Research Article - Biomedical Research (2017) Volume 28, Issue 16
Uterine resectoscopic myomectomy with and without microrelin pretreatment: a single-blinded randomized clinical trial
Abbas Norouzi Javidan1, Mina Jafarabadi2*, Sahar Latifi1, Masoumeh Farhadkhani1 and Mansoureh Gorginzadeh2
1Brain and Spinal Injury Research Center, Tehran University of Medical Sciences, Iran
2Reproductive Health Research Center, Tehran University of Medical Sciences, Iran
- *Corresponding Author:
- Mina Jafarabadi
Reproductive Health Research Center
Tehran University of Medical Sciences, Iran
Accepted date: June 08, 2017
Abstract
Study objective: To assess the outcomes of surgery in the group of patients who were submitted to preoperative GnRH agonists in comparison with those who received no medication. The levels of gonadotropin hormones (LH and FSH) and estradiol, which were measured 2 and 12 weeks after treatment, were also compared between two groups.
Design: Single-blinded randomized clinical trial. Design classification: Canadian Task Force Classification I.
Setting: University hospital.
Patients: A total of 60 patients participated in this clinical trial. Inclusion criteria were: age between 18-50 y (premenopausal women).
Interventions: Sixty patients were assigned randomly to intervention and control group. Intervention group received three doses of monthly intramuscular injections of Microrelin 3.75 mg (a sustained release formulation of triptorelin) before hysteroscopic myomectomy and control group received no medications. Uterine resectoscopic myomectomy was performed in both groups.
Measurements and main results: Operation characteristics including operation time and amount of absorbed fluid were measured. Level of difficulty and satisfaction from surgeon’s view were assessed based on Visual Analog Scale (VAS). Serum levels of LH, FSH and estradiol were measured at the baseline and after 2 and 12 weeks in both groups. A total of 60 patients participated in this clinical trial. The sizes of myomas were significantly reduced in microrelin group in comparison with control group (33% reduction, p<0.0001). The administration of Microrelin led to significant reduction of estradiol, LH and FSH levels after 12 weeks. However a transient flare-up in the level of estradiol was detected after 2 weeks. The satisfaction level from operation from surgeon’s view was significantly higher (p=0.01) and the level of difficulty was lower in Microrelin group (p=0.049). No significant difference in haemoglobin drop after operation was detected between two groups.
Conclusion: Our study showed that pretreatment with Microrelin before uterine resectoscopic myomectomy could lead to less surgical difficulties and subsequently lower operation time. The amount of haemoglobin (as an indicator of blood loss) was not affected with this intervention. Our study supports the beneficiary influence of GnRH agonists in outcomes of myomectomy.
Keywords
GnRH agonist, Myoma, Myomectomy, Clinical trial.
Introduction
Uterine leiomyomata are the most frequent benign tumour of uterus and is considered to be the most common cause of uterine surgery [1,2]. It has been previously reported that pretreatment with Gonadotropin Releasing Hormone (GnRH) analogs reduces the size of myomas and facilitate hysteroscopic surgery [3,4]. However, the numbers of randomized clinical trial confirming this beneficiary effect of GnRH analogs are so restricted. Some studies have supported the advantages of administration of GnRH analogs before operation since these medications have the potential to decrease the volume of distension fluid and the overall surgical time, but some other literatures have shown that these analogs may increase the recurrence of myomas because they make small myomas less visible during the surgery [5,6].
The growth of leiomyomas are stimulated by sex steroid hormones and therefore prescription of GnRH agonists leads to suppression of pituitary ovarian function which helps to temporary control of bleeding and correction of iron deficiency anaemia [2,7,8]. However, as these analogs may sometimes make the enucleation of myomas more difficult no definite recommendation of their administration has been made so far.
The purpose of this study was to evaluate and compare the clinical and surgical outcomes of uterine resectoscopic myomectomy with and without pretreatment with GnRH analogs. Here, we also tried to compare the difficulty of the operation with assessing the volume of the absorbed fluid and total operation time and also the level of satisfaction from surgeon’s view with and without pretreatment with GnRH analog.
Materials and Methods
Study design and participants
This investigation is a single-blinded randomized clinical trial with registration number: IRCT201408133950N3. Participants were premenopausal women selected for hysteroscopic myomectomy due to different complains related to uterine myomas.
Inclusion criteria: Age between 18-50 y (premenopausal women), patients who are candidate for surgical myomectomy, existence of myomatous uterus with sub-mucosal, intracavitarymyomas or myomas with less than 50% invasion into myometrium, less than 2 myomas with size range of 2-4 cm, using non-hormonal contraception methods and Body Mass Index (BMI) between 18 and 30.
Exclusion criteria: Previous history of endometrial, cervical or breast cancer, previous history of endometrial atypical hyperplasia or adenocarcinoma, history of severe coagulopathy, previous consumption of GnRH agonists to treat myomas, pregnancy, history of osteoporosis, liver dysfunction, alcohol consumption, drug abuse and necessity for urgent operation. Patients with a trial of embolization therapy in the preceding 2 y were excluded from the study.
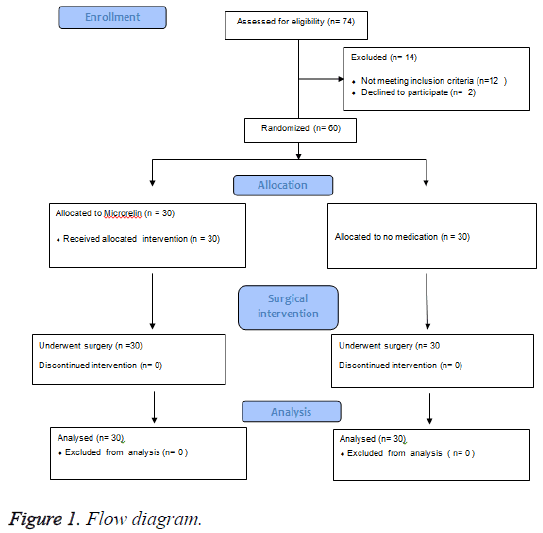
62 patients referred to gynaecology clinic were selected based on the inclusion and exclusion criteria and were randomly assigned to intervention and control group by using Permuted Balanced Block Randomization method. Adequate information about the trial was given to each patient before enrolment and written consent was obtained from each patient. The study was approved by the ethics committee of Tehran University of Medical Sciences. Data were collected from Jan to Dec 2014. Two patients refused to continue due to personal reasons, therefore 60 patients were finally allocated into intervention and control groups, 30 participants in each (Figure 1).
Intervention
Treatment group received 3.75 mg Microrelin (provided by Pooyesh Daru Corporation. Myomectomy was performed in both groups using monopolar resectoscope system. Procedures were scheduled in the early follicular phase for patients allocated to the control group and in the third or early fourth week after the second GnRHa administration in the treatment group. Resectoscopic resection was done in both groups using the slicing technique with a transcervical 9 mm, 0º fore oblique view, operative resectoscope, equipped with a unipolar electrosurgical loop (KARL STORZ GmbH and Co., Germany).
Measurements
Patient’s baseline characteristics including age, parity and history of reproductive disorders were indexed during face-toface interviews. The sizes of myomas were measured at the baseline (before treatment with GnRH analog) and before operation by ultrasound, which was performed by a single expert of radiology. Total surgical time and operation time were measured. The amount of absorbed fluid was calculated by reduction of the amount of fluid in the suction instrument from the total administered fluid. Circulatory estradiol, Luteinizing Hormone (LH) and the Follicle Stimulating Hormone (FSH) were measured at the baseline and then 2 and 12 weeks after administration of first dose of Microrelin.
Serum levels of LH and FSH were measured with immunometric ELIZA kit with detection range of 0.5 mIU/ml to 200 mIU/ml for LH and 0.6-100 mIU/ml for FSH. Estradiol parameter assay kit with sensitivity of 12.1 pg/mL was used to measure serum level of estradiol. The level of satisfaction from operation and the difficulty of the surgery were assessed according to surgeon’s view and were rated based on Visual Analog Scale (VAS) with the score range of 1-10.
Both the surgeon and the radiologist reporting the size of myomas before the treatment and the operation were not aware of the patient’s allocated groups.
Statistical analysis
All statistical analyses were performed using SPSS software, version 18.0 (SPSS Inc., Chicago IL, USA). Categorical values are reported by percentages and continuous data are expressed by mean ± standard deviation (SD). One-way analysis of variance (ANOVA) with test for linearity was used to compare means between groups. Pearson chi-square test was used to compare categorical data and Independent t-test with confidence interval of 95% was used to compare quantitative value of both groups. The value of p<0.05 was considered statistically significant.
Results
All patients were nulliparous. Table 1 shows the baseline characteristics of the patients in this clinical trial. The mean age in the Microrelin group was 32.10 ± 2.7 and 31.94 ± 2.4 in control group. No significant difference in the mean age was observed between two groups (p=0.82). History of reproduction disorders was detected in 9 (29%) patients in Microrelin group and in 8 (25.8%) participants in control group. Myomas were matched according to their topography in two groups. Cumulative topographic scores according to STEPW were 0.96 and 1.07 in agonist and control group respectively (p=0.12). There was no significant difference in the mean of pretreatment size of myomas between two groups (2.8 ± 0.79 cm and 1.9 ± 1.02 cm in the Microrelin and control groups, respectively). There was no statistically significant difference between the mean numbers of myomas between two groups. The mean size of myomas was significantly reduced in the Microrelin group in comparison with control group (P<0.0001). The mean size of myomas before operation was 1.9 ± 1.02 cm (range: 1.6-3.1 cm) in intervention group and 2.8 ± 0.79 (range: 2.4-3.4) in control group which illustrate the significant reduction of the size of the myomas with Microrelin administration.
| Category | Microrelin Group (n=30) |
Control Group (n=30) |
p value |
|---|---|---|---|
| Agea | 32.10 ± 2.7 | 31.94 ± 2.4 | 0.82 |
| History of reproductive disordersb | 9 (29%) | 8 (25.8%) | 0.13 |
| Myoma pretreatment sizea | 2.8 ± 0.79 | 1.9 ± 1.02 | 0.62 |
| Number of leiomyomataa | 1.3 ± 0.2 | 1.3 ± 0.4 | 0.55 |
| Myoma topographic scorec | 0.96 | 1.07 | 0.12 |
| Hemoglobin <10 g/dl the day before operationb | 3 (9.6%) | 4 (12.9%) | 0.06 |
Table 1: Baseline characteristics of the subjects in two groups.
The laboratory measurements showed haemoglobin (Hb) less than 10 g/dl in three (9.6%) patients in the Microrelin group and four (12.9%) in control group at the day before surgery. The day after operation, low levels of Hb (below 10 g/dl) were detected in 4 and 5 patients (12.9% and 16.1%) in intervention and control groups, respectively (Table 2). In the Microrelin group, the total surgical time was significantly lower (22.5 ± 3.1 min in the Microrelin group and 28.3 ± 3.7 min in control group) (Table 2).
| Outcome | Microrelin Group | Control Group | p value |
|---|---|---|---|
| Operation timea | 16.4 ± 2.5 b | 22.1 ± 2.4 | 0.0001 |
| Total Surgical Timea | 22.5 ± 3.1 | 28.3 ± 3.7 | 0.0001 |
| Administered Fluidc | 3398 ± 229 | 4100 ± 408 | 0.163 |
| Absorbed Fluidc | 374 ± 45 | 486 ± 65 | 0.045 |
| Level of Satisfactiond | 8.4 ± 0.6 | 7.8 ± 1.0 | 0.012 |
| Level of difficultyd | 4.6 ± 1.8 | 6.0 ± 2.9 | 0.049 |
| Hemoglobin<10 g/dl at the day after operatione | 4 (12.9) | 5 (16.1%) | 0.43 |
Table 2: Comparison of surgical outcomes between the two groups.
There was no significant difference in the levels of estradiol, LH and FSH at the baseline between two groups. However, concentrations of circulatory estradiol and LH were significantly reduced after 12 weeks of treatment with Microrelin in comparison with control group (P<0.0001). Similarly, the significant reduction of FSH occurred after 12 weeks in the Microrelin group in comparison with control group. (p<0.0001, Table 3).
| Serum hormone level | Baseline | Week 2 | Week 12 | p valuea |
|---|---|---|---|---|
| Serum estradiol levelb | 67.66 ± 10.23 | 75.87 ± 10.94 | 16.79 ± 8.56 | <0.0001c |
| Serum LH leveld | 6.77 ± 2.11 | 7.74 ± 2.95 | 2.05 ± 0.67 | <0.0001 |
| Serum FSH leveld | 6.74 ± 2.32 | 7.02 ± 2.65 | 2.95 ± 0.64 | <0.0001 |
Table 3: The amount of change in serum hormone levels after 2 and 12 weeks in the Microrelin group.
The satisfaction level from operation from surgeon’s view was significantly higher in the Microrelin group. Satisfaction level was measured based on VAS (scale: 1-10) and was 8.4 ± 0.6 in the Microrelin group and 7.8 ± 1.0 in the control group (p=0.01). Surgeon’s viewpoints about the difficulty level of the operation were also indexed based on VAS and was significantly lower in the treatment group (4.6 ± 1.8 and 6.0 ± 2.9 in the Microrelin and control groups, respectively. (p=0.049, Table 2).
Discussion
Although the advantages of administration of GnRH agonists before laparoscopic or abdominal excision of intramural or subserousal myomas have been demonstrated, still prescription of these agents has not been definitively recommended [9]. Dubuisson et al. showed that preoperative GnRH analogue results in a softening of the uterine myomas and facilitates morcellation [10]. However, Campo et al. illustrated that the operative time in patients who have received preoperative GnRH was higher than those who received no medication [9]. The results of Campo’s study are in controversy with our outcomes. Our investigation revealed significant reduced operation time and lower difficulty level from surgeon’s view n the group of patients submitted to preoperative GnRH therapy. Some other investigations have also reported that administration of preoperative GnRH analogs results in a more difficult identification of the cleavage plane and thus makes the surgical time longer [11-13]. In controversy with these reports, Perino et al. showed a decreased surgical time, bleeding, and volume of distension fluid for those patients to whom GnRH analogs had been administered in comparison to control group who received no medication which is in line with our results [5]. According to Campo et al. study in 2005, the cervical dilatation is harder and more time-consuming among those patients submitted to pre-operative GnRH analogs [13]. So, the reason for these reported controversies in surgical time can be differences in measuring surgical time (whether they have considered the duration of anaesthesia and cervical dilatation time or not). In fact, it seems that GnRH analogs reduce the size of myomas and thus facilitates surgical process and decreases the amount of bleeding. On the other hand, when considering the time that is required for cervical dilatation or the time that consumes in exploring the small-sized myomas for enucleating, pre-operative administration of GnRH analogs does not seem to be advantageous. In controversy with these results, our investigation showed reduced operation time both the surgical duration and total operation duration. Other operation features such level of satisfaction was significantly higher among the GnRH group compared with the control group.
Similar with previous reports that have shown that preoperative GnRH agonist increase the difficulties to identify and dissect the cleavage plan between leiomyoma and its pseudocapsule [14,15]. Vercellini et al. demonstrated that treatment with a GnRH agonist before abdominal myomectomy has no significant effect on intraoperative blood loss [16]. Here, we did not measure the volume of blood loss but the frequency of low Hb level (lower than 10 g/dl) the day before and the day after operation showed no significant difference between the Microrelin and control group. Our results are in line with Vercellini’s reports and it seems that pre-operative treatment with GnRH agonists does not have a beneficiary effect in reducing blood loss [16].
Uterine myomas are estrogen dependent tumours. Both receptors of estrogen and progesterone have been identified in these tumours [17]. Moreover, the estrogen secreted by myomas may reach a concentration to support its own growth [18]. It has been shown that GnRH agonists reduce estrogen levels to those occurring after menopause or after surgical oophorectomy [19]. In the present study we demonstrated an average reduction of 77% (16.7 pg/ml) in circulatory estradiol level after 12 weeks. GnRH agonists which initially stimulate the pituitary gland but continuous administration inhibits the secretion of gonadotropins as a result of receptor desensitization and/or down-regulation. Our investigation showed an increased level of estradiol after 2 weeks of treatment with GnRH agonists which is a result of the initial stimulation of pituitary glands. Similar to our results, Parsanezhad et al. demonstrated raised levels of estradiol after 2 weeks of GnRH analog administration [20]. The same phenomenon of hormone flare-up was also reported by Harding et al. who also demonstrated the probability of ovarian enlargement, especially when GnRH agonists are started at the early follicular phase [21]. Although increased levels of estradiol with treatment initiation can result in myoma growth, these consequences are usually transient. However, Friedman reported a case of rapid uterine enlargement during the first seven days of the administration of GnRH agonists [22].
In conclusion, our study shows that pretreatment with GnRH agonist before uterine resectoscopic myomectomy leads to less surgical difficulties and subsequently lower operation time. This study shows an average reduction of 33% (from 2.8 cm to 1.9 cm) in the sizes of uterus myomas with administration of GnRH agonist. Although, GnRH analogs led to more favourable surgery characteristics, the amount of Hb (as an indicator of blood loss) was not affected with this intervention.
Conclusion
In the present study, we tried to evaluate the effect of the administration of GnRH agonists (Microrelin 3.75 mg) on outcomes of resectoscopic myomectomy among premenopausal women with uterine myomas. A significant reduction of the mean myoma size (33%) was reached in the Microrelin group which, as a result, led to more favourable surgical outcomes. The duration of operation and the level of surgery difficulty from surgeon’s viewpoints were significantly lower in the Microrelin group. Moreover, the administration of these agents was associated with significant reduction in estradiol, LH and FSH levels after 12 weeks, though initially, a transient increase in these levels were detected by the end of the second week due to flare-up effect. Our study supports the beneficiary influence of GnRH agonists in the short-term. Longer follow-up studies, nonetheless, are required to evaluate long-term outcomes and the rate of recurrence.
References
- Fedele L, Bianchi S, DortaM, Brioschi D, Canotti F, Vercellini P. Transvaginal ultrasonography versus hysteroscopy in the diagnosis of uterine submucousmyomas. Obstet Gynecol 1991; 77: 745-748.
- Lethaby A, Vollenhoven B, Sowter M. Pre-operative GnRH analogue therapy before hysterectomy or myomectomy for uterine fibroids. Cochrane Library 2007.
- Donnez J, Schrurs B, Gillerot S, Sandow J, Clerckx F. Treatment of uterine fibroids with implants of gonadotropin-releasing hormone agonist: assessment by hysterography. Fertil Steril 1989; 51: 947-950.
- Mencaglia L, Tantini C. GnRH agonist analogs and hysteroscopic resection of myomas. Int J Gynaecol Obstet 1993; 43: 285-288.
- Perino A, Chianchiano N, Petronio M, Cittadini E. Role of leuprolide acetate depot in hysteroscopic surgery: a controlled study. Fertil Steril 1993; 59: 507-510.
- Fedele L, Vercellini P, Bianchi S, Brioschi D, Dorta M. Treatment with GnRH agonists before myomectomy and the risk of short-term recurrence. Br J Obstet Gynaecol 1990; 97: 393-396.
- Stewart EA. Uterine fibroids. Lancet 2001; 357: 293-298.
- Marianna De F, Stefania S, Massimo M, Chiara M, Luigi I, Francesca C, Andrea Di L. Leiomyoma pseudocapsule after pre surgical treatment with gonadotropin releasing hormone agonists: Relationship between clinical features and immunohistochemical changes. Eur J Obstet Gynecol Reprod Biol 2009; 144: 44-47.
- Campo S, Garcea N. Laparoscopic myomectomy in premenopausal women with and without preoperative treatment using gonadotrophin-releasing hormone analogues. Hum Reprod 1999; 14: 44-48.
- Dubuisson JB, Chapron C, Chavet X. Laparoscopic myomectomy: where do we stand? Gynaecol Endosc 1995; 4: 83-86.
- Acie´n P, Quereda F. Abdominal myomectomy: results of a simple operative technique. Fertil Steril 1996; 65: 41-51.
- Deligdisch L, Hirschmann S, Altchek A. Pathologic changes in gonadotropin releasing hormone agonist analogue treated uterine leiomyomata. Fertil Steril 1997; 67: 837-841.
- Campo S, Campo V, Gambadauro P. Short-term and long-term results of resectoscopic myomectomy with and without pretreatment with GnRH analogs in premenopausal women. Acta Obstet Gynecol Scand 2005; 84: 756-760.
- Dubuisson JB, Fauconnier A, Fourchotte V, Babaki-Fard K, Coste J, Chapron C. Laparoscopic myomectomy: predicting the risk of conversion to an open procedure. Hum Reprod 2001; 16: 1726-1731.
- Beyth Y. Gonadotropin-releasing hormone analog treatment should not precede conservative myomectomy. Fertil Steril 1990; 53: 1871-88.
- Vercellini P, Trespìdi L, Zaina B, Vicentini S, Stellato G, Crosignani PG. Gonadotropin-releasing hormone agonist treatment before abdominal myomectomy: a controlled trial. Fertil Steril 2003; 79: 1390-1395.
- Brandon DD, Erickson TE, Keenan EJ, Strawn EY, Novy MJ, Burry KA. Estrogen receptor gene expression in human uterine leiomyomata. J Clin Endocrinol Metab 1995; 80: 1876-1881.
- Bulun SE, Imir G, Utsunomiya H, Thung S, Gurates B, Tamura M. Aromatase in endometriosis and uterine leiomyomata. J Steroid Biochem Mol Biol 2005; 95: 57-62.
- Perry CM, Brogden RN, Goserelin. A review of its pharmacodynamics and pharmacokinetic properties, and therapeutic use in benign gynaecological disorders. Drugs 1996; 51: 319-346.
- Parsanezhad ME, Azmoon M, Alborzi S, Rajaeefard A, Zarei A, Kazerooni T, Frank V, Schmidt EH. A randomized, controlled clinical trial comparing the effects of aromatase inhibitor (letrozole) and gonadotropin-releasing hormone agonist (triptorelin) on uterine leiomyoma volume and hormonal status. Fertil Steril 2010; 93: 192-198.
- Harding SG, Pesce A, McMillan L. Symptomatic ascites complicating GnRH analogue use for myoma shrinkage. Br J Obstet Gynaecol 1993; 100: 1054-1056.
- Friedman AJ. Acute urinary retention after gonadotropin-releasing hormone agonist treatment for leiomyomata uteri. Fertil Steril 1993; 59: 677-678.