Research Article - Biomedical Research (2017) Volume 28, Issue 18
Small-bore versus large-bore catheters for talc pleurodesis of malignant pleural effusion: a tertiary hospital experience
Ülkü Aka Aktürk1*, Nagihan Durmuş Koçak1, Cansel Atinkaya Öztürk1, Aysun Şengül2, Dilek Ernam1, Merve Alan Tepetam3 and Elçin Ersöz1
1Chest Department, Süreyyapaşa Chest Disease and Thoracic Disease Education and Research Hospital, Health Science University, İstanbul, Turkey
2Chest Department, Derince Education and Research Hospital, Kocaeli, Turkey
3Allergy and Immunology Department, Süreyyapaşa Chest Disease and Thoracic Disease Education and Research Hospital, Health Science University, İstanbul, Turkey
- *Corresponding Author:
- Ülkü Aka Aktürk
Chest Department
Süreyyapaşa Chest Disease and Thoracic Disease Education and Research Hospital
Health Science University, İstanbul, Turkey
Accepted date: August 24, 2017
Abstract
Background: Malignant Pleural Effusion (MPE) is a complication of advanced malignancy occurring commonly in patients with lung cancer, lymphoma and breast cancer. Pleurodesis is often used to prevent re-accumulation of fluid and to help respiratory status. The size of the catheter (small versus large) to be used for pleurodesis, however, continues to be debated.
Objective: To describe a tertiary hospital experience in patients requiring pleurodesis for malignant pleural effusion, with a comparison of small-bore catheter and large-bore catheter outcomes.
Methods: Retrospective chart abstraction study of patients who received chemical pleurodesis with talc via catheter for malignant pleural effusions between 1 January 2012 and 31 December 2014 in our hospital. Small-bore (8-10 F) versus large-bore (28-32 F) catheter outcomes of interest were length of stay (LOS) post-catheterization and total LOS.
Results: A total of 185 patients were included in the study, of whom 67 (36.2%) received a small-bore catheter and 118 (63.8%) a large-bore catheter groups, respectively. Lung cancer was the most common primary malignancy. Using propensity-score matched analyses, post-catheterization LOS was shown to be significantly shorter in the small-bore catheter group (1.8 d; p<0.001), however, total LOS was not different between the two groups (p=0.89). The pain VAS score was significantly lower in small bore catheter group than in large bore catheter group (p<0.001). The success rates were similar in the two groups (p=0.68).
Conclusion: In our study post-procedure length of hospital stay is shorter when talc pleurodesis is conducted via small-bore catheter as compared to large-bore catheter. The success rates of pleurodesis were found to be similar regardless of the type of tube inserted. In addition, the pain VAS score was significantly lower in small bore catheter group than the large one.
Keywords
Talc pleurodesis, Malignant pleural effusion, Small-bore catheter
Introduction
Malignant pleural effusion is a complication of advanced malignancy, occurring commonly in patients with lung cancer, lymphoma and breast cancer, and is associated with a poor prognosis [1,2]. Malignant pleural effusion causes progressive dyspnoea, chronic cough, chest pain, and reduces physical activity, all of which significantly impact patient Quality of Life (QoL) [3,4]. To prevent re-accumulation, to improve respiratory status and to improve QoL, chemical pleurodesis is often performed in patients with malignant pleural effusion. This procedure employs chemical sclerosant agents such as talc, povidone-iodine, tetracycline derivatives, or bleomycin or physical abrasion of pleural surfaces [5-7]. Talc is preferred as it is associated with less malignant pleural effusion recurrence and a low risk of complications (e.g., acute respiratory failure, pneumonia, treatment-related death) [2,8].
Currently, both large and small-bore catheters are used in pleurodesis followed by pleural drainage. The main reason for using a large-bore catheter is the improved flow rate with more viscous effusions. However, large-bore catheters are uncomfortable and require a larger incision and tissue dissection. Previous studies support the hypothesis that small-bore catheters are as effective as large-bore catheters, and they have the advantages of being safe and well tolerated [9,10]. In this study, we describe clinical characteristics of patients receiving talc pleurodesis for malignant effusion at a tertiary referral hospital and further evaluate the length of hospital stay associated with small-bore versus large-bore catheters.
Material and Methods
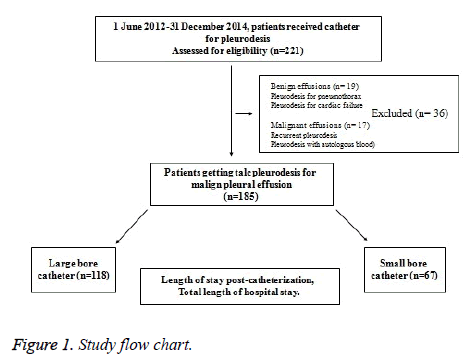
Study design and study population: We conducted a retrospective chart abstraction-based study of patients receiving chemical pleurodesis with talc via catheter. The inclusion criteria were adult patients over 18 y old with malignant pleural effusion that underwent pleurodesis via catheter in the Süreyyapaşa Chest and Thoracic Surgery Education and Research Hospital between 1 January 2012 and 31 December 2014. Malignant pleural effusions were proven pathologically by pleural fluid cytology and/or pleural biopsy. Patients whom performed pleurodesis in this study didn’t have re-expansion problem after pleural fluid drainage. Each patient was verified for re-expansion by postero anterior chest X-ray graphy after pleural drainage. We excluded patients who received chemical pleurodesis for etiologies other than malignancy, and additionally patients who were requiring recurrent pleurodesis.
Pleurodesis technique
After malignant pleural effusion was proven pathologically, pleural drainage was performed via small or large bore catheters by the thoracic surgeons. The thoracic surgeons decided the catheter type and replaced these catheters. After local anesthesia with lidocaine, a large-bore (size 28-32 F) or small-bore catheter (size 8F-10 F) was inserted into the mid-axillary line through the fifth or sixth intercostal space. The catheter was connected to a drainage system to completely drain the effusion and facilitate lung re-expansion. Total lung re-expansion was radiographically verified and pleurodesis was performed when drainage decreased to <200 mL/d. Patients were first sedated with 10 mg diazepam administered parenterally. Normal saline solution (50 mL) containing 2 mg/kg lidocaine was infused through the catheter as a local anaesthetic. After 15-20 min, 5 g sterile, asbestos-free talc (Steritalc® F2; Novatech, France) in 50 mL normal saline was instilled through the chest drain. After asbestos free talc slurry was instilled through the catheter, 50 ml normal saline was pushed afterward. Then catheter was clamped for 6 h after talc instillation. The catheter was withdrawn when<200 mL of fluid was drained per day.
Variables
Patients were classified into two groups according to whether they underwent pleurodesis with a small-bore (8-10 F) or large-bore (28-32 F) catheter. The primary outcome was LOS post-catheterization, and the secondary outcome was the total LOS. The pain Visual Analogue Scale (VAS) score was recorded. Chest x-rays following pleurodesis within 12 weeks were evaluated to detect efficacy of the procedure. We defined complete response as symptomatic improvement of dyspnoea with complete radiographic resolution of the effusion and partial response as symptomatic improvement of dyspnoea that did not require additional thoracentesis and fluid level below prepleurodesis level at the end of the 12 weeks follow up period. Total response rate was defined as the sum of both complete and partial response.
Confounders were determined as age, gender, previous chemotherapy, type of primary cancer, comorbidities and pleural fluid parameters. Patients’ demographic variables were recorded from their medical records. All laboratory results were collected from our hospital online database (HIS: Hospital Information System). Pleural fluid parameters included levels of adenosine de-aminase (ADA), lactate dehydrogenase (LDH), total protein, albumin, and glucose. Malignancy type, comorbidities, chemotherapy, in-hospital death, the pain VAS score, response rates, complication rates, total LOS, and post-catheterization LOS were recorded.
Ethical approvals
The study was approved by the local Ethics Committee of the Health Science University, Süreyyapasa Chest Disease and Thoracic Surgery Education and Research Hospital and conducted in accordance with the ethical principles stated in the Declaration of Helsinki.
Statistical analysis
Descriptive statistics were defined as frequencies and percentages, or mean values and standard deviations (SDs). Chi-square tests were performed for group comparisons of categorical variables. We performed t-tests for normally distributed continuous variables and non-parametric Mann- Whitney U tests for variables with non-normal distributions. Based on associated factors with catheter type selection (i.e., age, gender, hypertension, cardiac failure, and other types of cancers), propensity scores were calculated to control for potentially confounding variables and minimize the effect of selection bias on patient allocation. These groups were matched with regard to age, sex, pleural fluid parameters, comorbidities, hypertension, cardiac failure and other types of cancer. In this propensity analysis, 50 matched pairs were identified. Linear regression analysis adjusted to propensity scores was used for multivariate analyses. A p value<0.05 was considered significant. All statistical analyses were carried out using SPSS software (version: 16.0; SPSS Inc., Chicago, IL, USA).
Results
A total of 221 patients who received catheter for pleurodesis were identified during the study period. Of these, 185 had malignant pleural effusion and met inclusion and exclusion criteria, with 67 (36.2%) and 118 (63.8%) patients in the small and large-bore catheter groups, respectively (Figure 1). Patient demographics and clinical characteristics according to catheter type are shown in Table 1. Lung cancer was the most common primary malignancy leading to malignant effusions (61.6%), with adenocarcinoma being the most prevalent among lung cancers (27.6%).
| Small bore catheter group n: 67 |
Large bore catheter group n : 118 |
P value | |
|---|---|---|---|
| Age | 62 ± 13 | 65 ± 13 | 0.1 |
| Gender(male) | 33 (48.5) | 68 (57.6) | 0.29 |
| Presence of comorbidity | 29 (43.3) | 55 (46.6) | 0.36 |
| Prior systemic therapy | 40 (59.7) | 62 (52.5) | 0.39 |
| Lung cancer | |||
| Adenocarsinoma | 22 (32.8) | 29 (24.6) | 0.75 |
| Non-small cell carcinoma | 15 (22.4) | 25 (21.2) | |
| Small cell carcinoma | 8 (11.9) | 15 (12.7) | |
| Mesothelioma | 9 (13.4) | 18 (15.3) | |
| Extrapulmonary malignancy | |||
| Breast cancer | 8 (11.9) | 14 (11.9) | - |
| Ovarian cancer | 2 (2.9) | 5 (4.2) | |
| Lymphoma | 2 (2.9) | 3 (2.5) | |
| Colon cancer | 1 (1.4) | 4 (3.4) | |
| Gastric cancer | 0 (0.0) | 2 (1.7) | |
| Renal cancer | 1 (1.4) | 0 (0.0) | |
| Osteosarcoma | 0 (0.0) | 1 (0.8) | |
| Pancreas cancer | 0 (0.0) | 1 (0.8) | |
| Pleurla LDH | 635 ± 1937 | 619 ± 1124 | 0.93 |
| Pleural glucose | 99 ± 48 | 119 ± 53 | 0.032 |
| Pleural albumin | 4 ± 9 | 2 ± 1 | 0.92 |
| Pleural protein | 4 ± 1 | 4 ± 1 | 0.7 |
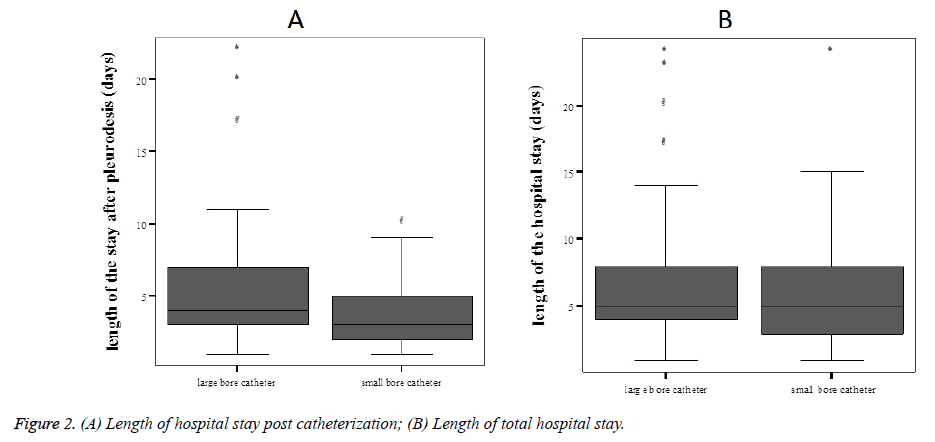
| Length of post-catheterization hospital stay(d) | 3.6 ± 2.2 | 5.3 ± 3.5 | <0.001 |
| Total length of hospital stay (d) | 6.5 ± 4.6 | 6.7 ± 4.5 | 0.89 |
| In-hospital death | 9 (13.4) | 15 (12.7) | 0.88 |
Table 1: Patient’s demographic and clinical characteristics.
In unadjusted analyses, post-catheterization LOS was significantly shorter in the small-bore catheter group (p<0.001), total LOS was not different between the small- and large-bore groups (p=0.89, Figures 2A and 2B).
Given concerns about confounding by indication, we developed a propensity score using age, gender, hypertension, cardiac failure and other types of cancers besides lung were chosen as propensity score calculation variables. The overlap range of propensity scores was found to be acceptable between the two groups. These groups were matched with regard to age, sex, pleural fluid parameters, comorbidities, hypertension, cardiac failure and other types of cancer. In this propensity analysis, 50 matched pairs were identified. The clinical and demographic characteristics of propensity score matched patients are shown in Table 2.
| Small-bore catheter group n:50 | Large-bore catheter group n:50 | P value | |
|---|---|---|---|
| Age | 63 ± 14 | 64 ± 14 | 0.64 |
| Gender (male) | 21 | 30 | 0.05 |
| Comorbidity (n) | 20 | 19 | 0.5 |
| Previous chemotherapy (n) | 30 | 28 | 0.42 |
| Lung carcinoma (n) | 32 | 32 | 0.91 |
| Adeno carcinoma | 16 | 15 | |
| Small cell carcinoma | 11 | 12 | 0.81 |
| Non-small cell carcinoma | 6 | 4 | |
| Mesotelioma | 7 | 8 | 0.9 |
| Breast carcinoma | 7 | 6 | |
| Lymphoma | 1 | 1 | |
| Kolon carcinoma | 2 | 2 | |
| Osteosarcoma | 0 | 1 | |
| Pancreas carcinoma | 1 | 0 | |
| Pleural LDH | 649 ± 952 | 877 ± 1520 | 0.37 |
| Pleural glucose | 99 ± 48 | 102 ± 52 | 0.79 |
| Pleural albumin | 3.6 ± 9.8 | 2.2 ± 0.7 | 0.33 |
| Pleural protein | 3.9 ± 1.1 | 4.1 ± 0.9 | 0.68 |
| Pleural ADA | 13 ± 11 | 16 ± 12 | 0.44 |
Table 2: Propensity score matched patient’s clinical and demographic characteristics.
Propensity score-adjusted linear regression analysis revealed that post-catheterization LOS was 1.8 d shorter in the smallbore catheter group (95% confidence interval: -0.9 to 2.8, p<0.001). The pain VAS score was significantly lower in small bore catheter group than in large bore catheter group (p<0.001, Table 3). Considering each group, pleurodesis as successful in 56 cases (83.6%) in small bore catheter group and in 103 cases (87.3%) in large bore catheter group. The total response rate was similar between the small-bore and large-bore catheter group and the success rates did not show a statistically significant difference (p: 0.68, Table 3).
| Small bore catheter group N: 50 | Large bore catheter group N: 50 | P value | |
|---|---|---|---|
| Pain VAS score (cm) | 3.9 ± 0.8 | 7.0 ± 0.8 | <0.001 |
| Post-catheterization LOS | 3.5 ± 2.2 | 5.4 ± 3.6 | <0.001 |
| Total LOS | 6.3 ± 4.5 | 7.6 ± 6.4 | 0.07 |
| Complete and partial response rate | 0.836 | 0.873 | 0.068 |
| VAS: Visual Analog Scale; LOS: Length of Hospital Stay. | |||
Table 3: Comparison of response rate, pain VAS score and length of hospital stay between propensity score matched catheter groups.
Discussion
In this study, we have shown that talc pleurodesis for malignant pleural effusion through a small-bore catheter resulted in a significantly shorter post-catheterization LOS as compared to those who received a large-bore catheter. Whereas the post-procedure LOS was 1.8 d shorter for small-bore catheter receiving patients, there was no difference between the two groups for total length of stay. This finding suggests that for small bore catheters receiving patients, there was a delay from time of hospitalization to time of procedure. The reasons for this are not clear, but may relate to the amount of effusion at time of admission. Nonetheless, the fact that post-procedure LOS was reduced is an important finding that supports the use of small-bore catheters, and ideally sooner during hospitalization. Although hospital stay cost data was not available in our study, the reduction in post-catheterization LOS and earlier discharge in the small-bore catheter group implies lower hospitalization costs, and additionally benefit to patients who often receive pleurodesis as a component of palliation care. In addition the mean pain VAS score was lower in small-bore catheter group than the large-bore catheter group. There is no statistically significant difference in total response between two groups. The response rates were similar.
Pleurodesis with large-bore (28 F to 36 F) or small-bore (7 F to 16 F) catheters is an important palliative procedure for recurrent pleural effusions due to malignant diseases. Usually, this procedure requires 5-7 d of hospitalization [11,12]. Once the drainage is decreased to 100-200 mL/d, a sclerosant agent is instilled. Talc is most often used due to its high success rate without relapse [4,13,14].
Boshuizen et al. described 381 patients with MPE who underwent pleurodesis. This cohort was largely comprised of females with breast cancer, and the median age was 61 y [15]. In another the study of chemical pleurodesis with iodopovidone, breast, lung, and ovary cancers were the most common malignancies with female dominance [10]. The mean age of our patients was similar to these two reports, but most of our patients were male, and lung cancer (adenocarcinoma) was the most common primary malignancy. These differences can be explained by the fact that our center is a pulmonary disease-specific hospital.
Patz et al. reported the first series of 19 patients with malignant effusions who underwent bleomycin pleurodesis using small-bore (10.3 F) catheters, and the length of catheterization was 2 to 11 d (mean 5.1 d) [16]. In a randomized controlled trial with talc pleurodesis, the mean LOS was 4.9 d in the chest tube group [17]. Fysh et al. reported longer pleural effusion-related hospital LOS (median, 10 d; interquartile range, 6-18 d) than previous studies of talc pleurodesis [18]. In 32 patients with known primary malignancies who underwent small-bore catheter for talc pleurodesis, the catheters remained in place for 2-9 d (mean 4.9 d) [9]. Catheterization time for both catheter types was similar in some previous studies. In a prospective analysis comparing large- and small-bore catheters for iodopovidone pleurodesis, the total tube durations were 5.6 and 5.7 d, respectively [10]. Another group concluded that tube duration and total LOS were not different between small- and large-bore catheter groups of patients with MPE [19]. Conversely, we found that patients in the small-bore catheter group had nearly 2 d shorter post-catheterization LOS compared to those in the large-bore catheter group. Interestingly, the secondary outcome of total LOS was not different between the two groups. We suspect that poor performance status of patients, large size of effusion, and loculated effusion are variables that could explain these discrepant findings. Correspondingly, it was reported that mean tube duration was significantly longer in patients with loculated pleural effusions than those with non-loculated effusions (means of 7.6 and 4.4 d, respectively) [9].
The main factors determining LOS are prolonged fluid drainage before pleurodesis and delayed chest tube removal after pleurodesis [20]. Difficulties with tube occlusion have been described when using chest tubes smaller than 12 F [20]. Conversely, we observed a shorter post-catheterization LOS for the small-bore catheter group. This suggests that the smaller size did not cause drainage difficulties, but we were unable to confirm this with data about the amount of fluid drained.
In the literature, increasing evidence shows that small bore catheters induce less pain to large bore catheters in the management of malignant effusion and pneomothoraces [21-23].
Conclusion
Pleurodesis with talc via small-bore catheter in malignant pleural effusion appears to result in a shorter post procedure length of hospital stay as compared to large-bore catheters, thus potentially reducing hospitalization costs. The success rates of pleurodesis were found to be similar regardless of the type of tube inserted. Pleurodesis via small-bore catheter seems to be less painful than the large-bore catheters.
Acknowledgement
The authors thank the American Thoracic Society for their support of Methods in Epidemiologic, Clinical and Operations Research (MECOR) Program, and specifically Dr. Payam Nahid, Dr. Ahmet Demir, Dr. Zuhal Karakurt for their invaluable help in the development of our research skills and our love for research.
References
- Marel M, Zrustova M, Stasny B. The incidence of pleural effusion in a well-defined region. Epidemiologic study in central Bohemia. Chest 1993; 104: 1486-1489.
- American Thoracic Society. Management of malignant pleural effusions. Am J Respir Crit Care Med 2000; 162: 1987-2001.
- Martinez-Moragon E, Aparicio J, Sanchis J. Malignant pleural effusion: prognostic factors for survival and response to chemical pleurodesis in a series of 120 cases. Respir 1998; 65: 108-113.
- Crnjac A, Sok M, Kamenik M. Impact of pleural effusion pH on the efficacy of thoracoscopic mechanical pleurodesis in patients with breast carcinoma. Eur J Cardiothorac Surg 2004; 26: 432-436.
- Rodriguez-Panadero F, Montes-Worboys A. Mechanisms of pleurodesis. Respir 2012; 83: 91-98.
- Ibrahim IM, Dokhan AL, El-Sessy AA. Povidone-iodine pleurodesis versus talc pleurodesis in preventing recurrence of malignant pleural effusion. J Cardiothorac Surg 2015; 10: 64.
- Xia H, Wang XJ, Zhou Q. Efficacy and safety of talc pleurodesis for malignant pleural effusion: a meta-analysis. Plos One 2014; 9: e87060.
- Roberts ME, Neville E, Berrisford RG. Management of a malignant pleural effusion: British Thoracic Society Pleural Disease Guideline. Thorax 2010; 65: 32-40.
- Marom EM, Patz EF, Erasmus JJ. Malignant pleural effusions: Treatment with small-bore catheter thoracostomy and talc pleurodesis. Radiol 1999; 210: 277-281.
- Caglayan B, Torun E, Turan D. Efficacy of iodopovidone pleurodesis and comparison of small-bore catheter versus large-bore chest tube. Ann Surg Oncol 2008; 15: 2594-2599.
- Filosso PL, Sandri A, Felletti G. Preliminary results of a new small bore percutaneous pleural catheter used for treatment of malignant pleural effusions in ECOG PS 3-4 patients. Eur J Surg Oncol 2011; 37: 1093-1098.
- Hunt BM, Farivar AS, Valliares E. Thoracoscopic talc versus tunnelled pleural catheters for palliation of malignant pleural effusions. Ann Thorac Surg 2012; 94: 1053-1059.
- Shaw P, Agarwal R. Pleurodesis for malignant pleural effusions. Cochrane Database Syst Rev 2004; 1: CD002916.
- Kilic D, Akay H, Kavukçu S. Management of recurrent pleural effusion with chemical pleurodesis. Surg Today 2005; 35: 634-638.
- Boshuizen RC, Burgers JA, Van den Heuvel MM. Comments on predictors of clinical use of pleurodesis and/or indwelling pleural catheter therapy for malignant pleural effusion. Chest 2015; 147: 232-233.
- Patz EF, McAdams HP, Goodman PC. Ambulatory sclerotherapy for malignant pleural effusions. Radiol 1996; 199: 133-135.
- Penz ED, Mishra EK, Davies HE. Comparing cost of indwelling pleural catheter vs. talc pleurodesis for malignant pleural effusion. Chest 2014; 146: 991-1000.
- Fysh ETH, Waterer GW, Kendall PA. Indwelling pleural catheters reduce inpatient days over pleurodesis for malignant pleural effusion. Chest 2012; 142: 394-400.
- Parulekar W, Primio GD, Matzinger F. Use of small bore and large bore chest tubes for treatment of malignant pleural effusions. Chest 2001; 120: 19-25.
- Sartori S, Tombesi P, Tassinari D. Sonographically guided small-bore chest tubes and sonographic monitoring for rapid sclerotherapy of recurrent malignant pleural effusions. J Ultrasound Med 2004; 23: 1171-1176.
- Kulvatunyou N, Erickson L, Vijayasekaran A, Gries L, Joseph B, Friese RF, Okeeeffe T, Tang AL,Wynne JL, Rhee P. Randomized clinical trial of pigtail catheter versus chest tube in injured patients with uncomplicated traumatic pneomothorax. Brit J Surg 2014; 101: 17-22.
- Fysh ET, Smith NA, Gary Lee YC. Optimal Chest Drain Size: The rise of the small-bore pleural catheter. Semin Respir Crit Care Med 2010; 31: 760-768.
- Horsley A, Jones L, White J, Henry M. Chest 2006; 130: 1857-1863.
- Sahin U, Unlü M, Akkaya A, Ornek Z. The value of small-bore thoracostomy in the treatment of malignant pleural effusions. Respir 2001; 68: 501-505.
- Parulekar W, Di Primio G, Matzinger F, Dennie C, Bociek G. Use of small-bore vs. large-bore chest tubes for the treatment of malignant pleural effusions. Chest 2001; 20: 19-25.
- Yildirim H, Metintas M, Ak G. Predictors of talc pleurodesis outcome in patients with malignant pleural effusions. Lung Cancer 2008; 62: 139-144.