- Biomedical Research (2014) Volume 25, Issue 4
The inhibitory activity of nutmeg essential oil on GABAA ?1?2?2s receptors.
Ahmad Tarmizi Che Has1, Mohammad Rafiqul Islam1,2, Igor Baburin3, Steffen Hering3, Hasnah Osman4, Habsah Mohamad5 and Jafri Malin Abdullah1*1Centre for Neuroscience Services and Research, Universiti Sains Malaysia, Jalan Hospital Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia Department of Neurosciences, School of Medical Sciences, Universiti Sains Malaysia, Jalan Hospital Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia Hospital Universiti Sains Malaysia, Jalan Hospital Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan
2Department of Anatomy and Histology, Bangladesh Agricultural University, Bangladesh
3Department of Pharmacology and Toxicology, University of Vienna, Austria
4School of Chemical Sciences, Universiti Sains Malaysia, 11800 Minden, Penang, Malaysia
5Faculty of Sciences and Technology, Universiti Malaysia Terengganu, 20130 Kuala Terengganu, Terengganu, Malaysia
- *Corresponding Author:
- Jafri Malin Abdullah
Centre for Neuroscience Services and Research
Universiti Sains Malaysia, Jalan Hospital Universiti Sains
Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia
Accepted date: August 11 2014
Abstract
The inhibitory effects of nutmeg essential oil and three components of this oil at different concentrations were tested by GABA-induced chloride current (I GABA) stimulation through gamma-aminobutyric acid type A (GABAA) receptors comprising of α1β2γ2s subtype using the two-microelectrode voltage clamp technique on Xenopus laevis oocytes. The essential oil of the kernels of Myristica fragrans was extracted by steam distillation and was analysed by gas chromatography-mass spectrometer (GC-MS) technique. A total of 16 compounds were isolated from the essential oil. The essential oil showed promising activity by enhanced I GABA up to 100% of stimulation when tested at 100 nL/mL. For dose dependent activity, α-terpineol and myristicin showed statistical significant potentiation of I GABA. α-Terpineol was found to induce an enhancement of I GABA modulation (EC5-10) by 229.6±23.8% and 326.3±43.8%, at 100 μM and 500 μM, respectively. Myristicin induced an enhancement up to 237.6±35.1% at 500 μM. In contrast, limonene exhibited weak modulation compared to GABA controls at the same concentration. The essential oil and its two components exhibited promising inhibitory activity by enhancement of I GABA that provides scientific evidence for its traditional use in the treatment of epilepsy.
Keywords
Myristica fragrans, α-Terpineol, gas chromatography-mass spectrometer (GC-MS), GABAA receptor, epilepsy
Introduction
Myristica fragrans is a tropical plant from the family of Myristicaceae. The fruit of this plant is well known as nutmeg, a yellow colour fruit that almost looks like an apricot. Nutmeg has a characteristic pleasant fragrance and has a slightly warm taste. It is used to flavour many kinds of baked goods, confections, pudding, meats, sausages, sauces, vegetables and beverages [1]. The pharmacological activities of nutmeg mainly exist in its essential oil fraction [2]. Nutmeg oil possesses a wide array of pharmacological actions including analgesic [3], antifungal [4], antimicrobial [5], anti-inflammatory [6], as well as hepatoprotective activity [7]. Different methods of nutmeg extraction showed different activities on the central nervous system. The chloroform extract of nutmeg inhibited the carrageenan-induced rat and produced a reduction in writhings induced by acetic acid in mice [6], the trimyristin and the acetone insoluble fraction of the hexane extract of nutmeg seed demonstrated anxiogenic and antidepressant activities in mice [3,8]. Nutmeg oil has shown the anticonvulsant activity against pentylenetetrazole (PTZ)-induced seizure and hind limb tonic extension phase in the maximal electric shock (MES) seizure test [2]. Recently, we found the dose dependent antiepileptic effects of α-terpineol, a major component of nutmeg oil, on genetic absence epileptic rat from Strasburg (GAERS) model [9].
Novel antiepileptic drugs acting selectively through the GABA-ergic system, such as Tiagabine, inhibits neuronal and glial uptake of GABA whilst Vigabatrin increases the synaptic concentration of GABA by inhibiting GABA-aminotransferase. Gabapentin also decreases influx of calcium ions into neurons via a specific subunit of voltage-dependent calcium channels. Another novel antiepileptic drug, Lamotrigine and Oxcarbazepine are mainly associated with an inhibition of voltage-dependent sodium channels. Conventional antiepileptic drugs generally inhibit sodium currents (carbamazepine, phenobarbital, phenytoin, valproate) or enhance GABA-ergic inhibition (benzodiazepine, phenobarbital, valproate) [10]. Unfortunately, the entire available antiepileptic drugs are synthetic molecules associated with side effects and approximately 30% of the patients continue to have seizures with this therapy [11]. Consequently, it is essential to design and explore the novel antiepileptic agents; which have the characteristics of being effective as well as safe. Nowadays, many people from the developing countries are using herbal medicines to treat epilepsy [12]. Therefore, the idea of managing epilepsy by using carefully analyzed and well-studied herbal medicines would be significant and advisable.
GABA exerts its physiological actions by activating the ionotropic GABAA receptors, which are the member of super family ligand-gated ion channels and the most widely distributed inhibitory receptor in the mammalian CNS. A total of 16 different GABAA receptor subunits have been identified in human. These inhibitory receptors are pentameric transmembrane protein complexes that form an intrinsic anion channel permeable to chloride ions [13-15]. The most abundant GABAA receptor subtype consists of 2α1, 2β2, and 1γ2 subunits [16,17]. GABAA channels are modulated by many drugs, used clinically to treat anxiety, insomnia, panic disorders, and epilepsy [18,19]. Many natural products modulating GABAA receptors include valerenic acid, a constituent of Valerian [20], piperine from Piper nigrum [21] and osthol and cnidilin (substances in the roots of Angelica pubescens) [22]. Therefore, the present study aimed to investigate the individual GABAA receptor modulator activities that expressed in Xenopus laevis oocytes of the different components of nutmeg essential oil.
Materials and Methods
Plant materials
Nutmegs were collected from Balik Pulau, Penang Island in Malaysia. About 8 kg of the dried kernels of the ripe seeds of Myristica fragrans (nutmeg) were used in this study. A voucher specimen was deposited at the Faculty of Sciences and Technology, Universiti Malaysia Terengganu (catalogue number; UMT 1001/170809).
Extraction and isolation
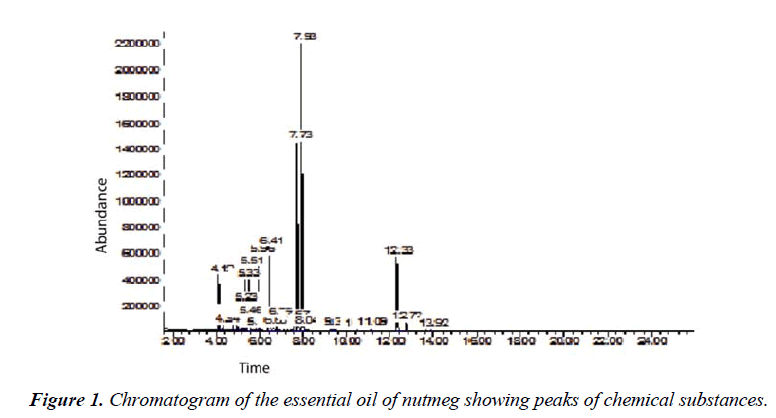
The kernels of nutmeg were chopped into small pieces and placed carefully into the water distiller up to the appropriate level. Distilled water was poured into the water distiller up to half full. At the end of the opening part, a small vial filled with hexane solvent was connected into the distiller. The boiling process takes approximately 6 to 8 hours. Once the water distiller was cooled, the mixture of hexane solvent containing essential oils in a small vial was concentrated using rotary evaporator to remove the waste of hexane. Then the pure essential oil of nutmeg was stored in a refrigerator at 8 °C. Structural elucidation of the compound was carried out by using Gas Chromatography-Mass Spectrometer (GC-MS; Agilent, Atlanta, GA, USA) with HP-5MS column (diameter 0.25 mm, length 30 m, film thickness 0.25 μm) and helium gas as the carrier. The inlet temperature of gas chromatography was set to 280 °C with split-less mode. A 1 μL of sample was injected into the GC-MS system. Gas flow was set to 1.2 mL/min. The initial oven temperature was 70 °C, increased to 280 °C and trap low temperature at –30 °C, and the final temperature was maintained for 18 min. According to Goren [23] the automatic peak detection and mass spectrum was performed for a total runtime of 26 min. During this period, a total of 16 compounds were detected in nutmeg oil (Table. 1 and Fig. 1).
Expression of GABAA receptors
The healthy oocytes of Xenopus laevis from stage V to VI were selected and cRNAs were injected as previously described by Khom et al. (2006). Female Xenopus laevis (NASCO) were anesthetized by exposing them for 15 min to a 0.2% MS-222 (3- aminobenzoic acid ethyl ester methane sulfonate, Sigma) solution before surgically removing parts of ovaries. Follicle membranes from isolated oocytes were enzymatically digested with 2 mg/mL collagenase from Clostridium histolyticum (Type 1A, Sigma). Synthesis of capped run-off poly (A+) cRNA transcripts was obtained from linearized cDNA templates (pCMV vector). One day after enzymatic isolation, the oocytes were injected 50 nL of DEPC-treated water (Sigma) containing different cRNAs at a concentration of approximately 300-3000 pg/nL/subunit. The amount of injected cRNA mixture was determined by means of a NanoDrop ND-1000 (Kisker Biotech). Rat cRNAs were mixed in a ratio of 1:1:10 to favour the incorporation of gamma subunit in α1β2γ2s receptors. Then the oocytes were incubated at 18°C in ND96 solution [24]. Voltage clamp measurements were performed between days 1 and 5 after cDNA injection.
Two-microelectrode voltage clamp studies
Electrophysiological experiments were performed by the two-microelectrode voltage clamp method. The amplifier used was a TURBO TEC 03X amplifier (NPI electronic, Tamm, Germany) at a holding potential of – 70 mV and pCLAMP 10 data acquisition software (Molecular Devices, USA). Currents were low-passfiltered at 1 kHz and sampled at 3 kHz. The ND96 bath solution contained 90 mM NaCl, 1 mM KCl, 1 mM MgCl2, 1 mM CaCl2 and 5 mM HEPES (pH 7.4).Electrode filling solution contained 2 M KCl. Oocytes with maximal current amplitudes > 3 μA were discarded to exclude voltage clamp errors.
Fast solution exchange during I GABA recordings
The test solutions (100 μL) were applied to the oocytes at a speed of 300 μL/s by the automated fast perfusion system [25]. In order to determine GABA EC5-10 (typically between 3 and 8 μM), a dose–response experiment with GABA concentrations ranging from 0.1 μM to 1000 μM was performed. Stock solution of nutmeg oil (10 μL/mL in DMSO) was diluted to a concentration of 100 nL/mL with bath solution and then mixed with GABA EC5-10 before dissolved in 2.97 mL of bath solution. In the second assay, for dose– dependent experiments of three components of nutmeg oil (α-terpineol, myristicin and limonene) were also diluted into a set of concentrations from 1 μM to 500 μM with bath solution and then mixed with GABA EC5-10. Compounds were applied for an equilibration period of 1 minute before concomitant application of GABA (EC5-10) and increasing concentrations of the compound (1, 10, 100 and 500 μM).
Data analysis
Enhancement of the chloride current (I GABA) was defined as (I(GABA+Comp) /I GABA) – 1, where I (GABA+Comp) is the current response in the presence of a given compound, and I GABA is the control GABA-induced chloride current. Concentration– response curves were generated and the data fitted by non-linear regression analysis. Data were fitted to the equation: 1/(1+(EC50/[Comp])nH),where EC50 is the concentration of the compound that increases the amplitude of the GABA evoked current by 50% and nH is the Hill coefficient. Data are expressed as mean ± SE of at least 4 oocytes and ≥ 2 oocyte batches. Statistical significance is calculated using unpaired Student t-test with a confidence interval of p < 0.05.
Results
Nutmeg oil analysis
We isolated 16 compounds from nutmeg oil by GC-MS analysis method. The chromatogram of identified components is shown in figure 1. The compounds isolated are α-pinene, β-pinene, β-terpinene, α-phellandrene, 3- carene, α-terpinene, p-cymeme, limonene, γ-terpinene, terpinolene, 2-norbornanol, borneol, 4-terpineol, α- terpineol, myristicin and elemicin (Table. 1). The rest of the compounds were considered as low in their presence within the oil constituents.
GABAA receptors activation
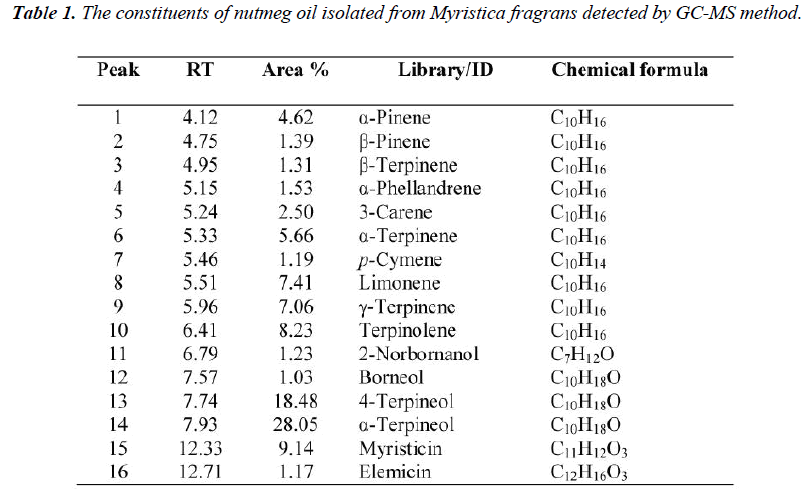
To determine the GABA EC5-10 at different concentration of GABA solution were tested on the Xenopus laevis oocytes containing of GABAA receptors α1β2γ2s subtype. A total of five concentrations (1 μM, 5 μM, 10 μM, 100 μM and 1000 μM) of GABA solution were used and the maximum potentiation was found at 1000 μM concentration. The concentration–response curve was plotted in figure 2.
GABAA receptor modulating property of nutmeg oil and its compounds
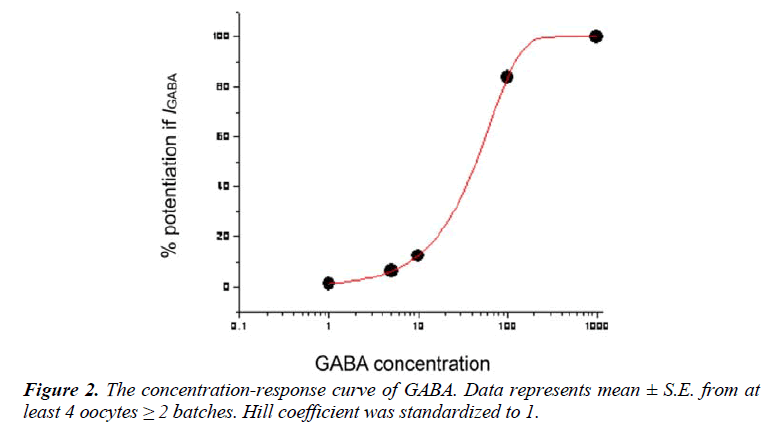
In the two-microelectrode assay, the essential oil of Myristica fragrans kernel significantly increased stimulation of I GABA through the GABAA α1β2γ2s receptors. A prominent current trace for the modulation of GABA-induced chloride currents through GABAA α1β2γ2s receptors was found in the essential oil when tested at 100 nL/mL in comparison to GABA control (Figure 3). The essential oil at this concentration induced I GABA by approximately 100%. The essential oil was submitted to GC-MS profiling to identify the molecular structure of its components. In the second assay of screening, three pure compounds from the essential oil of nutmeg were tested on GABAA receptors in order to find which compound showed promising activity in the oil constituents. α-Terpineol, myristicin, and limonene were selected because they represent the high, moderate and low in percentage of oil component. The chemical structures of these are shown in figure 4.
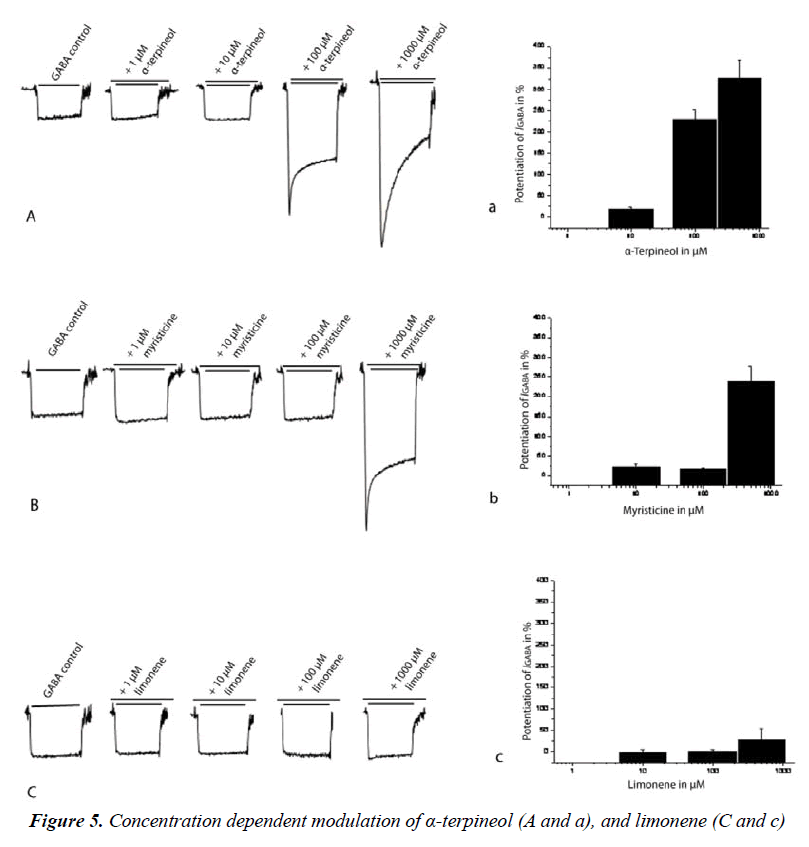
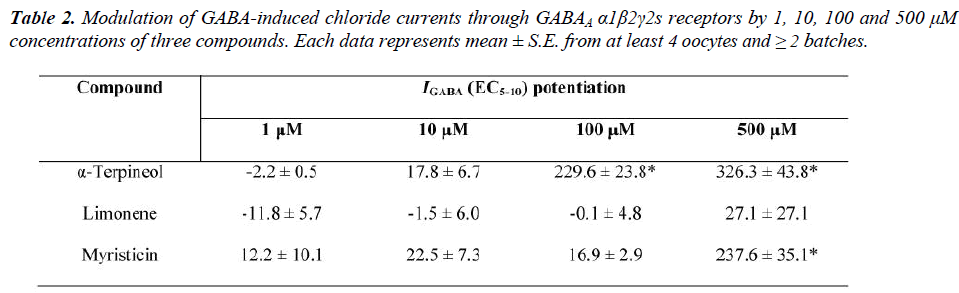
For a quantitative determination of GABAA receptor activity of compounds of α-terpineol, myristicin and limonene were tested on GABAA receptors α1β2γ2s subtype in the presence of GABA EC5-10 at several concentrations (1, 10, 100 and 500 μM) in the oocytes assay. α-Terpineol was most efficient as it potentiated I GABA by 326.3±43.8% at 500 μM, while myristicin at the same concentration was less efficient (potentiation of I GABA by 237.6±35.1%). Weak enhancement of I GABA (27.1±27.1%) was observed for limonene at same concentration (Table 2). Only α-terpineol and myristicin enhanced statistical significance of I GABA, and a weak enhancement of I GABA was observed for limonene when compared to GABA controls (Figure 5).
The EC50 value was determined by fitting the concentration- effect data to the Hill equation, for α-terpineol the EC50 was ≈ 88 μM and myristicin showed a fivefold increase than α-terpineol (≈ 403 μM), while the maximum I GABA stimulation by limonene (27.1±27.1% at 500 μM) was considered as low (below than 40% of enhancement). Therefore the EC50 value was not determined.
Discussion
Nutmeg oil is traditionally used to manage seizure symptoms [2]. In our previous work we found α-terpineol, one of the major components of nutmeg oil, showed a dose dependent anticonvulsive activity in GAERS animal model [9]. We hypothesized that the nutmeg oil has some compounds; which may show inhibitory activities in GABAA receptors like many of the conventional antiepileptic drugs. In the present study we found a positive modulation in GABAA α1β2γ2s receptors for nutmeg oil and its compounds α-terpineol and myristicin. Several chemical compounds can modulate functions of GABAA receptors. Benzodiazepines, imidazopyridines [26], barbiturates [27], alcohol [28], general anesthetics [29] and neurosteroids [30] act as positive modulators. Conventional antiepileptic drugs generally inhibit sodium currents (carbamazepine, phenobarbital, phenytoin, vaproate) or enhance GABA-ergic inhibition (benzodiazepine, phenobarbital, valproate) [10].
Three compounds of nutmeg oil are tested in this study; α-terpineol and myristicin acted as positive allosteric modulators by potentiating I GABA at GABAA receptors α1β2γ2s subtype. For α-terpineol, potentiation was observed at concentrations of 100 μM and 500 μM with GABA EC5-10 elicited I GABA 229.6±23.8% and 326.3±43.8%, respectively. This enhancement is likely to be the result of a positive allosteric of α-terpineol. In contrast, at 100 μM of concentration myristicin showed weak I GABA potentiation, and at 500 μM concentration showed relatively high enhancement of I GABA. To determine the potency and efficiency (maximum ability to enhance the GABA EC5-10 response) we studied their concentration-effect for the receptors. α-Terpineol displayed the highest potency (EC50 ≈ 88 μM) followed by myristicin (EC50 ≈ 403 μM) and therefore the most potent modulator between both compounds on the receptors was α-terpineol which exhibited approximately 4.6 times higher potentiation than myristicin.
The monoterpenoid compounds such as menthol [31], borneol [32], thymol [33], thujone [34], and itronello and pinene [35] act as potential modulators of GABAA receptors. In this study, we found that α-terpineol which is one of the monoterpenoids, positively modulates I GABA at GABA EC5-10. The present finding reinforces the argument that the monoterpenoid acts as a modulator of GABAA receptor but it is a questionable fact, whether this compound shares a similar site of action with other recognized e.g. benzodiazepine, sites on the receptor. Priestly [33] suggested that thymol (monoterpenoid) has another novel binding site. Further research is required to explore this issue.
Myristicin is a phenylpropene; which is a natural organic compound and is a major component of the nutmeg seed. Myristicin is the principle aromatic constituents of the essential oil and it has been implicated to manage the adverse effects such as detachment from reality, tachycardia, flushing, hypotension, drowsiness, confabulation, gagging, vomiting, ileus, paresthesia, numbness, blurred vision, hypothermia and sweating [36−38]. Recently Leiter [39] examined the purported anxiolytic properties of myristicin, and its potential interaction with GABAA receptor site [37]. Sonavane [40] also suggested that myristicin may act at GABAA receptor sites other than the benzodiazepine site. The present findings of Xenopus oocytes expression system using GABAA receptor α1β2γ2s type at concentration 500 μM of myristicin potentiated I GABA at GABA EC5-10 by 237.6±39.% supported the previous studies. The weak potentiation of limonene revealed that its affinity for the binding site of GABAA receptor was not as strong as in the case of α-terpineol or myristicin. Furthermore we can consider that it is an isomer and shares the same functional group in the structure. This finding might lead to the investigation about the functional group of the compounds in terms of activity on the receptor sites. Further studies are required to elucidate the potentiality of other components of nutmeg oil.
Conclusion
The present study presumes that nutmeg essential oil has promising inhibitory activity by enhancement of I GABA. α-Terpineol is the important constituent of nutmeg oil; which has the maximum inhibitory activity on GABAA receptor modulators in Xenopus oocytes. This finding also provides scientific evidence for the dose dependent antiepileptic effects of α-terpineol on GAERS rat model that we found in our previous study [9] and its traditional use for the treatment of epilepsy.
Acknowledgements
The study was supported by RU grant (Grant No. 1001/PSKBP/812019) of Universiti Sains Malaysia. The authors wish to thank Dr. Annette Hohaus, Institute of Pharmacology and Toxicology, University of Vienna, Austria for supplying clone of GABAA receptors subunit.
References
- Panayotopoulos DJ, Chisholm DD. Hallucinogenic effect of Nutmeg. Brazilian Med J 1970; 1:754-760.
- Wahab A, Haq RU, Ahmed A, Khan R, Raza M. Anticonvulsant activities of nutmeg oil of Myristica fragrans. Phytother Res 2009; 23: 153-158.
- Sonavane GS, Sarveiya V, Kasture VS, Kasture SB. Anxiogenic activity of Myristica fragrans seeds. Pharmacol Biochem Behav 2001; 71:239−44.
- Nadkarni K, Indian Materia Medica, Bombay Popular Prakashan, Mumbai,India 1998; Vol. 3 p 830.
- Takikawa A, Abe K, Yamamoto M, Ishimaru S, Yasui M, Okubo Y. Antimicrobial activity of nutmeg against Escherichia coli0157. J Biosci Bioengineer 2002; 94: 315-320.
- Olajide OA, Ajiya FF, Ekhelar AI, Awe SO, Makinde JM, Alada ARA. Biological effects of Myristica fragrans (nutmeg) extract. Phytother Res 1999; 13: 344-345.
- Morita T, Jinno K, Kawagishi H, Arimoto Y, Suganuma H, Inakuma T. Hepatoprotective effect of myristicin from nutmeg (Myristica Fragrans) on lipopolysacharide/D-galactosamine-induced liver injury. J Agric Food Chem 2003; 51: 1560-1565.
- Dhingra D, Sharma A. Antidepressant-like activity of n-hexane extract of nutmeg (Myristica fragrans) seed in mice. J Med Food 2006; 9: 84-89.
- Islam MR, Muthuraju S, Tarmizi CH, Zulkifli MM, Osman H, Mohamad H, Abdullah JM. Anticonvulsant activity of α-terpineol isolated from Myristica fragrans in GAERS model of absence epilepsy. ASM Science J 2012; 6: 95--102.
- Czapiński P, Blaszczyk B, Czuczwar SJ. Mechanisms of action of antiepileptic drugs. Curr Trop Med Chem 2005; 5: 3-14.
- Bailer M, White HS .Key factors in the discovery and development of new antiepileptic drugs. Nature Rev Drug Discover 2010; 9:68-82.
- Abdullah JM. Interesting Asian plants: Their compounds and effects on electrophysiology and behaviour. Malaysian J Med Sci 2011;18: 1-4.
- Hevers W, Lüddens H . The diversity of GABAA receptors. Pharmacological and electrophysiological properties of GABAA channel subtypes. Mol Neurobiol 1998; 18: 35-86.
- Simon J, Wakimoto H, Fujita N, Lalande M, Barnard EA. Analysis of the set of GABAA receptor genes in the human genome. J Biol Chem 2004; 279: 422-435.
- Olsen RW, Sieghart W. GABAA receptors: Subtypes provide diversity of function and pharmacology. Neuropharmacol 2009; 56: 141-148.
- Benke D, Mertens S, Trzeciak A, Gillessen D, Möhler H (1991) GABAA receptors display association of γ2-subunit with α-1 and β2/3-subunits. J Biol Chem 1991; 266: 4478-4483.
- Benke D, Fritschy JM, Trzeciak A, Bannworth W, Möhler H. Distribution,prevalence and drug binding profile of gamma-aminobutyric acid type A receptor subtypes differing in the β-subunit variant. J Biol Chem 1994; 269: 27: 100-107.
- Whiting PJ. GABA(A) receptors: a viable target for novel anxiolytics? Curr Opin Pharmacol 2006; 6: 24-29.
- Riss J, Clyod J, Gates J, Collins S. Benzodiazepines in epilepsy: pharmacology and pharmacokinetics. Acta Neurol Scandinavia 2008;118: 69-86.
- Khom S, Baburin I, Timin EN, Hohaus A, Trauner G, Kopp B, Hering S. Valerenic acid potentiates and inhibits GABAA receptors: molecular mechanism and subunit specificity. Neuropharmacol 2007; 53: 178-187.
- Zaugg J, Baburin I, Strommer B, Kim HJ, Hering S, Hamburger M. HPLCbased activity profiling: discovery of piperine as a positive GABA (A) receptor modulator targeting a benzodiazepine-independent binding site. J Nat Prod 2010; 73: 185-191.
- Zaugg J, Eickmeier E, Rueda DC, Hering S, Hamburger M. HPLC-based activity profiling of Angelica pubescens roots for new positive GABAA receptor modulators in Xenopus oocytes. Fitoterap 2011; 82: 434-440.
- Goren AC. Analysis of Essential Oil of Coridothymus capitatus (L.) and Its Antibacterial and Antifungal Activity. Z Naturforsch 2003; 58: 687-690.
- Methfessel C, Witzemann V, Takahashi T, Mishina M, Numa S, Sakmann B. Patch clamp measurements on Xenopus laevis oocytes: currents through endogenous channels and implanted acetylcholine receptor and sodium channels. Euro J Physiol 1986; 407: 577-588.
- Baburin I, Beyl S, Hering S (2006) Automated fast perfusion of Xenopus oocytes for drug screening. Europ J Physiol 2006; 453: 117-123.
- Möhler H, Fritschy JM, Rudolph U (2002) A new benzodiazepine pharmacology. J Pharmacol Exp Therap 2002; 300: 2–8.
- Chebib M, Johnston GA. The ‘ABC’ of GABA receptors: a brief review. Clin Exp Pharmacol Physiol 1999; 26: 937-940.
- Milnic SJ, Ye Q, Wick MJ, Kaltchine VV, Krasowski MD, Finn SE. Sites of alcohol and volatile anaesthetic action on GABA (A) and glycine receptors. Nature 1997; 389: 385-389.
- Mascia MP, Trudell JR, Harris RA. Specific binding sites for alcohols and anaesthetics on ligand-gated ion channels. Proceed Nat Acad Sci 2000; 97: 9305-9310.
- Belelli D, Lambert JJ .Neurosteroids: endogenous regulators of the GABA (A)receptor. Nature Rev Neurosci 2005;6: 565-675.
- Watt EE, Betts BF, Kotey O, Humbert DJ, Griffith TN, Kelly EW. Menthol shares general anaesthetic activity and sites of action on the GABAA receptor with the intravenous agent, propofol. Euro J Pharmacol 2008; 590: 120-126.
- Granger RE, Campbell EL, Johnston GAR. (+)-and (-)-Borneol: efficacious positive mudulators of GABA action at human recombinant α1β2γ2L GABAA receptors. Biochem Pharmacol 69: 1101-1111.
- Priestly CM, Williamson EM, Wafford KA, Sattelle DB .Thymol, a constituent of thyme essential oil, is a positive allosteric modulator of human GABAA receptors and a homo-oligomeric GABA receptor from Drosophila melanogaster. Braz J Pharmacol 2003; 140: 1363-1372.
- Hold KM, Sirisoma NS, Ikeda T, Narahashi T, Casida JE (2000) α-Thujone (the active component of absinthe): γ-aminobutyric acid type A receptor modulation and metabolic detoxification. Proceed Nat Acad Sci 2000; 97: 3826-3831.
- Aoshima H, Hamamoto K. Potentiation of GABA-A receptors expressed in Xenopus oocytes by perfume and phytoncid. Biosci Biotech Biochem 1999; 63:743–748.
- Hallström H, Thuvander A. Toxicological evaluation of myristicin. Natural Toxin 1997; 5: 186-192.
- Sangalli BC, Chiang W. Toxicology of nutmeg abuse. Toxicol Clin Toxicol 2000; 38: 671-678.
- Grover JK, Khandar S, Vats V, Dhunnoo Y, Das D. Pharmacological studies on Myristica fragransantidiarrheal, hypnotic analgesic and hemodynamic (blood pressure) parameters. Exp Clin Pharmacol 2002; 24: 675-680.
- Leiter E, Hitchcock G, Godwin S, Johnson M, Sedgwick W, Jones W. Evaluation of the Anxiolytic Properties of Myristicin, a component of Nutmeg, in the Male Sprague-Dawley Rat. AANA J 2001; 79: 109-114.
- Sonavane GS, Palekar RC, Kasture VS, Kasture SB .Anticonvulsant and Behavioral actions of Myristica fragrans seeds. Indian J Pharmacol 2002; 34: 332-338.