Research Article - Biomedical Research (2017) Volume 28, Issue 20
Role of YAP-siRNA in reverse sensitivity of human lung cancer A549/TAX cells to taxol and its possible mechanism
Dan Xue1,2, Qiongying Wei1,2, Xiaoping Li1,2 and Tingyan Lin1,2*
1Department of Respiratory Medicine, Fujian Medical University Union Hospital, Fuzhou, China
2Department of Geriatrics, Fujian Medical University Union Hospital, Fuzhou, China
- *Corresponding Author:
- Tingyan Lin
Department of Respiratory Medicine
Fujian Medical University Union Hospital, China
Accepted on September 21, 2017
Abstract
Lung cancer is a most common malignancy in both men and women. Studies have indicated that YAP gene was closely related to chemo-resistance of cancers. However, whether YAPS expression participates in the resistance of human lung cancer A549 cells to Taxol (TAX) remains unclear. The aim of this study is to investigate the role of YAP in the resistance of A549 cells to TAX. The sensitivity of cells to TAX was firstly confirmed by MTT assay. The expression levels of YAP in A549 cells and A549/TAX cells were examined by QRT-PCR and Western blot. Small interference RNAs (siRNAs) were used to knock down the gene expression of YAP in A549/TAX cells. Then the cell viability and apoptosis were assessed following treatment with taxol using MTT assay and flow cytometry. The levels of drug resistance related genes, MDR1, MRP1 and BCRP, were examined in YAP-siRNA A549/TAX cells. Furthermore, Western blot analysis was utilized to determine ERK pathway following YAP knockdown. The results demonstrated that the expression of YAP was significantly increased in A549/TAX cells. YAP gene knockdown significantly decreased the IC50 value of A549/TAX cells to TAX and increased taxolinduced apoptosis in the A549/TAX cells. The expression of MDR1, MRP1 and BCRP significantly decreased, and YAP knockdown markedly decreased the activation of ERK pathway. In conclusion, YAP knockdown could reverse the resistance of A549/TAX cells to taxol, thus it may act as a candidate for therapeutic target of lung cancer.
Keywords
YAP, Lung cancer, Chemo-resistance, Taxol, MEK/ERK pathway.
Introduction
Lung cancer is the most common cancer in the world with more than 1 million cases diagnosed every year, and remains the second leading cause of cancer death in both men and women [1]. Advanced Non-Small Cell Lung Carcinoma (NSCLC) accounts for >80% of lung cancer cases [2]. More than half of these patients have developed metastasis at the time of diagnosis and chemotherapy remains the most effective treatment option [3]. Platinum-based doublets are considered to be the standard treatments for NSCLC [4]. The clinical effectiveness of the chemotherapeutic agents used in the treatment of NSCLC is often negated by drug resistance [5,6].
Yes-Associated Protein (YAP) was first discovered and named in 1994, it is a multifunctional intra-cellular junction protein and transcriptional co-activator. YAP plays important roles in regulating signal transduction and gene transcription in normal cells. Studies have confirmed that YAP is the target molecules in Hippo pathway which is a newly discovered cell signal transduction pathway and plays a critical role in regulating the volume and size of organs [7,8]. Researches have shown that YAP was over-expressed in a variety of cancers including liver cancer, gastric cancer, ovarian cancer, prostate cancer, lung cancer, etc., and it was related to the differentiation, proliferation, metastasis, and chemotherapy sensitivity, indicating that YAP is involved in the development of tumor [9-13]. Recent studies indicated that YAP could confer the resistance of cancers to chemotherapy drugs [14], and high expression of YAP was associated with poor patient prognosis [15].
Taxol is an important anti-tumor agent that is particularly effective in the treatment of ovarian and breast cancers, although its usefulness is often hampered by the development of drug resistance. As a new anti-tubulin drug [16], taxol can enhance the binding ability of 14-3-3 protein with YAP without changes of tublins stability and YAP phosphorylation status [17]. This limits the function of YAP as a transcription factor. However, the suppression of taxol on YAP function can be reversed by YAP up-regulation [18].
Based on previous reports, we hypothesized that YAP may be aberrantly expressed in lung cancer cells with chemo-resistant ability and regulation of YAP may affect the chemo-sensitivity of lung cancer cells to chemotherapeutic agents. In this study, we firstly evaluated the YAP expression in A549 (normal lung cancer cells) and A549/TAX (lung cancer cells resistant to taxol). Then YAP-siRNA was transfected into A549/TAX cells to decrease the expression of YAP. The cellular sensitivity to taxol was measured by MTT assay and cell apoptosis was examined by flow cytometry. The underlying molecular mechanism was revealed through QRT-PCR and Western blot.
Materials and Methods
Cells and cell culture
The human non-small cell lung cancer (NSCLC) A549 cell line and the taxol-resistant A549/TAX cell line were purchased from American Type Culture Collection (Manassas, VA, USA) and were cultured in RPMI-1640 medium (Gibco, Gaithersburg, MD, USA) supplemented with 10% Fetal Bovine Serum (FBS) (Cor., New York, NY, USA), 100 mg/ml streptomycin and 100 U/ml penicillin, and cells were incubated in a humidified atmosphere with 5% CO2 at 37°C. Cells were passaged every 2-3 d.
Cell transfection
For cell transfection, Lipofectamine2000 transfection reagent (Invitrogen) was used. Negative control-siRNA or YAP-siRNA (Santa cruz, CA, USA) was transiently transfected into A549/TAX cells using 20μl transfection reagent according to the manufacturer’s instructions. Cells were incubated for 48 h before the following tests.
MTT cell viability assay
To determine the drug resistance of taxol to A549 cells, halfmaximal inhibitory concentration (IC50) values were calculated. Cells were seeded into a 96-well plate and cultured in RPMI-1640 media for 24 h, then the cells were treated with various concentrations of taxol (Sigma, Santa Clara, Utah, USA) for 48 h. Subsequently, MTT solution (5 mg/ml) (Eli Lilly, Co. Ltd., Indianapolis, IN, USA) was added to culture medium and incubated at 37°C for 4 h. The Optical Absorbance (OD) at 450 nm was measured using a SynergyTM 2 Multi-function Microplate Reader (Bio-Tek Instruments, Winooski, VT, USA). Each experiment was independently repeated three times.
Flow cytometry assay
Annexin V/propidium iodide double staining method was performed to examine cell apoptosis. 48 h after cell transfection, cells were treated with 5 μl TAX for 24 h. Then cells were labeled with annexin V-FITC and Propidium Iodide (PI) for cell apoptotic rate detection following the manufacturer’s instructions, and flow cytometry (Becton Dickinson, New Jersey, USA) was performed. Each experiment was independently repeated three times.
Western blot analysis
Total cellular protein was extracted using RIPA buffer containing 1 mM Phenylmethanesulfonyl Fluoride (PMSF), and equal protein samples were standardized using spectrophotometer. Samples were resolved by using SDSPAGE analysis and transferred to Polyvinylidene Fluoride (PVDF) membranes (Millipore). The membranes were blocked at room temperature for 1.5 h in PBS solution containing 0.1% Tween-20 and 5% fat-free milk, and incubated overnight with primary antibody (1:1000) at 4°C followed by secondary antibody (1:5000) at room temperature for 2 h. The TS380-H automatic washing machine (Taixing TaiSheng Medical Equipment Factory, China) was used to analyse the protein bands.
QRT-PCR analysis
After certain treatment, qRT-PCR was performed for gene mRNA expression level detection. GAPDH acted as an internal control. Total RNA was obtained from cells using TRIZOL reagent (Takara, Japan) in accordance with the manufacturer’s instructions. Then, RNA was transcribed into cDNA using RTPCR detection kit (Invitrogen, Carlsbad, CA, USA) according to manufacturer’s instructions. Real-time PCR was performed to analyse the synthesized cDNA and the conditions were 95°C for 10 min, followed by 40 cycles of 95°C for 10 s, 60°C for 60 s. All the primers were performed by Sangon Biotech (Shanghai, China). The primer sequences of real-time PCR are as shown in Table 1.
| Sequence (5'-3') |
|---|
| YAP-forward: 5'ACGTTCATCTGGGACAGCAT3' |
| YAP-reverse: 5'GTTGGGAGATGGCAAAGACA3' |
| GAPDH-forward: 5'CTTTGGTATCGTGGAAGGACTC3' |
| GAPDH-reverse: 5'GTAGAGGCAGGGATGATGTTCT3' |
Table 1. Primer sequence for PCR.
Statistical analysis
All quantitative data were displayed as the mean ± Standard Deviation (SD). Student's t-test was used to identify significant differences in multiple comparisons. For all statistical analyses, P<0.05 was considered statistically significant.
Results
Yap is upregulated in taxol resistant lung cancer cells
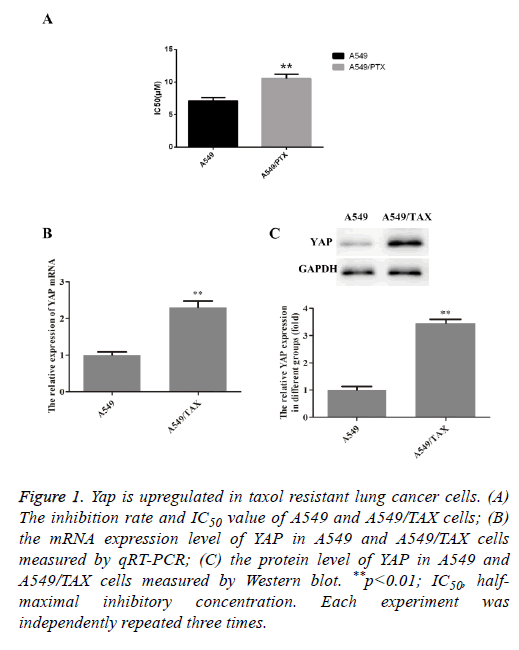
To confirm the taxol resistant ability of A549/TAX cell line, which was obtained from ATCC, MTT assay were firstly performed on A549 and A549/TAX cell lines after treated with taxol. As shown in Figure 1A, a significant inhibiting effect of taxol on cell viability was observed in A549 cell line, thus suggesting that A549 cells were more sensitive to taxol. That is, the obtained A549/TAX cell line was resistant to taxol as expect. To investigate the biological role of YAP in the taxol resistance of non-small cell lung carcinoma, we compared the expression level of YAP in A549 and A549/TAX. The results were shown in Figures 1B and 1C. Compared with A549 cells, the YAP expression was significantly upregulated in A549/TAX cells, indicating a possible involvement of YAP in drug resistance in lung cancer, especially NSCLC.
Figure 1: Yap is upregulated in taxol resistant lung cancer cells. (A) The inhibition rate and IC50 value of A549 and A549/TAX cells; (B) the mRNA expression level of YAP in A549 and A549/TAX cells measured by qRT-PCR; (C) the protein level of YAP in A549 and A549/TAX cells measured by Western blot. **p<0.01; IC50, halfmaximal inhibitory concentration. Each experiment was independently repeated three times.
Decrease of YAP mRNA and protein expression after YAP-siRNA
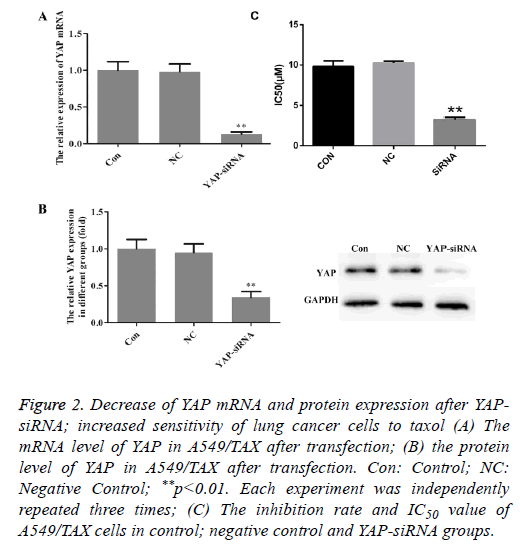
To further investigate the role of YAP in A549/TAX cells, YAP gene was knockdown using siRNA technology. The YAP mRNA and protein expression was examined by QRT-PCR and Western blot after siRNA transfection. As shown in Figure 2, the mRNA and protein expression levels were decreased to a relatively low level after transfection. It was considered that YAP gene expression was efficiently suppressed and siRNA transfection could be used as an effective method to inhibit the expression of YAP.
Figure 2: Decrease of YAP mRNA and protein expression after YAPsiRNA; increased sensitivity of lung cancer cells to taxol (A) The mRNA level of YAP in A549/TAX after transfection; (B) the protein level of YAP in A549/TAX after transfection. Con: Control; NC: Negative Control; **p<0.01. Each experiment was independently repeated three times; (C) The inhibition rate and IC50 value of A549/TAX cells in control; negative control and YAP-siRNA groups.
Increased sensitivity of lung cancer cells to taxol after YAP-siRNA transfection
MTT assay was utilized to determine the change of sensitivity to taxol after silence of YAP expression in A549/TAX cells. As shown in Figure 2C, a significant inhibiting effect of taxol on cell viability was observed in YAP-siRNA transfection group, indicating that YAP knockdown significantly increased the sensitivity of A549/TAX cells to taxol.
Increase of apoptosis in YAP-knockdown A549/TAX cells
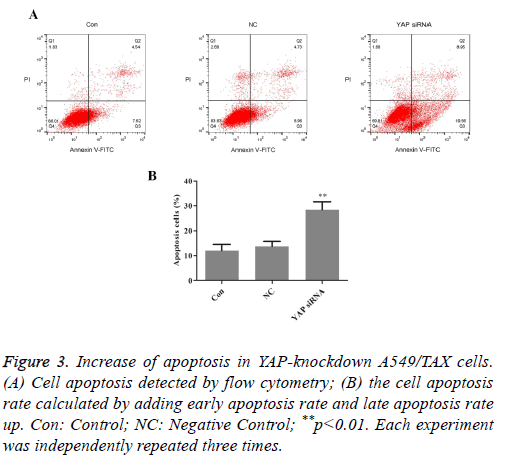
The effects of YAP knockdown on cell apoptosis in the A549/TAX cells were evaluated by flow cytometric analysis. The results were exhibited in Figure 3. After treated with taxol, the apoptosis rates were 12.16% in the parental A549/TAX cells and 13.69% in the A549/TAX cells transfected with control siRNA (P>0.05), whereas the apoptotic cells were 28.51% in A549/TAX YAP-siRNA cells (P<0.05). Compared with parental A549/TAX cells, the number of apoptotic cells was markedly increased in the YAP-knockdown A549/TAX cells. The results demonstrated that A549/TAX cells with downregulation of YAP were more sensitive to taxol compared with the control group.
Figure 3: Increase of apoptosis in YAP-knockdown A549/TAX cells. (A) Cell apoptosis detected by flow cytometry; (B) the cell apoptosis rate calculated by adding early apoptosis rate and late apoptosis rate up. Con: Control; NC: Negative Control; **p<0.01. Each experiment was independently repeated three times.
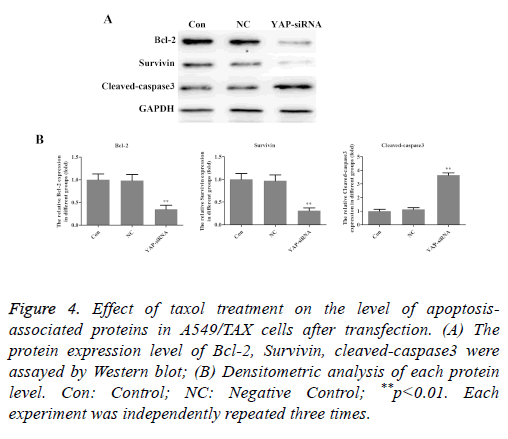
In order to investigate the mechanisms of sensitization induced by taxol in the A549/TAX cells, the expression levels of apoptosis-associated proteins were investigated following taxol treatment. As shown in Figure 4, YAP knockdown significantly decreased the expression level of Bcl-2 and survivin in A549/TAX cells, while the expression level of cleavedcaspase- 3 was increased remarkedly. In short, these results indicated that YAP is an inducer of taxol resistance and involves in regulating cell apoptosis.
Figure 4: Effect of taxol treatment on the level of apoptosisassociated proteins in A549/TAX cells after transfection. (A) The protein expression level of Bcl-2, Survivin, cleaved-caspase3 were assayed by Western blot; (B) Densitometric analysis of each protein level. Con: Control; NC: Negative Control; **p<0.01. Each experiment was independently repeated three times.
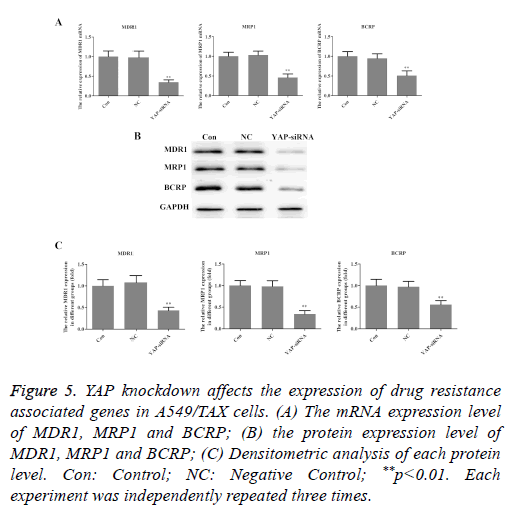
YAP knockdown affects the expression of drug resistance associated genes in A549/TAX cells
To explore the underlying molecular mechanisms of the roles that YAP played in taxol resistance, the expression of drug resistance related genes, MDR1, MRP1 and BCRP was examined in A549/TAX cells. As illustrated in Figure 5, both the mRNA and protein levels of MDR1, MRP1 and BCRP were decreased in A549/TAX YAP-siRNA cells. These findings suggested that YAP may induce taxol resistance through regulating drug transportation.
Figure 5: YAP knockdown affects the expression of drug resistance associated genes in A549/TAX cells. (A) The mRNA expression level of MDR1, MRP1 and BCRP; (B) the protein expression level of MDR1, MRP1 and BCRP; (C) Densitometric analysis of each protein level. Con: Control; NC: Negative Control; **p<0.01. Each experiment was independently repeated three times.
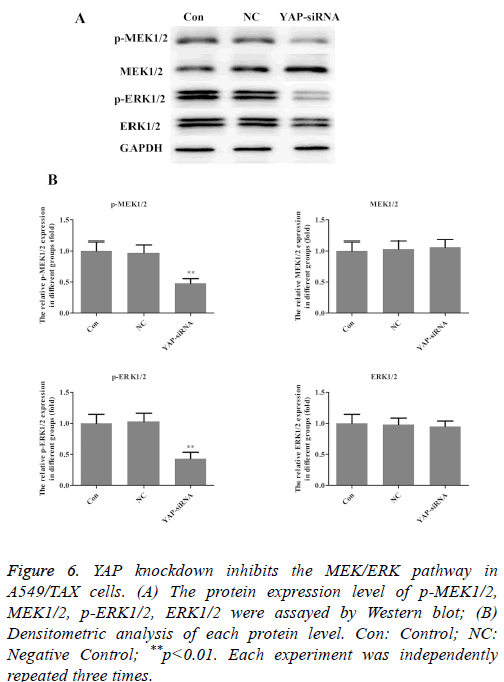
YAP knockdown inhibits the MEK/ERK pathway in A549/TAX cells
Aberrant activation of the ERK pathway has been reported in various carcinomas and it plays a critical role in carcinogenesis by regulating cell proliferation, invasion and migration [19-21]. To further elucidate the mechanism involved in the effect of YAP on taxol resistance in lung cancer, MEK/ERK pathway was explored further. By Western blot analysis, we found that activation (phosphorylation) of MEK and ERK was reduced in YAP-siRNA group (Figure 6). That is, YAP knockdown inhibits the MEK/ERK pathway in A549/TAX cells. The results indicated that MEK/ERK pathway participated in the effect of YAP on taxol resistance in lung cancer.
Figure 6: YAP knockdown inhibits the MEK/ERK pathway in A549/TAX cells. (A) The protein expression level of p-MEK1/2, MEK1/2, p-ERK1/2, ERK1/2 were assayed by Western blot; (B) Densitometric analysis of each protein level. Con: Control; NC: Negative Control; **p<0.01. Each experiment was independently repeated three times.
Discussion
Drug resistance is the major factor that affects the efficacy of chemotherapy in patients with lung cancer. There are many factors that affect the resistance of NSCLC drug resistance, including the increases of the expression of resistant multi-drug (MDR) gene, multidrug resistance associated protein (MRP) genes and their product, as well as the decreased expression of Topoisomerase II (Topo II) and Glutathione (GSH) and the increased expression of Glutathione S Transferase (GST) system. In addition, studies have shown that the abnormal expression of related factors in cell signal transduction, the abnormal of DNA repair in tumor cells and the aberrant expression of other related genes are closely related to lung cancer drug resistance [22]. Therefore, exploring novel therapeutic targets and effective molecular markers to predict the sensitivity of patients to drugs is extremely critical for NSCLC treatment.
YAP, a critical effecter of Hippo signaling pathway, is one of the major transcription activation factors and is highly active. Studies have suggested that YAP is highly expressed in both nuclear and cytoplasm of lung cancer cells and involved in the development of lung cancer [23]. Recently, a growing number of evidence indicated that the expression of YAP may associate with multi-drug resistance [24]. However, the relationship of YAP gene expression and Taxol chemoresistance in human lung cancer cells is still unclear. In the present study, the expression of YAP was significantly elevated in A549/TAX cell line both in protein and gene levels, thus indicating a possible involvement of YAP in drug resistance in lung cancer.
As an effective antitumor drug, taxol is an effective therapeutic agent to various tumor cells and it can limit the function of YAP as a transcription factor [25]. In order to precisely and definitely elucidate the role of YAP on the resistance of taxol in lung cancer, we suppressed YAP gene by siRNA in A549/TAX cells and then examined the chemo-sensitivity to taxol of A549/TAX cells. We found that YAP knockdown significantly increased the sensitivity of A549/TAX cells to taxol. Intrinsic or acquired drug resistance limits the effectiveness of chemotherapy. One of the most studied mechanisms of drug resistance in cancer cells is associated with altered anticancer drug transport, mediated by members of the ATP-Binding Cassette (ABC) superfamily of transport protein such as Multidrug Resistance Protein-1 (MRP1) and Breast Cancer Resistance Protein (BCRP) [26-28]. This resistant type has been termed Multidrug Resistance (MDR) [29]. Multi Drug Resistant Protein 1 (MDR1) has been linked to multi drug resistance and disease progression in cancers such as NSCLC [30]. Overexpression of MRP1 has been detected in a variety of tumor types, and has also been associated with drug resistance or poor patient outcome in lung cancer [31]. BCRP, which is a novel ABC transport protein, functions as an energy-dependent efflux pump. Elevated expression of BCRP in vitro causes resistance against at least two classes of anticancer drugs, anthracyclines and topoisomerase I inhibitors [32]. In the present study, we found that both the mRNA and protein levels of MDR1, MRP1 and BCRP were decreased in A549/TAX YAP-siRNA cells, thus suggesting YAP may induce taxol resistance through regulating drug transportation.
Apoptosis is cell suicide under the control of the activities of individual development. Apoptosis disorder is an important reason for tumorigenesis. caspases play the role of initiators and executioners in programmed death. And caspase-3 is directly responsible for cell demise [33]. As a well-known member of the inhibitor of apoptosis protein family, Survivin is completely down-regulated and undetectable in normal adult tissue and becomes prominently overexpressed in many human malignancies, including NSCLC [34]. The anti-apoptotic proteins Blc-2 serving as a binding groove for pro-apoptotic factors belongs to the Bcl-2 family, which is regarded as a central regulator of the apoptosis process [35]. Literatures have revealed one of the main mechanisms of chemoresistance is related to defects in the apoptotic pathway especially the alteration of apoptotic markers caspase-3, survivin and Blc-2 [36,37]. In this study, YAP knockdown significantly decreased the expression level of Bcl-2 and survivin in A549/TAX cells, while the expression level of cleaved-caspase-3 was increased remarkedly, which indicating YAP may involve in the regulation of cell apoptosis in A549/TAX cells.
The MEK/ERK signaling pathway is one of the bestcharacterized kinase cascades in cancer cell biology [38]. MEK1 and MEK2 are tyrosine and serine/threonine dualspecificity kinases and subsequently phosphorylate ERK1 and ERK2, resulting in their activation. Upon activation, they activate multiple nuclear and cytoplasmic targets (>600), including transcription factors, kinases, phosphatases and cytoskeletal proteins [39]. Taru et al. [40] have revealed that the activity of MEK/ERK pathway positively influences drug resistance and YAP can be regulated at several levels to promote resistance to multiple targeted therapies including through MEK/ERK pathway. In our experiments, we found that phosphorylation of MEK and ERK were reduced in YAPsiRNA group, which suggested YAP knockdown inhibits the MEK/ERK pathway in A549/TAX cells.
In conclusion, we show that YAP knockdown could reverse the resistance of A549/TAX cells to TAX. Therefore, YAP may be used as a novel therapeutic target in the treatment of lung cancer in the future, as well as an effective molecular marker to predict the sensitivity of patients to drugs, which can better guide the individual treatment, improve the efficacy of chemotherapy.
Conclusion
Acknowledgements
This study was sponsored by the funds from National and Fujian Province’s Key Clinical Specialty Discipline Construction Programs and Fujian Natural Science Foundation (No.2017J01295).
Declaration of Conflict of Interest
None
References
- Torre LA, Bray F, Siegel RL. Global cancer statistics 2012. Cancer J Clin 2015; 65: 87-108.
- Collins LG, Haines C, Perkel R, Enck RE. Lung cancer: diagnosis and management. Am Fam Physician 2007; 75: 56-63.
- Reck M, Heigener DF, Mok T, Soria JC, Rabe KF. Management of non-small-cell lung cancer: recent developments. Lancet 2013; 382: 709-719.
- Giaccone G. Clinical perspectives on platinum resistance. Drugs 2009; 59: 9-17.
- Sequist LV, Yang JC, Yamamoto N, OByrne K, Hirsh V; Mok T. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol 2013; 31: 3327-3334.
- Inoue A, Kobayashi K, Maemondo M, Sugawara S, Oizumi S, Isobe H. Updated overall survival results from a randomized phase III trial comparing gefitinib with carboplatin-paclitaxel for chemo-naïve non-small cell lung cancer with sensitive EGFR gene mutations (NEJ002). Ann Oncol 2013; 24: 54-59.
- Liu AM, Xu Z, Luk JM. An update on targeting Hippo-YAP signaling in liver cancer. Expert Opin Ther Targets 2012; 16: 243-247.
- Hong L, Cai Y, Jiang M, Zhou MT, Chen DWL. The Hippo signaling pathway in liver regeneration and tumorigenesis. Acta Biochim Biophys Sin (Shanghai) 2015; 47: 46-52.
- Xu MZ, Yao TJ, Lee NP, Ng IOL, Chan YT, Zender L. Yes-associated protein is an independent prognostic marker in hepatocellular carcinoma. Cancer 2009; 115: 4576-4585.
- Zhang J, Xu ZP, Yang YC, Zhu JS, Zhou CWX. Expression of Yes-associated protein in gastric adenocarcinoma and inhibitory effects of its knockdown on gastric cancer cell proliferation and metastasis. Int J Immunopathol Pharmacol 2012; 25: 583-590.
- Zhang X, George J, Deb S, Degoutin JL, Takano EA, Fox SB. The Hippo pathway transcriptional co-activator, YAP, is an ovarian cancer oncogene. Oncogene 2011; 30: 2810-2822.
- Zhang L, Yang SP, Chen XC, Seth S, Yu F, Subodh ML. The hippo pathway effector YAP regulates motility, invasion, and castration-resistant growth of prostate cancer cells. Mol Cell Biol 2015; 35: 1350-1362.
- Su LL, Ma WX, Yuan JF, Shao Y, Xiao W, Jiang SJ. Expression of Yes-associated protein in non-small cell lung cancer and its relationship with clinical pathological factors. Clin Med J 2012; 125: 4003-4008.
- Pan W, Wang Q, Zhang Y, Zhang NS, Qin JM, Li W. Verteporfin can reverse the paclitaxel resistance induced by YAP over-expression in HCT-8/T cells without photoactivation through inhibiting YAP expression. Cell Physiol Biochem 2016; 39: 481-490.
- Xia Y, Cheng T, Wang Y, Liu YX, Li WH, Li M. YAP promotes ovarian cancer cell tumorigenesis and is indicative of a poor prognosis for ovarian cancer patients. PloS One 2014; 9: 91770.
- Ivery MT, Le T. Modeling the interaction of paclitaxel with beta-tubulin. Oncol Res 2003; 14: 1-19.
- Joo Y, Schumacher B, Landrieu I, Bartel M, Smet-Nocca C, Ahram J. Involvement of 14-3-3 in tubulin instability and impaired axon development is mediated by Tau. FASEB J 2015; 29: 4133-4144.
- Jinawath N, Vasoontara C, Yap KL, Thiaville MM, Nakayama K, Wang TL. NAC-1, a potential stem cell pluripotency factor, contributes to paclitaxel resistance in ovarian cancer through inactivating Gadd45 pathway. Oncogene 2009; 28: 1941-1948.
- Yan Z, Jun Y, Xinli Z, Futang Y, Minggang H, Tao W. Angiomotion promotes the malignant potential of colon cancer cells by activating the YAP-ERK/PI3K-AKT signaling pathway. Oncol Rep 2016; 36: 3619-3626.
- Ye Q, Cai W, Zheng Y, Evers BM, She QB. ERK and AKT signaling cooperate to translationally regulate survivin expression for metastatic progression of colorectal cancer. Oncogene 2014; 33: 1828-1839.
- Zhao D, Sui Y, Zheng X. Mir-331-3p inhibits proliferation and promotes apoptosis by targeting HER2 through the PI3K/Akt and ERK1/2 pathways in colorectal cancer. Oncol Rep 2016; 35: 1075-1082.
- Yano S. Molecular mechanisms of targeted drug resistance in lung cancer and its therapeutic strategy. Nihon Rinsho 2015; 73: 1378-1383.
- Steinhardt AA, Gayyed MF, Klein AP, Dong J, Maitra A, Pan D. Expression of Yes-associated protein in common solid tumors. Hum Pathol 2008; 39: 1582-1589.
- Fujimoto D, Ueda Y, Hirono Y, Goi T, Yamaguchi A. PAR1 participates in the ability of multidrug resistance and tumorigenesis by controlling Hippo-YAP pathway. Oncotarget 2015; 6: 34788-34799.
- Crown J, Oleary M. The taxanes: an update. Lancet 2000; 355: 1176-1178.
- Borst P, Evers R, Kool M, Wijnholds J. A family of drug transporters: the multidrug-resistance-associated proteins. J Natl Cancer Inst 2000; 92: 1295-302.
- Doyle LA, Yang W, Abruzzo LV, Krogmann T, Gao YM, Rishi AK. A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc Natl Acad Sci USA 1998; 95: 15665-70.
- Jian L, Zhen-Nan L, Yong-Jie D, Xiao-Qin L, Qian-Lei B, Ping C. Expression of MRP1, BCRP, LRP, and ERCC1 in advanced non-small-cell lung cancer: correlation with response to chemotherapy and survival. Clin Lung Cancer 2009; 10: 414-421.
- Yvan C, Francis B, Susan PCC, Roger GD, James HG, Gerard B, Francisco A, Marie FP. Multidrug resistance genes (MRP) and MDR1 expression in small cell lung cancer xenografts: relationship with response to chemotherapy. Cancer Lett 1998; 130: 133-141.
- Syed S, Archana J, Kaiser J. Homology modelling and molecular docking of MDR1 with chemotherapeutic agents in non-small cell lung cancer. Biomed Pharmacother 2015; 71: 37-45.
- Jian L, Zhen-Nan L, Li-Chao Y, Qian-Lei B, Jian-Rong W, Shun-Bing S, Xiao-Qin L. Association of expression of MRP1, BCRP, LRP and ERCC1 with outcome of patients with locally advanced non-small cell lung cancer who received neoadjuvant chemotherapy. Lung Cancer 2010; 69: 116-122.
- Kawabata S, Oka M, Soda H, Shiozawa K, Nakatomi K, Tsurutani J. Expression and functional analyses of breast cancer resistance protein in lung cancer. Clin Cancer Res 2003; 9: 3052-3057.
- Park HH, Lo YC, Lin SC, Wang L, Yang JK, Wu H. The death domain superfamily in intracellular signaling of apoptosis and inflammation. Annu Rev Immunol 2007; 25: 561-586.
- Tang XP, Li J, Yu LC, Chen YC, Shi SB, Zhu LR. Clinical significance of survivin and VEGF mRNA detection in the cell fraction of the peripheral blood in non-small cell lung cancer patients before and after surgery. Lung Cancer 2013; 81: 273-279.
- Reed JC, Zha H, Aime-Sempe C, Takayama S, Wang HG. Structure-function analysis of Bcl-2 family proteins: Regulators of programmed cell death. Adv Exp Med Biol 1996; 406: 99-112.
- Kontos CK, Christodoulou MI, Scorilas A. Anticancer agents. Med Chem 2014; 14: 353-374.
- Ping L. Inhibitory effects of hyperoside on lung cancer by inducing apoptosis and suppressing inflammatory response via caspase-3 and NF-kB signaling pathway. Biomed Pharmacother 2016; 82: 216-225.
- Cindy N, Annemilai TR, Louis de M, Jerome C, Sandrine F, Eric R. MEK in cancer and cancer therapy. Pharmacol Ther 2014; 141: 160-171.
- Ramos JW. The regulation of extracellular signal-regulated kinase (ERK) in mammalian cells. Int J Biochem Cell Biol 2008; 40: 2707-2719.
- Taru M, Laura MS, Julie H, Lisa LG, Jonathan LC, Carson CT, Seong AK, David MS, Gordon BM, Joan SB. ERK and p38 MAPK activities determine sensitivity to PI3K/mTOR inhibition via regulation of MYC and YAP. Cancer Res 2016; 76: 15.