Research Article - Biomedical Research (2017) Volume 28, Issue 15
The investigation of blood drug levels and course of acetaminophen poisoning due to application time
Abdullah Algin1* and Mehmet Ozgur Erdogan21Emergency Medicine, Adiyaman University Training and Research Hospital, Adiyaman, Turkey
2Emergency Medicine, Haydarpasa Numune Training and Research Hospital, Istanbul, Turkey
- *Corresponding Author:
- Abdullah Algin
Emergency Medicine
Adiyaman University Training and Research Hospital
Adiyaman, Turkey
Accepted date: June 27, 2017
Abstract
Background: Analgesics are reported to be responsible for 11.3% of the poisoning events in the 20 y data obtained from the Poison Counseling Centers. In this study, we aimed to investigate the relationship between epidemiological characteristics of patients, drug levels detected in acetaminophen poisoning and course of the disease due to timely, late admission and severity of intoxication.
Method: Data of the patients admitted to emergency department of Haydarpasa Numune Training and Research Hospital between Jan 2013 and Dec 2015 were retrospectively analyzed with 142 (79 f/63 m) timely and 48 (29 f/19 m) late applicants. Liver enzymes, INR, platelets, venous blood gas and demographics of these two groups were evaluated.
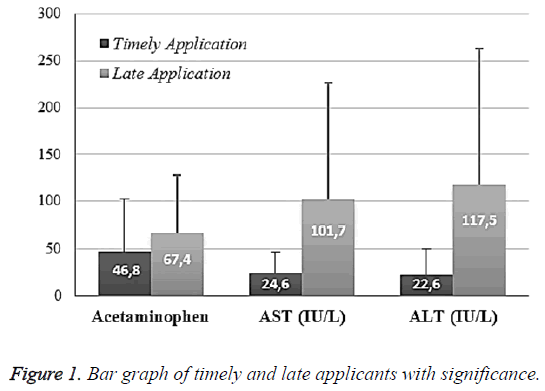
Results: The average acetaminophen value was 46.8 ± 55.1 in TA and 67.4 ± 60.1 in LA (p: 0.039; p<0.05). The average aspartate aminotransferase (AST) level was 24.6 ± 22.2 in timely applicants while the average AST level was 101.7 ± 125.9 in late applicants (P: 0.00011). The average alanine aminotransferase (ALT) level was 22.6 ± 27.5 in timely applicants while it was 117.5 ± 146.8 in late applicants (P: 0.00012). There was no correlation relation among those. The mean INR value was 1.11 ± 0.26 in timely applicants while it was 1.08 ± 0.13 in late applicants (p: 0.471). Platelet level distribution was 273.6 ± 63.6 in timely applicants while it was 279.4 ± 69.9 in late applicants (p: 0.609). The venous blood gas pH value of timely applicants was 7.38 ± 0.053 whereas the venous blood gas pH value was 7.37 ± 0.0518 in late applicants (p: 0.43). There was a positive significant correlation between acetaminophen levels and blood gas pH value (p: 0.000001; r: 0.445).
Conclusion: Late admission of the patient who has been admitted to emergency services due to acetaminophen poisoning is related to higher levels of acetaminophen, higher hospitalization rates, and lower discharge rates. It is useful to apply more careful and concerned approach to the patients especially if they apply late, and it will contribute to the treatment of patients.
Keywords
Acetaminophen poisoning, Paracetamol intoxication, Application time.
Introduction
Poisoning has been one of the major problems affecting human health since ancient times and the frequency varies according to the cultural and socio-economic structure of societies [1]. It is known that poisonings for suicidal purposes constitute 95% of all cases of poisoning, with more frequent in women. Drugs being used for preventing ache are the leading cause of poisoning [2].
In Turkey, there are 150.000-200.000 cases of acute poisoning every year; 58.6% of them are caused by drugs [3]. In the United States, analgesics are reported to be responsible for 11.3% of the poisoning events in the 20 y data obtained from the Poison Counselling Centers [4]. It was reported that acetaminophen accounts for 47.1%, other Non-Steroidal Anti- Inflammatory Drugs (NSAID) account for 36.1%, salicylates account for 7.7% and opioids account for 8.4% of analgesics poisoning [5].
Acetaminophen is a pain reliever with analgesic, antipyretic and weak anti-inflammatory effects, and poisonings related with this drug are very common in our country [3]. When taken from the oral route, acetaminophen is absorbed quickly and the effect starts timely [6]. Plasma level reaches the most effective level within 30-60 min. Absorption is reduced by nutrients [7]. After one dose the analgesic effect lasts for 3-4 h. The half-life at the usual dose is 2.4 h; it can be up to 7.3 h in overdose due to nonlinear elimination kinetics. Due to the low level of gastrointestinal toxicity, full doses can be combined with low doses of anti-inflammatory analgesics [6-8].
In this study, we aimed to investigate the relationship between epidemiological characteristics of patients, drug levels detected in acetaminophen poisoning and the course of the disease due to timing, late admission and severity of intoxication.
Materials and Methods
This retrospective observational study complied with the guidelines of the Declaration of Helsinki and was approved by the Public Ethics Committee of the Haydarpasa Numune Training Hospital, which is a tertiary referral center located in the Istanbul City. We reviewed the data of the patients admitted to the emergency department of Haydarpasa Numune Training and Research Hospital between Jan 2013 and Dec 2015. Because this study involved a retrospective review of existing data, specific informed consents were not obtained from the patients. However, informed consent of the risks of acute acetaminophen poisoning and all treatment modalities was obtained from all patients upon their initial admission.
All demographic, clinical and laboratory data were collected and the mortality rate was analysed. Liver enzymes, INR, platelets, venous blood gas and demographics of two groups were evaluated through the timely and late applicants. The diagnoses of acetaminophen intoxication were based on clinical, physical and laboratory examinations and confirmed by plasma acetaminophen levels (fluorescence polarization immunoassay). Patients were excluded if they were younger than 18 y or did not have detectable acetaminophen levels in the blood despite a positive history of ingestion or if they had major systemic comorbidities, such as cancer or heart, lung, renal or liver diseases.
The protocols that were used to treat patients included gastric lavage with large amounts of normal saline, which was followed by the infusion of 1 g/kg activated charcoal and 250 ml magnesium citrate through a nasogastric tube. Magnesium citrate was used to prevent constipation after the charcoal administration. The use of the N-acetyl cysteine antidote was determined with toxicity estimations from the Rumack- Matthew nomogram for single acute acetaminophen overdose/ ingestion.
Data was analysed with Statistical Package for Social Sciences (SPSS) version 17. The continuous variables are expressed as means and standard deviations, and categorical variables are expressed as numbers with percentages in brackets. All data were tested for the normality of the distribution and the equality of standard deviations before the analysis. Parametric continuous data was analysed by student-T test, whereas nonparametric data were analysed by Kolmorov Similgrow test. Ki-Square test was used to analyse categorical data. P<0.05 results were accepted as statistically significant.
Results
While the mean age of 142 timely applicants (TA’s) (79 females/63 males) was 26.4 ± 10.1 y, the mean age of 48 late applicants (LA’s) (29 females/19 males) was 24.8 ± 8.4. There were no statistically significant differences in terms of age (p: 0.294) and gender (p: 0.615) distribution between timely admitted patients and those with late admission.
The mean acetaminophen value was 46.8 ± 55.1 in TA and 67.4 ± 60.1 in LA (p: 0.039; p<0.05). The mean Aspartate Aminotransferase (AST) level was 24.6 ± 22.2 in timely applicants while the AST level was 101.7 ± 125.9 in late applicants (p: 0.00011). The Alanine Aminotransferase (ALT) level was 22.6 ± 27.5 in timely applicants while it was 117.5 ± 146.8 in late applicants (p: 0.00012). There was no correlation among those (Figure 1).
The mean INR value was 1.11 ± 0.26 in timely applicants while it was 1.08 ± 0.13 in late applicants (p: 0.471). Platelet level distribution was 273.6 ± 63.6 in timely applicants while it was 279.4 ± 69.9 in late applicants (p: 0.609). The venous blood gas pH value of timely applicants was 7.38 ± 0.053 whereas the venous blood gas pH value was 7.37 ± 0.0518 in late applicants (p: 0.43). There was a positive significant correlation between acetaminophen levels and blood gas pH value (p: 0.000001; r: 0.445) (Table 1).
| Timely application (n:142) | Late application (n:48) | P | |
|---|---|---|---|
| Age (y) | 26.4 ± 10.1 | 24.83 ± 8.4 | 0.294 |
| Gender (f/m) | 76/63 | 29/19 | 0.615 |
| Acetaminophen | 46.8 ± 55.1 | 67.4 ± 60.1 | 0.039 |
| AST (IU/L) | 24.6 ± 22.2 | 101.7 ± 125 | 0.00011 |
| ALT (IU/L) | 22.6 ± 27.5 | 117.5 ± 146 | 0.00012 |
| INR | 1.11 ± 0.26 | 1.08 ± 0.13 | 0.471 |
| PLT | 273.6 ± 63 | 279.4 ± 69 | 0.609 |
| pH | 7.38 ± 0.052 | 7.37 ± 0.051 | 0.434 |
Table 1: Test results of timely and late applicants with significance.
In the assessment among patients in terms of discharge from emergency service, the rates of discharge from emergency service in LAs (23/48; 48%) were less than those in TA’s (102/142; 72%). The late applicants had statistically higher internal branch hospitalization rates (p: 0.004).
N-Acetyl Cysteine (NAC) treatment was applied to 102 of 142 timely applicants, while NAC treatment was applied to 35 of 48 late applicants in emergency service. There was no statistically significant difference in the onset of NAC treatment in emergency service (p>0.05). Nasogastric catheter insertion and gastric lavage were applied to 114 of 142 timely applicants in emergency service, while they were applied to 23 of 48 late applicants in emergency service. Nasogastric catheter insertion and gastric lavage were found to be statistically higher in timely applicants (p: 0.00003).
Discussion
In studies conducted, it was reported that most of the analgesic poisonings occur with acetaminophen [4]. Acetaminophen is a pain reliever with analgesic, antipyretic and weak antiinflammatory effects and is widely preferred in our country [9]. Similar to the poisoning rates in the world, this drug-related poisoning is also common in Turkey [3]. The results of this study are very valuable in terms of evaluating the epidemiological characteristics of patients in our country for acetaminophen-induced poisoning and have importance for evaluating timely and late application of acetaminophen poisoning and its course.
Acute liver failure or fulminant hepatic insufficiency is an important and serious complication of acute acetaminophen poisoning [7]. At therapeutic doses of acetaminophen, 90% is metabolized to sulphate and glucuronide conjugates in the liver and excreted into the urine [10,11]. Very little is metabolized by the cytochrome P450 CYP2E1 subtype in the liver. In this way, also known as the mixed-function oxidase pathway, Nacetyl- p-benzoquinonimine (NAPQI), a toxic, highly reactive and electrophilic intermediate product is formed [12]. NAPQI is arylated and binds covalently to hepatocyte macromolecules, leading to oxidative damage and hepatocellular necrosis [5]. At normal doses, NAPQI rapidly converts to non-toxic cysteine and mercaptan compounds as a conjugate of hepatic glutathione and is excreted in the urine [13]. At toxic doses, when the sulphate and glucuronide pathway is saturated, it causes an increase in acetaminophen fraction, which is metabolized via the cytochrome P450 enzymes [14]. When the liver glutathione stores are reduced by 70%, NAPQI accumulates and leads to liver damage. The severity of the resulting liver necrosis depends on the activity of the glutathione stores, the cytochrome P450 system, and the glucuronization system [10,12].
Acute liver failure has a serious mortality, but the causes vary from country to country and/or time interval [6]. There are no sufficient data on the etiological studies of acute liver failure in our country. According to the study of Kayaalp et al. drugrelated conditions were shown as 19% among the reasons of adult acute liver failure in our country [15]. In our study, only 3 of 190 patients developed acute hepatic failure as a result. Our study group demonstrated acute acetaminophen poisoning as one of the reasons for acute hepatic failure in our country. However, one of the major known causes of acute hepatic failure in our country is hepatic viruses. In some studies, it appears to be an important cause of acute liver failure at 30% [16]. In our study, due to the low number of developing acute hepatic failure, it could not be found that acetaminophen intoxication causes acute hepatic failure more frequently in timely applicants and/or late applicants.
There was no difference in age distribution between acetaminophen intoxication of timely applicants and acetaminophen intoxication of late applicants in the current study. No difference was observed in gender distribution between acetaminophen intoxication of timely applicants and acetaminophen intoxication of late applicants in our study. This is an important data indicating that the distribution between the two groups is normal, and at the same time suggesting that age and gender are not important factors when applying to hospital after people attempt suicide. The idea of going to a hospital after a suicide attempt is a multifactorial process and depends on the psychological state of the person, the suicide method and the effect of people who are in touch with. Age and gender were not found to be important factors for hospital admission time.
INR and Platelet values do not differ in timely and late application [17]. This is because of the fact that INR values begin to increase at the end of the second month in cases of acetaminophen-related intoxication [18]. Our study is based on the 4th h as a timely and late application. The application of any patients after 1st day was not registered as a late application. For this reason, the INR value was not registered into the study after 24 h. According to our study, the increases in INR levels are not expected according to this level.
The treatment decision for acute acetaminophen overdose is usually made using the Rumack-Matthew Nomogram [19]. Generally, NAC treatment should be started when the concentration of plasma acetaminophen in a blood sample taken at 4th h exceeds 150 mg/l [20]. It is not possible to start NAC therapy on time by using the Rumack-Matthew Nomogram in patients with late applications [21]. Therefore, levels of acetaminophen that have reached toxic levels as a result of late admission have a higher risk of acute hepatic failure in patients. In our study, late applicants showed significantly higher levels of AST and ALT compared to timely applicants. Plasma levels of acetaminophen in late applicant patients were also significantly higher than in timely applicant patients.
There are many reasons to be able to explain higher levels of blood plasma acetaminophen levels in late applicant patients, such as not being able to apply gastric lavage and nonactivated carbon delivery, the patient being more stable for suicide, and the longer waiting time to come to the hospital. When looked at the distribution of acetaminophen levels in late applicant patients, it was observed that blood levels were very low in the major part of the patients, while blood levels reached toxic levels in the other part. As the reason for this, it may be considered that some patients may receive low doses of acetaminophen, reflect the situation as a suicide attempt and obtain secondary gains, and be brought to the hospital with the persistence of their relatives. The patient’s stability in the suicide attempt, taking more medication, staying away from medical help, not seeking for medical help and to not treating with active carbon with gastric lavage may be considered as a reason in order to explain the high levels of acetaminophen in late applicant patients.
When the timely and late applicant patients with intoxication as a result of acetaminophen use are compared, it is observed that gastric lavage was applied more to the timely applicant patients compared to late applicant patients. Timely admission gives the opportunity to intervene in precious hours for gastric lavage. Late applicants cannot be benefited from the gastric lavage for drug elimination and they expose to drug doses that can be removed from the body. When looked at the late applicant patient group, hospital admission rates were significantly higher compared to timely applicants, and those discharged from emergency service were lower than timely applicants. A late applicant patient is faced with more risks of acetaminophen poisoning, and NAC treatment cannot be started in time, no active charcoal can be given by gastric lavage. Such situations result in more hospitalization and less discharge from the emergency service of late applicants. Despite intravenous administration of NAC treatment in emergency service in our study, no NAC-related anaphylactic reaction was observed.
Phase-1 of acetaminophen poisoning usually involves the first 24 h of symptoms [9]. Symptoms usually include nausea, vomiting, anorexia, abdominal pain, lethargy, and diaphoresis. In this stage, elevation in hepatic levels and deterioration of blood values are not expected [22]. However, according to our study, results of liver function tests in the first 24 h were significantly higher in late applicants compared to timely applicants [11]. This situation is related with late-onset NAC treatment, not being able to treat with activated carbon through gastric lavage and much higher blood plasma acetaminophen levels in late applicants [23]. Hepatotoxicity is rarely seen below 12 g and is usually found in doses above 10 g. According to the Rumack-Matthew Nomogram in phase 1, the initiation of treatment according to the 4th h is recommended; but it is clear that late application in phase 1, for example at 12 or 18 h, includes differences with timely application, and the duration of the treatment and the severity of both the treatment and the liver effects [21,24]. A statistical difference was observed in both levels of acetaminophen and liver enzymes between the timely applicant and late applicant patients in phase-1 in our study.
Phase-2 includes the first 24-72 h in acetaminophen poisoning. Symptoms are so low as to disappear [6]. Liver enzymes, bilirubin, PTZ and INR values are at abnormal levels. Some patients complain of right upper quadrant pain, and when they have a liver biopsy, centrilobular necrosis is observed [18]. In phase-3, nausea and vomiting recur. The somnolence associated with the neurological system, confusion and even coma findings appear. At this phase hepatocellular damage is severe and liver enzymes can rise to over 10000 IU/lt. Phase-4 is the recovery phase and 70% of patients with acute hepatic failure can reach to this phase [1,14,22].
Acetaminophen is a drug that can be easily accessed worldwide with sales over the counter. The fact that toxic doses of this drug are sold in a single package increases the acetaminophen-related mortality. Selling in quantities that are not toxic to adults in a single package by reducing the packet doses of this drug, at least preparing low-dose packages for sales over the counter may contribute to reducing drug-related mortality.
Conclusion
Late admission of patients who admitted to emergency services due to acetaminophen poisoning is related to higher levels of acetaminophen, higher hospitalization rates, and lower discharge rates. It is useful to apply more careful and concerned approach to the patients, especially if they apply late, and it will contribute to the treatment of patients.
References
- Heard K, Green J. Acetylcysteine therapy for acetaminophen poisoning. Curr Pharma Biotechnol 2012; 13: 1917-1923.
- Adibelli D, Olgun S. Knowledge, attitude and behavior of health college students related to drug abuse. Ulutas Med J 2016; 2: 90-100.
- Oguz M, Mihciokur H. Environmental risk assessment of selected pharmaceuticals in Turkey. Environ Toxicol Pharmacol 2014; 38: 79-83.
- Hodgman MJ, Garrard AR. A review of acetaminophen poisoning. Crit Care Clin 2012; 28: 499-516.
- Washio M, Inoue N. The risk factors of death from the acetaminophen poisoning with antipyretic-analgesic drugs in Japan. Fukuoka Igaku Zasshi 1997; 88: 352-357.
- Stevens A. Paracetamol recall: a natural experiment influencing analgesic poisoning. Med J Aust 2002; 176: 562.
- Bateman DN, Dear JW. Limitations of AST/ALT ratio in paracetamol poisoning. Clin Toxicol 2015; 53: 580.
- Park BK, Dear JW, Antoine DJ. Paracetamol (acetaminophen) poisoning. BMJ Clin Evid 2015.
- Tsui MS. Management of acute paracetamol poisoning. Hong Kong Med 2015; 21: 388.
- Serjeant L, Evans J, Sampaziotis F, Petchey WG. Haemodialysis in acute paracetamol poisoning. BMJ Case Rep 2017.
- Kazemifar AM, Hajaghamohammadi AA, Samimi R, Alavi Z, Abbasi E, Asl MN. Hepatoprotective property of oral silymarin is comparable to N-acetyl cysteine in acetaminophen poisoning. Gastroenterol Res 2012; 5: 190-194.
- Kalsi SS, Wood DM, Waring WS, Dargan PI. Does cytochrome P450 liver isoenzyme induction increase the risk of liver toxicity after paracetamol overdose? Open Access Emerg Med 2011; 3: 69-76.
- McGill MR, Jaeschke H. Metabolism and disposition of acetaminophen: recent advances in relation to hepatotoxicity and diagnosis. Pharma Res 2013; 30: 2174-2187.
- Villeneuve JP, Pichette V. Cytochrome P450 and liver diseases. Curr Drug Metab 2004; 5: 273-282.
- Kayaalp C, Ersan V, Yılmaz S. Acute liver failure in Turkey: A systematic review. Turk J Gastroenterol 2014; 25: 35-40.
- Özçay F, Eda Karadağ-Öncel ZB, Canan O, Moray G, Haberal M. Etiologies, outcomes, and prognostic factors of paediatric acute liver failure: A single centre’s experience in Turkey. Turk J Gastroenterol 2016; 27: 450-457.
- Shalansky S, Lynd L, Richardson K, Ingaszewski A, Kerr C. Risk of warfarin-related bleeding events and supratherapeutic international normalized ratios associated with complementary and alternative medicine: a longitudinal analysis. Pharmacotherapy 2007; 27: 1237-1247.
- Brown D. Potentiation of warfarin by acetaminophen. J Fam Pract 1998; 46: 456-457.
- Vassallo S, Khan AN, Howland MA. Use of the Rumack-Matthew nomogram in cases of extended-release acetaminophen toxicity. Ann Intern Med 1996; 125: 940.
- Schwarz ES, Mullins ME. Introduction of an N-acetylcysteine weight-based dosing chart reduces prescription errors in the treatment of paracetamol poisoning: Reply. Emerg Med Aust 2013; 25: 285-286.
- Fukumoto M. Are nomograms available for the treatment of acetaminophen poisoning? Limitation of Rumack & Matthew nomogram for evaluation of hepatotoxicity. Japanese J Toxicol 2010; 23: 111-115.
- Lucyk SN, Hoffman RS, Nelson LS, Fuentes M, Tavangarian K. Challenges with AST/ALT ratio in acetaminophen poisoning. Clin Toxicol 2015; 53: 786.
- Heard K, Rumack BH, Green JL, Bucher-Bartelson B, Heard S, Bronstein AC. A single-arm clinical trial of a 48 h intravenous N-acetylcysteine protocol for treatment of acetaminophen poisoning. Clin Toxicol 2014; 52: 512-518.
- White SJ, Rumack BH. The acetaminophen toxicity equations: “solutions” for acetaminophen toxicity based on the Rumack-Matthew nomogram. Ann Emerg Med 2005; 45: 563-564.