staff, needle introducing tubercle bacilli from skin surface to injured tissue etc. [2,4,7-9].
Shah et al. (2015) describes few cases of tubercular thigh abscess formation in infants less than 6 months old following hepatitis B vaccine. Here diagnosis was made by histopathology report stating granulomatous changes along with caseous necrosis. This case report suggests probable haematogenous spread from primary focus somewhere else in body [10].
Possibility of contaminated needle use, lack of aseptic precautions and inadvertent BCG vaccination were ruled by parents who accompanied baby at the time of vaccination.
Thus, we are unable to explain origin of infection and also its association with vaccine administration. Pharmacovigilance of adverse events following immunization are essential with newly introduced vaccines recommended for a routine immunization. A large scale surveillance studies are needed to detect rare and serious adverse events.
We suggest occult haematogenous dissemination of Tubercle bacilli from primary focus somewhere else in body may get lodge in hematoma at injection site and multiply to form an abscess. Such dissemination of tubercle bacilli from primary focus may take one week to three months and last for a very short time [1].
To conclude, Tuberculosis abscess in immunocompetent infant is less common and poses diagnostic challenge as it’s not considered in initial differential diagnosis. High degree of clinical suspicion is required for early diagnosis and complete treatment for better outcome.
References
- Gupta P, Menon PSN, Ramji S, et al. PG Textbook of Pediatrics: Infections and Systemic Disorders; 2:1139-1147.
- Mishra D, Mohta A, Arora P. Cold abscess of the thigh in an infant. Kathmandu Univ Med J. 2013;41:86-87.
- Agrawal A, Jain A. Tuberculosis Cold Abscess. Indian J Pediatr 2007;74:771-773.
- Dixit R, Dixit K, Patil CB, et al. Injection site abscess due to Mycobacteriology tuberculosis following DPT vaccinations. Indian J Tuberc 2014;61:246-249.
- Gondal KM, Iqbal Y, Khan UA, et al. Frequency of Contributing Factorsin Intramuscular Injection Abscess. PJMHS 2014; 569-572.
- Tan NWH, Pan WJ, Yunos HBM, et al. Postvaccination abscesses requiring surgical drainage in a tertiary childrens hospital in Singapore. J Pediatr Infect Dis 2010;5:139-147.
- Namshan M, Oda O, Zamakhshary M, et al. Bacillus Calmette Guerin related cold thigh abscess as an unusual cause of thigh swelling in infants following BCG vaccine administration: a case series. J Med Case Reports 2011;5:472.
- Verma S, Didel S. Cold abscess of thigh following inadvertent vaccination in an infant; Indian J Child Health 2016;3:76-78.
- Sharma J, Sharma T, Bhatt GC, et al. Isolated cold abscess of the thigh in an immunocompetent infant. Trop Doct 2014;44:221-222.
- Shah A, Dash B. Primary Tubercular Abscess of Thigh in Infancy. Indian Pediatr 2015;52:992.