- Biomedical Research (2013) Volume 24, Issue 1
The effects of low level laser therapy (LLLT) on the testis in elevating serum testosterone level in rats.
Jin-Chul Ahn1,2, Young-Hoon Kim3 and Chung-Ku Rhee1,2,*1Medical Laser Research Center, Dankook University, Cheonan, Korea 330-714.
2Department of Otolaryngology-Head & Neck surgery, Dankook University College of Medicine, Cheonan, Korea 330 714.
3Department of Otolaryngology-Head & Neck surgery, Wallace Memorial Baptist Hospital, Busan, Korea.
- *Corresponding Author:
- Chung-Ku Rhee
Medical Laser Research Center
Dankook University College of Medicine
Cheonan, Chungnam-do, 330-714, Korea
Accepted Date: October 07 2012
Abstract
Testosterone (T) plays a vital role in the sexual function and many other health-related phenomena. We conducted this study to examine the effects of low level laser therapy (LLLT) on the testis in elevating serum T level in rats. We performed the current experimental study using 30 male Sprague-Dawley rats (Orient Bio, Korea) aged six weeks, weighing 200 g. In rats of two laser groups, a 670-nm diode laser or an 808-nm one were irradiated to the testes at an intensity of 360 J/cm2/day (200 mW ´ 30 min) for five days. This was followed by the measurement of the depth of tissue penetration, that of serum T level and histopathological examination. Our results showed that the rate of tissue penetration was significantly higher in the 808 nm wavelength group as compared with the 670 nm wavelength group (P<0.05); serum T level was not significantly higher in the experimental groups as compared with the control group; but serum T level was significantly elevated in the 670 nm wavelength group on day 4. Thus the LLLT using a 670-nm diode laser was effective in increasing serum T level without causing any visible histopathological side effects. In conclusion, the LLLT might be an alternative treatment modality to the conventional types of testosterone replacement therapy.
Keywords
Testosterone, testis, LLLT and diode laser.
Introduction
A steroid hormone from the androgen group, testosterone (T) plays a vital role in the sexual function and many other health-related phenomena. Gonadotropin-releasing hormone (GnRH) is secreted from the hypothalamus, and it promotes the secretion of luteinizing hormone (LH) by stimulating the pituitary gland. Then, LH promotes the synthesis of T by stimulating the Leydig cells of the testis. In humans, T plays a very critical role in the development of male reproductive tissues and the expression of secondary sexual characteristics such as muscle, bone mass and hair growth [1].
Men with a low T level presented with such symptoms as decreased sex drive, impotence, fatigue, loss of lean muscles, fat accumulation and weight gain, an increased risk of osteoporosis and bone fractures and an increased risk of cardiovascular disease [2]. The Baltimore Longitudinal Study of Aging (BLSA) was conducted in a total of 890 male subjects, which showed that 20% of men in their 60s, 30% in their 70s and 50% in their 80s were hypogonadal [3].
To compensate for the abnormalities, T replacement therapy has been performed by the oral or parenteral route. Because there is no need for the assistance of physicians, oral administration is convenient for patients. But early pharmaceutical preparations entered blood circulation by the liver. Therefore, there is a possibility that this might cause cholestatic jaundice, hepatocellular neoplasms, peliosis hepatis and hepatocellular carcinoma. To minimize the toxicity, T undecanoate (TU) has been developed and it is absorbed by the lymphatic system [4]. Nevertheless, it is also not free from the possibility of side effects [5].
Parenteral methods are not so convenient as oral administration, which include syringe injections or transdermal applications such as gels, cream and patches. There is no denying their effectiveness. Following syringe injections of T enanthate and T cypionate, however, serum T level is abruptly increased or decreased. They are also known to cause side effects such as pain, edema or jaundice [6].
Low-level laser therapy (LLLT) has a wavelengthdependent capability to alter cellular behavior in the absence of significant thermal effects. To date, LLLT has been used for therapeutic purposes in treating wound healing, musculoskeletal pain and dental diseases [7,8]. Some studies have examined the effects of LLLT in augmenting the regeneration of various tissues such as skin, bone, skeletal muscle and nerve [7,9]. Cellular functions are altered by LLLT irradiation, which include an increase in protein synthesis, cell growth and differentiation, cell motility, membrane potential, binding affinities, neurotransmitter release, phagocytosis, ATP synthesis and prostaglandin synthesis [11]. But there are no established mechanisms by which these alterations occur. Components of the mitochondrial respiratory chain exhibit frequency- dependent spectra of the action, which leads to the speculation that the respiratory chain plays a vital role in the effects of LLLT [9].
Given the above background, we conducted this study to examine the effects of LLLT on the testis in elevating serum T level in rats.
Materials and Methods
1. Experimental animals
Prior to the experiment, 30 Sprague-Dawley male rats (Orient Bio, Korea) aged six weeks, weighing 200 g, were given a 1-week adaptation period. These experimental animals were divided into three groups: the control group, the 670 nm wavelength group and the 808 nm wavelength group. Each group comprised ten rats. The rats were anesthetized with KetaminÒ (Yuhan Corp., Korea) 30 mg/kg and RumpunÒ (Bayer, Leverkusen, Germany) 2 mg/kg before the experimental procedures. At the end of experimental procedures, rats were sacrificed for histopathologic examination. The current experimental study was conducted in compliance with all the ethical guidelines of the Dankook University Institutional Animal Care and Use Committee.
2. Laser irradiation and the measurement of tissue penetration
In rats of two laser groups, a 670-nm diode laser or an 808-nm one (LAS-30A, Daedeok Laser, Daejeon, Korea) were irradiated to the testes at an intensity of 360 J/cm2/day (200 mW ´ 30 min) for five days. But rats of the control group were placed on the operation table for laser therapy for 30 minutes without any laser irradiations.
The tissue samples were harvested from the tissues surrounding the testis, including the epidermis and dermis from five rats. To measure the rate of tissue penetration of a 670-nm and an 808-nm diode laser, a laser power meter (Coherent, USA) was placed underneath the samples. To make sure that lasers should be irradiated perpendicularly to the samples, a laser bare fiber was fixed. Following the dissection of the scrotal skin covering the testis in rats, the laser was irradiated as described above. Thus the depth of tissue penetration was confirmed. Then, to confirm the depth of tissue penetration by the laser irradiation, the testis was divided into three parts depending on the thickness: the surface (0/4), the middle part (2/4) and the terminal part (4/4). This was followed by a comparison of the depth of tissue penetration between the irradiation effects. This was followed by the irradiation with laser therapy of two wavelengths perpendicularly from the top of the samples.
3. Blood sampling and histopathological examination
Blood samples were collected at 08:00 A.M. before each session of laser irradiation on days 2, 3, 4, 5 and 6. Blood samples were not drawn on day 1, but laser irradiation was given to both wavelength groups (670 nm and 808 nm). Serum T level was measured with a radioimmunoassay using COBRA II Gamma counter (PacKard, USA) at the Seoul Medical Science Institute (SMSI, Seoul, Korea). On day 6, the testis tissue samples were obtained from all the three groups for a histolopathological examination. In each group, the testis tissue samples were fixed with 3.7% formaldehyde and prepared into a 5-mm thick slide through a paraffin block. This was followed by hematoxylin and eosin (H&E) staining.
4. Statistical analysis
All data are expressed as mean±SD (SD: standard deviation). Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) program. A P-value of <0.05 was considered statistically significant.
Results
1. The rate of tissue penetration
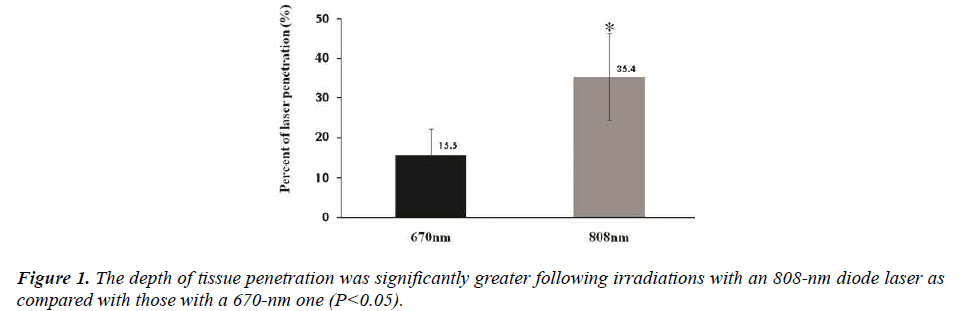
Following the irradiation of the tissue surrounding the testis with lasers of two different wavelengths (670 nm and 808 nm), the rate of tissue penetration was 15.5±6.7% and 35.4±11.0% in the corresponding order (Fig. 1). These results indicate that the rate of tissue penetration was significantly higher in the 808 nm wavelength group as compared with the 670 nm wavelength group (P<0.05).
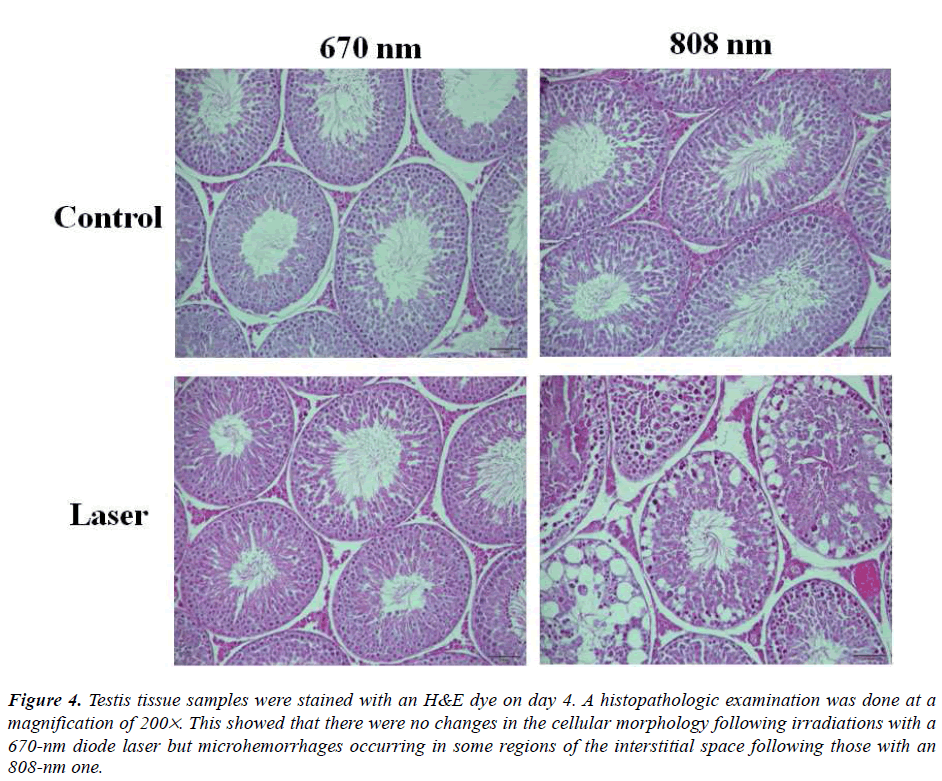
Figure 4: Testis tissue samples were stained with an H&E dye on day 4. A histopathologic examination was done at a magnification of 200´. This showed that there were no changes in the cellular morphology following irradiations with a 670-nm diode laser but microhemorrhages occurring in some regions of the interstitial space following those with an 808-nm one.
Discussion
It is nowadays well established that the light of infrared wavelength has a higher rate of tissue penetration as compared with that of visible one. In the current study, we therefore compared the rate of tissue penetration between the two wavelengths (670 nm and 808 nm). In the 808 nm wavelength group, the rate of tissue penetration was significantly higher as compared with the 670 nm wavelength group (P<0.01). The light of wavelength ranging from 400 to 600 nm is absorbed in the dermis by blood chromophores: hemoglobin, oxyhemoglobin, bilirubin and carotene. Huang YY et al. reported that the light of wavelength ranging from 700 to 1,300 nm is weakly absorbed by the blood, but its scattering is decreased in the dermis [16]. These authors also noted that there was a significant difference in the rate of tissue penetration between the two wavelengths (670 nm and 808 nm).
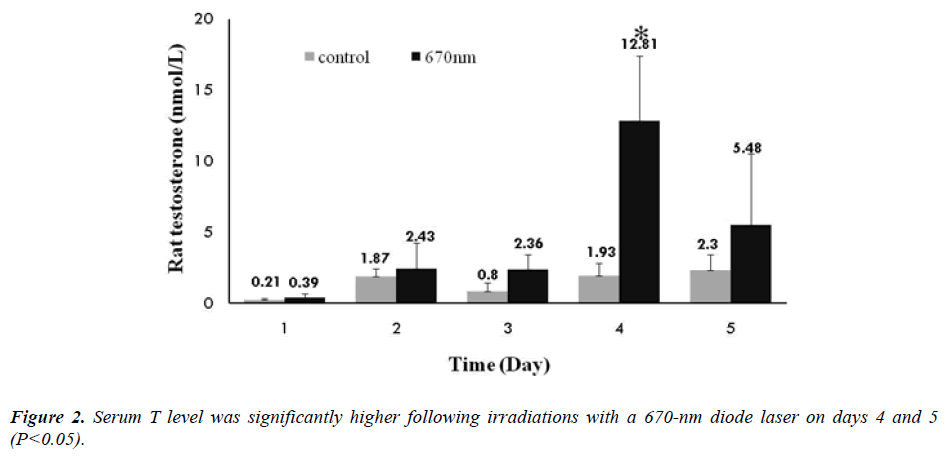
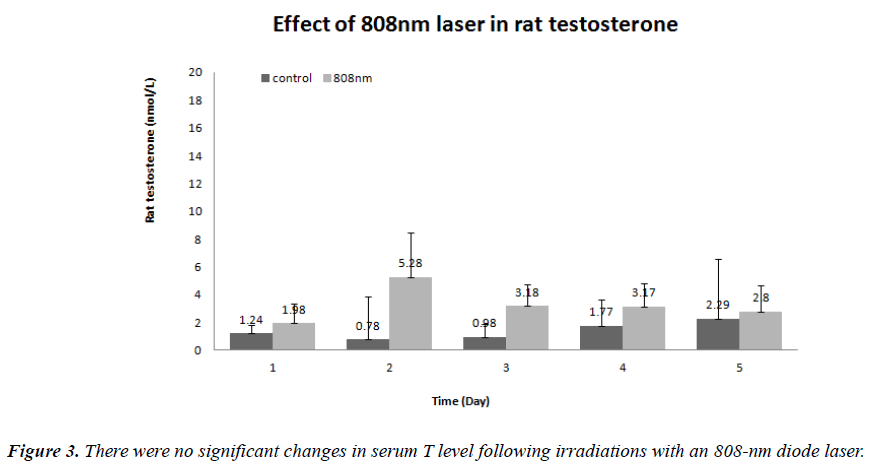
Despite a higher rate of tissue penetration, however, serum T level was significantly increased in the 808 nm wavelength group. In the 670 nm wavelength group, serum T level was also significantly increased the testosterone levels at the same intensity of 360 J/cm2/day. On histopathological examination, there were no definite changes in the 670 nm wavelength group. In the 808 nm wavelength group, there were such findings as an atrophy of the seminiferous tubules, disarrangement of sertoli cells, generation of giant multinucleated bodies and other deformities. It is apparent that the adverse effects occurred but serum T level was not increased following a 5- day course of irradiation with an 808-nm wavelength at n intensity of 360 J/cm2/day. Taha MF et al. showed similar histopathological findings [15]. According to these authors, on histopathological examination of the testis irradiated with an 830-nm wavelength at lower doses (28.05 J/cm2), there were normal appearances of the seminiferous epithelium and interstitial tissue. These authors also noted, however, that the seminiferous epithelium and interstitial tissue were irregularly arranged following the irradiation with an 830-nm wavelength at higher doses (46.8 J/cm2). It has been reported that laser irradiation of lower doses has a positive biostimulatory effect. According to these reports, its positive effects in controlling the cell metabolism occur only in a narrow range of doses. That is, laser irradiation of higher doses is associated with the growth inhibition and cell lethality [11]. A biphasic dose response has been frequently observed where laser irradiation of lower doses have a much better effect in stimulating and repairing tissue as compared with that of higher doses [16]. Previous studies have shown that laser irradiation of lower doses have positive and negative biostimulatory effects on various endocrine glands such as thyroid glands and pancreatic islet [17,18]. The current study also showed that there were the same types of the positive and negative biostimulatory effects.
Our results indicate that the LLLT might be an alternative modality to the conventional testosterone medications. In addition, there were different reactions of the tissues to the light of different wavelengths. To confirm the effectiveness of the laser treatments, various types of laser should be tested. This should also be followed by the establishment of a safe, solid protocol depending on the wavelength, power intensity and an ability of the different types of lasers to penetrate the tissue. Further studies are also warranted to examine the mechanisms by which the laser treatments affect serum T level as compared with the conventional types of testosterone replacement therapy. In addition, it should also be thoroughly examined for whether the LLLT has any adverse effect on the testis.
Conclusion
Our results showed that the LLLT using a 670-nm diode laser was effective in increasing serum T level without causing any visible histopathological side effects to the tissue. Thus the low level laser therapy may be an alternative treatment modality for conventional types of testosterone replacement therapy.
Acknowledgement
Sincere appreciation and thanks to the Dankook University Medical Laser and Device Research Center for their financial support.
Disclosure Statement
No conflicting financial interest exists.
References
- Kishore ML, Shehzad B. Safety and efficacy of testosterone gel in the treatment of male hypogonadism. Clinical Interventions in Aging 2009; 4: 397-412.
- Stephen EB, Thomas M. Testosterone replacement therapy for older men. Clinical Interventions in Aging 2007; 2: 561-566.
- Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab 2001; 86: 724-731.
- Kıhn FM, Schill WB. A new oral testosterone undecanoate formulation. World J Urol 2003; 21: 311-315.
- Kloer H, Hoogen H, Nieschlag E. Trial of high-dose testosterone undecanoate in treatment of male infertility. Int J Androl 1980; 3: 121-129.
- Nieschlag E. Testosterone Treatment Comes of AgeL New Options for Hypogonadal Men. Clinical Endocrinology 2006; 65: 275-281.
- Hansen HJ, Thoroe U. Low power laser biostimulation of chronic oro-facial pain: A double-blind placebo controlled cross-over study in 40 patients. Pain 1990; 43: 169-179.
- Simunovic Z. Low level laser therapy with trigger point technique: A clinical study on 243 patients. J. Clin. Laser 1996; 14: 163-167.
- Basford JR. Low intensity laser therapy: Still not an established clinical tool. Lasers Surg Med 1995; 16: 331-341.
- Michael R, Hamblin et al. Low-Level Laser Therapy Activates NF-kB via Generation of Reactive Oxygen Species in Mouse Embryonic Fibroblasts. PLoS One 2011; 6(7): e22453.
- Karu TI. Molecular mechanism of low-intensity laser radiation. Lasers Life Sci 1988; 2: 53-74.
- Dougherty TJ. Therapy and detection of malignant tumors. Photochem Photobio 1987; 45: 879.
- Dougherty TJ. Photodynamic therapy. In: Innovations in Radiation Oncology. Withers H, Peters L, eds. Berlin/ Heidelberg: Springer-Verlag 1988;. p175.
- Kolarova H, Ditrichova D, Waqner J. Penetration for the laser light into the skin in vitro. Lasers in surg Med 1999; 24: 231.
- Taha MF, Valojerdi MR. Quantitative and qualitative changes of the seminiferous epithelium induced by Ga. Al. As. (830 nm) laser irradiation. Lasers in surgery and medicine 2004; 34: 352-359.
- Huang YY, Chen ACH, Carroll JD, Hamblin MR. Biphasic dose response in low level light therapy. Dose -Response 2009; 7: 358-383.
- Azevedo LH, Aranha AC, Stolf SF, Eduardo CD, Vieira MMF. Evaluation of low intensity laser effects on the thyroid gland of male mice. Photomedicine and laser surgery 2005; 23: 567-570.
- Irani S, Monfared SSMS, Akbari-Kamrani M, Abdollahi M, Larijani B. Effect of low-level laser irradiation on in vitro function of pancreatic islets. Transplantation Proceedings 2009; 41: 4313-4315.