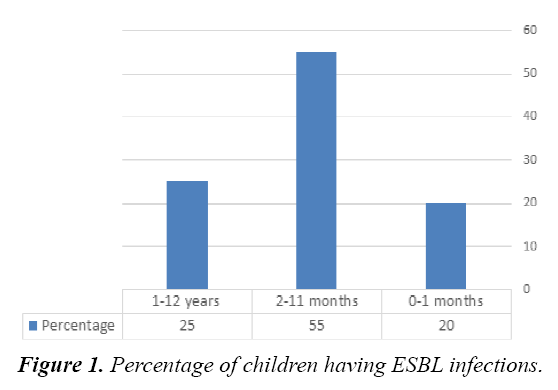Research Article - Current Pediatric Research (2018) Volume 22, Issue 4
Serious infection due to extended-spectrum β-lactamase producing bacteria in children.
Elham Bukhari*
Department of Pediatrics, King Saud Medical City, Saudi Arabia
- Corresponding Author:
- Elham Bukhari
Associate consultant Pediatrician, Department of Pediatrics
King Saud Medical City, Qurtuba street, 13244, Riyadh Saudi Arabia
Tel: +966114679963
E-mail: ebukhari@ksu.edu.sa
Accepted date: November 15, 2018
Abstract
Background: The prevalence of Extended Spectrum β-Lactamase (ESBL) producing bacteremia is increasing worldwide. The emergence ESBL-producing bacteria in children with severe infection like bacteremia and CNS infection needs further study. Here, we determine and characterize ESBL-producing bacteria in hospitalized children. Methods: Clinical and laboratory data of hospitalized pediatric patients aged 0-14 years diagnosed with ESBL-producing bacteria and serious infection (bacteremia and CNS infection) were collected over one year at King Khalid University Hospital. Results: We identified 20 episodes of ESBL-bacteremia among pediatric patients. Most patients were boys (n=17). Most 11 (55%) were 2-11 months of age, and preterm babies were almost half of the patient (50%). Two cases had serious CNS infection (brain abscess) caused by ESBL E. coli. Other examples include 14 K. pneumoniae (70%) isolates, 5 E. coli (25%) isolates and one Enterobacter aerogenes (5%). Bacteremia was diagnosed predominantly with K. pneumoniae (12 episodes) followed by E. coli (4 episodes) and E. aerogenes (1 episode). The ESBL-bacteremia documented in preterm babies was late-onset bacteremia. Three patients (15%) had underlying renal anomalies. Only one patient died (5%). Twelve patients (60%) were treated successfully with meropenem with or without amikacin. There was two dominant risk factor that occurred in all of the patient, Intensive care admission and prior usage of antibiotics. Conclusion: ESBL-producing bacteria tended to increase infection rates of both gramnegative bacteremia and brain abscesses in pediatric populations.
Keywords
Extended-spectrum β-lactamase, Bacteremia, Brain abscess.
Introduction
Extended-Spectrum β Lactamases (ESBLs) are a major group of enzymes that confer resistance to many types of β-lactam antibiotics including third generation cephalosporins (e.g., ceftazidime, cefotaxime, and ceftriaxone) [1,2]. Extended-Spectrum β-Lactamase (ESBL)-producing Gram-negative bacilli are an important cause of nosocomial infections [3,4]. The emergence of Gram-negative bacteria multidrug resistance producing infection is an important clinical problem worldwide. Enterobacteriaceae including Escherichia coli and Klebsiella pneumoniae are the dominant ESBL producing organisms isolated [5]. Treatment options for ESBLproducing Gram-negative bacteremia and CNS infection are often difficult because of the resistance to several antimicrobials including trimethoprim/sulfamethoxazole, aminoglycosides, tetracyclines, and quinolones [6]. Several studies have evaluated ESBL-producing bacteria urinary tract infections in Saudi Arabia [7,8]. However, there are few studies about the clinical features and prognosis of serious infection caused by ESBL-producing bacteria in children. Some studies have investigated the risk factors underlying infection and the clinical outcomes [9,10]. Other studies reported the risk factors for ESBL-producing Enterobacteriaceae and the antimicrobial resistance in a neonatal intensive care unit [11-16]. We conducted this study to assess the clinical features, associated risk factors, and prognosis of serious ESBL-producing bacterial infection in pediatric subjects.
Methodology
A case series study was conducted to explore the total number of episodes of bacteremia, CNS infection (meningitis/brain abscess) caused by ESBLproducing bacteria among pediatric patients aged 0-14 years between January 2017 and February 2018 at King Khalid University Hospital, a tertiary care center, with 150 pediatric beds located in Riyadh, KSA.
Demographic characteristics were collected from medical records, including sex, age, clinical status, underlying diseases, Risk factors like exposure to antibiotics, Central Venous Catheters (CVC), or the use of mechanical ventilation was recorded. The study was designed according the Institutional Review Board protocol.
Statistical analysis
All statistical analyses were performed using the SPSS version 15.0 (IBM SPSS Statistics, IBM Corporation, Armonk, NY, USA).
Microbiological Characterization
All blood samplings were ordered by the attending physicians based on suspected infection. Blood cultures were obtained via peripheral venous puncture (never through a CVC) and then performed using the BACTEC 9240 system. Antibiotic susceptibility patterns were determined according to methods recommended by the National Committee for Clinical Laboratory Standards Institute (CLSI) including the disk diffusion method; categorical assignment used CLSI breakpoints [17]. ESBL production was screened and confirmed in all isolates with a profile suggestive of resistance by performing a doubledisc synergy test according to CLSI guidelines [18].
Results
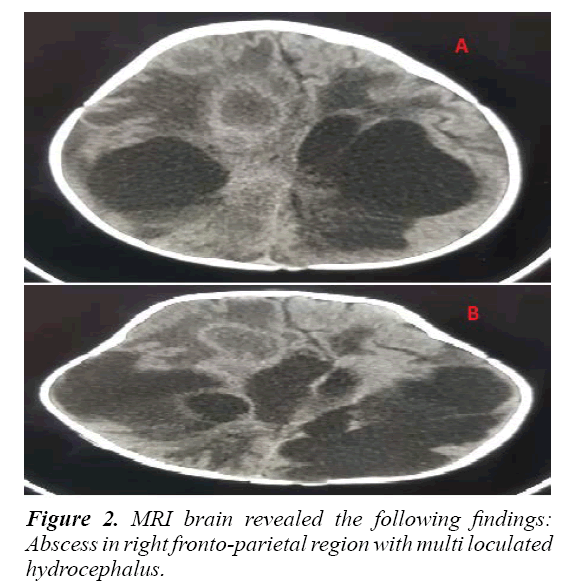
We enrolled 20 pediatric patients admitted in the hospital and diagnosed with ESBL Gram-Negative producing bacteremia and CNS infection. All patients were males except three; 55% were between 2-11 month of age (Figure 1), and 50% were preterm babies. Two patient were diagnosed with CNS infection (meningitis/brain abscess). The types of the isolated ESBL Gram–Negative bacteria were: K. pneumoniae was isolated from 14 cases (70%), E. coli was isolated from 5 (25%), and Enterobacter aerogenes was isolated from 1 (5%) (Table 1). Bacteremia was diagnosed predominantly with K. pneumoniae (12 episodes) followed by E. coli (4 episodes), and E. aerogenes with 1 episode. Two cases of brain abscess were seen-both with E. coli bateremia. Only one patient died (5%) (Table 2). Twelve patients were successfully treated with meropenum with or without amikacin, and 8 patients were treated with amikacin and tazocin. Three patients had underlying renal anomalies (hydronephrosis). One patient had neurodegenerative disease, and one patient had a malignancy (acute myeloid leukemia). One patient was diagnosed with cystic fibrosis. For the risk factor we found that, all patients 20 (100%) had previous admission to the intensive care unit. Mechanical ventilation was used in 8 (40%) patients, and all 20 (100%) had prior use of antibiotics. Around 12 (60%) patient had Central Venous Catheters (CVC) inserted for at least 2 weeks. Two patients had bacteremia complicated with meningitis and brain abscess. The first was a preterm baby with E. coli bacteremia complicated by ventriculitis and brain abscess. Brain MRI revealed an abscess in the right fronto-parietal region with multi loculated hydrocephalus (Figures 2A and 2B). Aspiration of the abscess showed growth of E. coli. The abscess was drained, and an occipital VP shunt was inserted after treating with appropriate antibiotics.
| Species | N (%) | Blood | CSF | Wound | Ascetic Fluid |
|---|---|---|---|---|---|
| K. pneumoniae | 14 (70) | 12 | 0 | 3 | 1 |
| E. coli | 5 (25) | 4 | 2 | 1 | 0 |
| E. aerogenes | 1 (5) | 1 | 0 | 0 | 0 |
Table 1: Species and specimen distribution among ESBL infected children.
| Characteristics | Percentage |
|---|---|
| Sex | |
| Male | 17 (85%) |
| Female | 3 (15%) |
| Age | |
| 0-1 month | 4 (20%) |
| 2-11 month | 11 (55%) |
| 1-13 year | 5 (25%) |
| Types and number of ESBL bacteria | |
| Klebsiella pneumoniae | 14 (70%) |
| E. coli | 5 (25%) |
| Enterobacter aerogenes | 1 (5%) |
| Risk factors | |
| Prematurity | 10 (50%) |
| Admission to Intensive Care (ICU) | 20 (100%) |
| Prior Antibiotics | 20 (100%) |
| Central Venous Line (CVC) | 12 (60%) |
| Mechanical Ventilation | 8 (40%) |
| Renal Disease | 3 (15%) |
| Mortality | 1 (5%) |
Table 2: Demographic and clinical characteristics of all study subjects, including 20 episodes of serious infection due to ESBL producing bacteria.
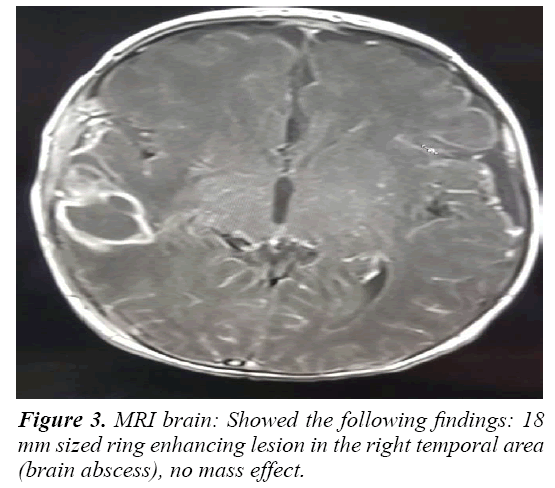
The second patient was a term baby referred from a private hospital. He had a history of E. coli bacteremia in the first week of life and after a short course of antibiotics (Gentamicin and Ampicillin). Within 48 hours of stopping the antibiotics, he developed a fever and was diagnosed to have CNS infection. Both blood and cerebrospinal fluid CSF showed growth of ESBL E. coli. Brain MRI showed an 18 mm sized ring-enhancing lesion in the right temporal area (brain abscess) with no mass effect (Figure 3). After 4 weeks of treatment, a repeat MRI brain revealed remarkable improvement in the brain abscess. The patient received 12 weeks of antibiotics and was discharged home in good condition.
Discussion
ESBL producing bacteria have been described in Saudi Arabia in the eastern province the prevalence of Enterobacteriaceae isolates was 10.1% [19]. These Enterobacteriaceae isolates have with a high level of carbapenem resistance (20%) [20]. In another study in central Saudi Arabia, 57% of ESBL-producing E. coli isolates were of community origin [21]. In a Taiwanese study by Tsai et al., ESBL Gram-negative bacteria accounted for one-seventh of all neonatal Gram-negative bacteremia particularly in neonates who received broadspectrum cephalosporins. Neonates with ESBL producing Gram-negative bacteremia were associated with a higher rate of infectious complications and adverse outcomes [22]. Shaikh et al. and his colleague noted that 55.69% (n=93) of E. coli and 44.31% (n=74) of K. pneumoniae isolates were ESBL producers. In contrast, we noted that ESBL production was more common in K. pneumoniae isolates than E. coli isolates [23]. The clinical characteristics and risk factors of preterm babies in this study agreed with other studies that investigated ESBL producing isolates in infants admitted to a neonatal intensive care unit [13- 16]. Brain abscess is rare in neonates. The most commonly implicated organisms causing neonatal brain abscess are Citrobacter koseri and Proteus. Feather cut, Layer cut, straight cut, U and V cut, Hair colouring, these rarely produce ESBL [24]. While E. coli is a common cause of neonatal sepsis in newborns, it rarely causes brain abscesses [25]. ESBL-producing K. pneumoniae species was the main causative organism. Some of these cases led to brain abscesses in neonates [26].
Conclusion
ESBL-producing isolates causes severe infections that can lead to increased morbidity and mortality; however, only one patient (10%) in this study died, this is a significant finding. Prematurity, intensive care admission, and prior exposure to antibiotics were significant risk factors.
References
- Drawz SM, Papp-Wallace KM, Bonomo RA. New beta-lactamase inhibitors: A therapeutic renaissance in an MDR world. Antimicrob Agents Chemother 2014; 58: 1835-46.
- Rawat D, Nair D. Extended-spectrum beta-lactamases in gram negative bacteria. J Glob Infect Dis 2010; 2: 263-74.
- Badal RE, Bouchillon SK, Lob SK, et al. Etiology, extended-spectrum β-lactamase rates, and antimicrobial susceptibility of Gram-negative bacilli causing intra-abdominal infections in patients in general pediatric and pediatric intensive care units-Global data from the Study for Monitoring Antimicrobial Resistance Trends (SMART) 2008-2010. Pediatr Infect Dis J 2013; 32: 636-40.
- Katsuta T, Shoji K, Watanabe Y, et al. (2013) Treatment of pyelonephritis caused by extended spectrum β-lactamase-producing entero-bacteriaceae in children. Pediatr Infect Dis J 2013; 32: 417-9.
- Shaikh S, Fatima J, Shakil S, et al. Antibiotic resistance and extended spectrum beta-lactamases: Types, epidemiology and treatment. Saudi J Biol Sci 2015; 22: 90-101.
- Paterson DL, Bonomo RA. Extended-spectrum-β-lactamases: A clinical update. Clin Microbiol Rev 2005; 18: 657-86.
- Alyamani EJ, Khiyami AM, Booq RY, et al. The occurrence of ESBL producing Escherichia coli carrying aminoglycoside resistance genes in urinary tract infections in Saudi Arabia. Ann Clin Microbiol Antimicrob 2017; 16: 1.
- Mashwal FA, El-Safi SH, George SK, et al. Incidence and molecular characterization of the extended spectrum beta lactamase-producing Escherichia coli isolated from urinary tract infections in Eastern Saudi Arabia. Saudi Med J 2017; 38: 811-5.
- Memon JI, Rehmani RS, Ahmed MU, et al. Extended spectrum beta-lactamase producing Escherichia coli and Klebsiella pneumoniae bacteremia: Risk factors and outcome in the eastern region of Saudi Arabia. Saudi Med J 2009; 3: 803-6.
- Panhotra BR, Saxena AK, Al-Ghamdi AM. Extended-spectrum betalactamase-producing Klebsiella pneumoniae hospital acquired bacteremia: Risk factors and clinical outcome. Saudi Med J 2004; 25: 1871-6.
- Somily AM, Alsubaie SS, BinSaeed AA, et al. Extended spectrum β-lactamase-producing Klebsiella pneumonia in the neonatal intensive care unit: Does vancomycin play a role?. Am J Infect Control 2014; 42: 277-82.
- Giuffré M, Cipolla D, Bonura C, et al. Outbreak of colonization by extended-spectrum β-lactamase Escherichia coli sequence type 131 in a neonatal intensive care unit, Italy. Antimicrob Resist Infect Control 2013; 2: 8.
- Shakil S, Akram M, Ali SM, et al. Acquisition of extended spectrum β-lactamase producing Escherichia coli strains in male and female infants admitted to a neonatal intensive care unit: Molecular epidemiology and analysis of risk factors. J Med Microbiol 2010; 59: 948-54.
- Singh N, Patel KM, Léger MM, et al. Risk of resistant infections with Enterobacteriaceae in hospitalized neonates. Pediatric Infect Dis J 2002; 21: 1029-33.
- Linkin DR, Fishman NO, Patel JB, et al. Risk factors for extended-spectrum beta-lactamase-producing Enterobacteriaceae in a neonatal intensive care unit. Infect Control Hosp Epidemiol 2004; 25: 781-3.
- Le J, Nguyen T, Okamoto M, et al. Impact of empiric antibiotic use on development of infections caused by extended spectrum beta-lactamase bacteria in a neonatal intensive care unit. Pediatric Infect Dis J 2008; 27: 314-8.
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: Twenty-second informational supplement M100-S22. CLSI, Wayne, PA, USA. 2012.
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial disk susceptibility tests: Approved standard-tenth informational supplement M02-A10. CLSI, Wayne, PA, USA. 2009.
- Kader AA, Kumar AK. Prevalence of extended spectrum betalactamase among multidrug resistant gram-negative isolates from a general hospital in Saudi Arabia. Saudi Med J 2004; 25: 570-4.
- Kandeel A. Prevalence and risk factors of extended-spectrum -lactamases producing Enterobacteriaceae in a general hospital in Saudi Arabia. J Microbiology and Infectious Diseases 2014; 4: 50-4.
- Al-Otaibi FE, Bukhari EE. Clinical and laboratory profiles of urinary tract infections caused by extended-spectrum beta-lactamase producing Escherichia coli in a tertiary care center in central Saudi Arabia. Saudi Medical Journal 2013; 34: 171-6.
- Tsai MH, Lee IT, Chu SM, et al. Clinical and molecular characteristics of neonatal extended spectrum β-lactamase-producing gram-negative bacteremia: A 12-year case-control-control study of a referral center in Taiwan. PLoS One 2016; 9: e0159744.
- Shaikh S, Fatima J, Shakil S, et al. Risk factors for acquisition of extended spectrum beta lactamase producing Escherichia coli and Klebsiella pneumoniae in North-Indian hospitals. Saudi J Biol Sci 2015; 22: 37-41.
- Biswas B, Mondal M, Thapa R, et al. Neonatal brain abscess due to extended-spectrum beta-lactamase producing Klebsiella pneumoniae. J Clin Diagn Res 2014; 8: PD01-2.
- Erdemir A, Kahramaner Z, Cosar H, et al. Escherichia coli brain abscess in a twin pair associated with TLR4 gene mutation. Pediatr Int 2013; 55: 516-8.
- Wojsyk-Banaszak I, Szczapa J. Central nervous system infections in neonates caused by multiresistant Klebsiella pneumoniae. Ginekol Pol 2000; 71: 975-8.