Short Communication - Annals of Cardiovascular and Thoracic Surgery (2018) Volume 1, Issue 2
Wall motion score index predicts mortality after coronary artery bypass grafting in patients with viable non-functioning myocardium
Yasser Ali Kamal1*, Shady EM Al-Elwany1, Ahmed MF Ghoneim2 and Ahmed MK El-Minshawy2
1Department of Cardiothoracic Surgery, Minia University, Egypt
2Department of Cardiothoracic Surgery, Assiut University, Egypt
- *Corresponding Author:
- Dr. Yasser Ali Kamal
Department of Cardiothoracic Surgery
Minia Cardiothoracic University Hospital
Minia 61768, Egypt
Tel: 20-01123234736
E-mail: yaser_ali_kamal@yahoo.com
Accepted on July 23, 2018
Citation: Kamal YA, Al-Elwany SEM, Ghoneim AMF et al. Wall motion score index predicts mortality after coronary artery bypass grafting in patients with viable non-functioning myocardium. Ann Cardiovasc Thorac Surg 2018;1(2):39-40.
DOI: 10.35841/cardiovascular-surgery.1.2.39-40
Visit for more related articles at Annals of Cardiovascular and Thoracic SurgeryAbstract
Coronary artery bypass grafting (CABG) has better survival than medical treatment in patients with left ventricular (LV) dysfunction. Assessment of myocardial viability is crucial to predict survival benefit after CABG. Our recent work determined higher prognostic value of echocardiographic wall motion score index (WMSI) than left ventricular ejection fraction (LVEF) in patients underwent CABG with viable myocardium and LVEF <50%. Thus, it is important to determine the extent of nonviable as well as viable myocardium in patients with low LVEF. Also, in the absence of high modality imaging studies like cardiac magnetic resonance (CMR) and to avoid the radiation hazards of radionuclide studies, assessment of WMSI in addition to LVEF can give accurate data of the magnitude of myocardial damage and can predict survival after CABG in patients with systolic LV dysfunction.
Keywords
Myocardial revascularization, left ventricular dysfunction, Low ejection fraction, Myocardial viability.
Commentary
Coronary artery disease (CAD) is a leading cause of left ventricular (LV) systolic dysfunction which results in higher mortality rates than non-ischemic etiologies [1]. Also, LV systolic dysfunction is known as a predictor of in-hospital mortality after CABG, and associated with multiple perioperative risk factors that affect the survival benefit of CABG [2]. However, in patients with LV systolic dysfunction, coronary artery bypass grafting (CABG) provides the potential for incremental survival benefit compared to medical treatment [1,3]. Moreover, a recent study demonstrated a greater postprocedural short and long-term survival benefit of CABG over PCI in patients with poor LV function (ejection fraction <30%) [4].
The predominance of tissue viability has a good impact on safety and efficacy of CABG in patients with LV dysfunction. Thus, myocardial viability is crucial for selection of patients with nonfunctioning myocardium to undergo CABG [5]. The use of noninvasive techniques to determine the myocardial viability, particularly echocardiography, provides important information to guide clinicians in selecting patients with LV dysfunction likely to receive benefit from CABG [6].
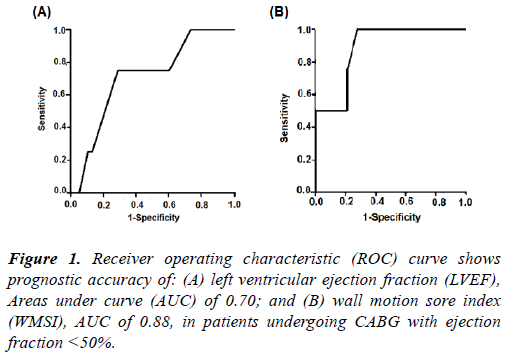
Recently, we published a study aiming to determine the perioperative predictors of adverse outcome after coronary artery bypass grafting (CABG) in patients with reduced left ventricular ejection fraction (LVEF; <50%) [2]. All of our patients had viable nonfunctioning myocardium. Wall motion score index (WMSI) determined on dobutamine stress echocardiography (DSE) in patients undergoing CABG with viable non-functioning myocardium had a good prognostic value (Figure 1).
Wall motion score index (WMSI) reflects the magnitude of myocardial damage and total extent of wall motion abnormalities. Our results indicate that not the presence of viable myocardium or improved LVEF on stress echocardiography but the magnitude of myocardial damage, has a positive impact on survival after revascularization. In other words, measurement of regional variability of LV function by WMSI predicts in-hospital mortality after CABG, but not a global measurement using end-systolic and enddiastolic volume indices.
As recommended by the American Society for Echocardiography a 16-segment model was used for left ventricular segmentation. Each segment was analyzed individually and scored on the basis of its motion and systolic thickening. Each segment’s function was confirmed in multiple views. Segments were scored are as: normal or hyperkinesis=1, hypokinesis=2, akinesis=3 and dyskinesis (or aneurysmatic)=4. WMSI was derived as the sum of all scores divided by the number of segments visualized [7].
It has been described that a combined analysis of LVEF and WMSI seems preferable to the measurement of LVEF alone. The presence of hypercontractile segments may limit the reduction in systolic function measured by LVEF without limiting it when measured with WMSI since it is based on the contractility of each segment and scores equal the hypercontractile and normal segments. The main difference between WMSI and LVEF is that the WMSI rates equally normokinesia and hyperkinesia, avoiding the compensation that hypercontractile segments make on the dysfunctional ones in the measurement of LVEF and therefore assessing more directly the intensity and extent of the myocardial damage [8].
It was postulated that non-invasive methods should identify and assess non-viable myocardium as well as viable myocardium since the combined data appear to provide a more accurate and comprehensive evaluation of myocardial viability [9]. The new imaging techniques, such as strain and cardiac magnetic resonance imaging (CMR) are more precise for determining myocardial damage. However, these are less accessible techniques for daily practice. A good correlation of the echocardiographic WMSI with the strain has been reported [8]. Thus, in the absence of CMR, assessment of WMSI by echocardiography provides important data about the magnitude of regional myocardial damage.
In conclusion, preoperative assessment of WMSI using stress echocardiography has an important prognostic role in patients with LV dysfunction undergoing CABG, and it helps identify patients who may benefit most from CABG. However, our finding is limited by small size of our patients, which indicates further large-scale studies with the same concern.
References
- Bonow RO. Indications for revascularization in patients with left ventricular dysfunction: Evidence and uncertainties. J Thorac Cardiovasc Surg. 2014;148(6):2461- 5.
- Kamal YA, Al-Elwany SM, Ghoneim AM, et al. Predictors of adverse effects after coronary artery bypass grafting in patients with reduced left ventricular ejection fraction. Journal of the Egyptian Society of Cardio-Thoracic Surgery. 2017;25:20-7.
- Velazquez EJ, Lee KL, Deja MA, et al. Coronary-artery bypass surgery in patients with left ventricular dysfunction. N Engl J Med. 2011;364:1607-16.
- Shah S, Benedetto U, Caputo M, et al. Comparison of the survival between coronary artery bypass graft surgery versus percutaneous coronary intervention in patients with poor left ventricular function (ejection fraction <30%): a propensity-matched analysis. Eur J Cardiothorac Surg. 2018.
- Salehi M, Bakhshandeh A, Rahmanian M, et al. Coronary Artery Bypass Grafting in Patients with Advanced Left Ventricular Dysfunction: Excellent Early Outcome with Improved Ejection Fraction. J Tehran Heart Cent. 2016;11(1):6-10.
- Meluzin J, Cerny J, Frelich M, et al. Prognostic value of the amount of dysfunctional but viable myocardium in revascularized patients with coronary artery disease and left ventricular dysfunction: Investigators of this Multicenter Study. J Am Coll Cardiol. 1998;32(4):912-20.
- Lang RM, Bierig M, Devereux RB, et al. Recommendations for chamber quantification: A report from the American society of Echocardiography’s guidelines and standards committee and the chamber quantification writing group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18:1440-63.
- Jurado-Roman A, Agudo-Quilez P, Rubio-Alonso B, et al. Superiority of wall motion score index over left ventricle ejection fraction in predicting cardiovascular events after an acute myocardial infarction. Eur Heart J Acute Cardiovasc Care. 2016. pii: 2048872616674464.
- Kim RJ, Shah DJ. Fundamental concepts in myocardial viability assessment revisited: when knowing how much is “alive” is not enough. Heart. 2004;90(2):137-40.