Research Article - Biomedical Research (2017) Volume 28, Issue 8
Role of resveratrol and miRNA-126 in vascular endothelial cells of hypertensive rats
Guoqiang Liu1,2, Hengli Li1,2, Fengyong Yang3, Yajing Wang1, Junhao Zhang1, Haichu Yu1 and Qixin Wang1*1Department of Emergency, Affiliated Hospital of Qingdao University, Qingdao, Shandong Province, PR China
2Department of Emergency, the Central Hospital of Tai’an, Tai’an, Shandong Province, PR China
3Intensive Care Unit, Laiwu City People's Hospital, Laiwu, Shandong Province, PR China
- *Corresponding Author:
- Qixin Wang
Department of Emergency
Affiliated Hospital of Qingdao University
PR China
Accepted date: December 27, 2016
Abstract
Aims: This study is to investigate the role of miRNA-126 (miR-126) and resveratrol in vascular endothelial cells of hypertensive rats. .
Methods: Totally 30 male spontaneously hypertensive rats (SHR) were equally divided into 3 groups at random: SHR group, resveratrol treatment group and miR-126 knock-out group. Rat tail artery noninvasive blood pressure meter was applied to measure systolic blood pressure (SBP), mean blood pressure (MBP) and diastolic blood pressure (DBP) of the tail arteries. HE staining was applied to detect the vascular structural changes of mesenteric arteries. Chemical assay was applied to measure nitric oxide content in mesenteric arteries. qRT-PCR was applied to detect the expression of miR-126 in rat serum. Western blot was applied to detect the expression of VEGF in mesenteric arteries.
Results: The expression levels of miR-126 in serum were significantly decreased in miR-126 knock-out group and resveratrol treatment group, compared with SHR group (P<0.05). Compared with those in SHR group, the SBP, MBP and DBP were gradually decreased in miR-126 knock-out group after 13 weeks and in resveratrol treatment group rats after 6 weeks of administration (P<0.05). The vascular hypertrophy of mesenteric arteries, lumen stenosis, the wall-to-lumen area ratios (W/L) and wall thickness to lumen radius ratios (WT/LR) were significantly decreased (P<0.05). The nitric oxide contents in mesenteric arteries and the expression levels of VEGF in mesenteric arteries were significantly higher in resveratrol treatment group and miR-126 knock-out group rats (P<0.05).
Conclusions: MiR-126 knockout and resveratrol treatment can decrease blood pressure in hypertensive rats, and improve mesenteric vascular structures. This may be related to the promotion of NO production and VEGF expression.
Keywords
Resveratrol, miRNA-126, Vascular endothelial cells, Hypertensive rats
Introduction
MicroRNAs (miRs), a class of highly conserved non-coding small RNA, play regulatory role in post-transcriptional gene silencing and are involved in many important biological processes [1]. As an important member of microRNA family, miR-126 plays an important role in the regulation of vascular integrity, endothelial cell proliferation, migration ability, etc [2,3]. miR-126 is highly expressed in hypertensive patients, and expression of miR-126 in vascular endothelial cells of hypertensive rats can down regulate the expression of vascular endothelial growth factor (VEGF), inhibit endothelial proliferation and injury cell repair, promote blood vessel thickening, and reduce nitric oxide (NO) generation [4-6]. Resveratrol is a powerful antioxidant extracted from grapes and other plants, which plays many important roles, such as anti-oxidant and anti-free radicals, anti-inflammation, cardiovascular protective effects, etc [7,8]. In this study, the role of miR-126 and resveratrol in vascular endothelial cells of hypertensive rats was investigated. And, the underlying mechanism was further discussed.
Materials and Methods
Experimental animal
Totally, 30 healthy 8-week-old SPF grade male spontaneously hypertensive rats (SHR) were provided by the animal experiment center of Sichuan Medical University (Animal certification number: SCXK (Chuan) 2013-065), and equally divided into 3 groups at random: SHR group, resveratrol treatment group (40 mg/Kg) and miR-126 knock-out group. All animal experiments were conducted according to the ethical guidelines of Affiliated Hospital of Qingdao University.
Construction of miR-126 knock-out and resveratrol treatment rat model
Lentiviral vector was applied for construction of miR-126 knock-out rat model. Lentiviral vector containing miR-126 was diluted to 350 μL with PBS buffer to the desired titer of 1 × 107 IU/mL, and then injected into rats by tail vein injection to construct the miR-126 knock-out rat model. Tail vein injection was performed for 8 weeks. The rats in the resveratrol treatment group were treated by intragastrical administration with resveratrol at the dose of 40 mg/Kg once a day, and lasted for 20 weeks. The SHR group rats were treated by intragastrical administration with same amount of normal saline.
Sampling
After 20 weeks of treatment, rats were sacrificed. Serum and mesenteric artery were isolated. Serum was used for detection of miR-126 expression, and mesenteric artery was used for detections of W/L, WT/LR, NO and VEGF expression.
Rat blood pressure monitoring
Rat tail artery noninvasive blood pressure meter BP-100A (Chengdu Techman Software Co., Ltd., Chengdu, China) was applied to measure systolic blood pressure (SBP), mean blood pressure (MBP) and diastolic blood pressure (DBP) of the tail arteries in awake rats. At least 3 parallel measurements for each rat were performed, and the average value was used. The blood pressure monitoring was lasted for 8 weeks.
qRT-PCR
Total RNA was extracted from the serum by Trizol according to the manufacturer’s protocol. Then RNA was reverse transcribed into cDNA. The 10 μL reverse transcription reaction system included: 4.625 μL ddH2O, 2 μL 10 × RT buffer, 0.375 μL dNTP Mix, 0.25 μL RNase inhibitor, 0.5 μL reverse transcription primers (1 μmol/L), 0.25 μL reverse transcripatase, and 2 μL total RNA. The reverse transcription reaction was performed with the following procedure: 16°C for 30 min, 42°C for 30 min and 80°C for 1 min. The HsamiR- 126 primer sequence was as follows: 5’-GTC GTA TCC AGT GCG TGT CGT GGA GTC GGC AAT TGC ACT GGA TAC GAC CGC ATT-3’.
The cDNA was used as a template for qRT-PCR to detect the expression of miRNA-126. The expression of U6 was set as internal controls. The 10 μL qPCR reaction system including 2 μL cDNA, 5 μL Maxima SYBR Green/Fluorescein qPCR Master Mix, 0.25 μL sense and 0.25 μL anti-sense primer, and 2.5 μL ddH2O. The primer sequences were as follows:
Hsa-miR-126
Sense: 5’-GTCCGCTCGTACCGTGAGTAATA-3’
Anti-sense: 5’-GTGCGTGTCGTGGAGTC-3
U6
Sense: 5’- CTCGCTTCGGCAGCACA-3’
Anti-sense: 5’-AACGCTTCACGAATTTGCGT-3’
The qRT-PCR reaction was performed on DNA Engine Opticon2 real time PCR system (MJ Research, Inc., Waltham, MA, USA) with the following procedure: 95°C for 5 min, followed by 40 cycles of 95°C for 12 s and 65°C for 50 s. The relative expression was calculated by 2-ΔΔCT method.
HE staining
HE staining was performed with HE staining kit (Nanjing KG Biological Technology Development Co., Ltd., Nanjing, China). Briefly, mesenteric arteries were fixed with 4% poly formaldehyde, dehydrated, embedded in paraffin and cut into sections with the thickness of 4 μm. Then HE staining was performed following the instructions provided by the kit. Morphological changes of mesenteric arteries were observed in all groups. The wall-to-lumen area ratios (W/L) and wall thickness to lumen radius ratios (WT/LR) were analyzed and calculated by Image pro-plus 7.0 software (Media Cybernetics, Inc., MD, Rockville, USA).
Nitrate reductase method for NO detection
The separated clean mesenteric arteries were homogenized on ice by cell lysis solutions and centrifugated at 1500 g for 10 min. The supernatant was collected and was added to a 96-well plate, with 50 μL in each well. The Griess Reagent I (Nanjing KG Biological Technology Development Co., Ltd., Nanjing, China) (50 μL/well) was added to each well and incubated for 60 min. Then, Griess Reagent II (Nanjing KG Biological Technology Development Co., Ltd., Nanjing, China) (50 μL/ well) was added and incubated for 30 min. OD value of each well at 540 nm wavelength was determined.
Western blot
The total protein of each mesenteric artery sample was extracted and the protein concentration was determined by BCA protein assay kit (Abcam Company, Cambridge, England). Proteins were subjected to SDS-PAGE and then transferred to PVDF membrane. The primary antibodies, including anti-VEGF antibody (ab46154, Abcam Company, Cambridge, England) (1:1000) and anti-β-actin antibody (AA128, Beyotime Biological technology co., LTD, Nanjing, China) (1:1000) were added and incubated overnight at 4°C. After washed with TBST buffer for 3 times, secondary goat anti-rabbit antibody (A0192, Beyotime Biological technology co., LTD, Nanjing, China) (1:500) was added and incubated. Membrane was washed for color development. And, image was obtained by Bio-rad geldoc XR+ image lab imaging system (Bio-Rad Laboratories, Inc., Hercules, California,USA) and analyzed by Image pro-plus 7.0 software (Media Cybernetics, Inc., MD Rockville, USA). The relative content of VEGF protein was calculated as the ratio of VEGF gray value to β-actin gray value.
Statistical analysis
All statistical analyses were performed by using the Statistical Package for Social Sciences software (SPSS, Windows version release 18.0; SPSS Inc.; Chicago, IL, USA). Data were presented as mean ± standard deviation. All data were analyzed with normality test. One-way ANOVA was applied for multiple sets of measurement data analysis. SNK method was applied when there was homogeneity of variance, otherwise, Tamhane's T2 method. A P value<0.05 was considered statistically significant.
Results
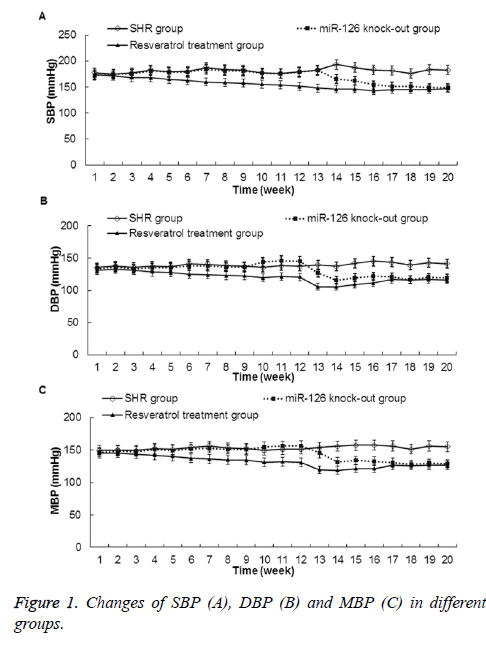
Effects of miR-126 knock-out or resveratrol treatment on rat blood pressure
Effects of miR-126 knock-out or resveratrol treatment on blood pressure of the rat tail arteries were studied. As shown in Figure 1, compared with those in SHR group, the SBP, DBP, and MBP were gradually decreased in miR-126 knock-out group and resveratrol treatment group rats. And, these differences were statistically significant after thirteenth weeks of feeding for the miR-126 knock-out group rats and 6 weeks of administration for the resveratrol treatment group rats (P<0.05). These results indicate that both miR-126 knock-out and 6 weeks of resveratrol treatment can lower rat blood pressure.
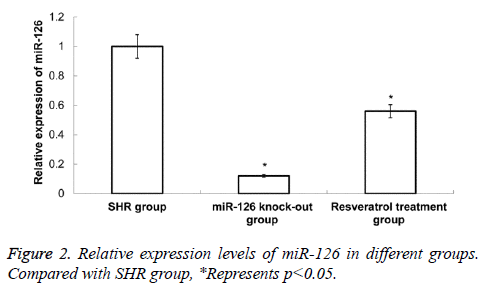
Expression changes of miR-126 in different rat serums
The qRT-PCR was applied to detect the expression of miR-126 in rat serum. Compared with SHR group, the expression level of miR-126 in miR-126 knock-out group and resveratrol treatment group was significantly decreased (P<0.05) (Figure 2). These results indicate that the decrease of rat blood pressure by resveratrol treatment may be related to the significantly decreased of miR-126 expression.
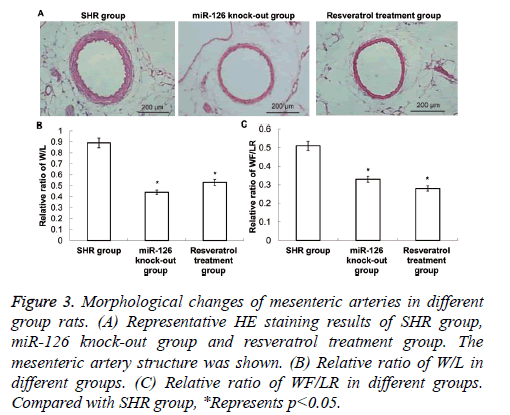
Morphological changes of rat mesenteric arteries
HE staining was applied to observe the morphological changes of mesenteric arteries. As shown in Figure 3, in the SHR group, there was vascular hypertrophy and lumen stenosis, and W/L and WT/LR were relatively high (P<0.05).
Figure 3: Morphological changes of mesenteric arteries in different group rats. (A) Representative HE staining results of SHR group, miR-126 knock-out group and resveratrol treatment group. The mesenteric artery structure was shown. (B) Relative ratio of W/L in different groups. (C) Relative ratio of WF/LR in different groups. Compared with SHR group, *Represents p<0.05.
Compared with those in SHR group rats, the vascular hypertrophy of mesenteric arteries, lumen stenosis, W/L and WT/LR were significantly decreased in resveratrol treatment group and miR-126 knock-out group (P<0.05). These results indicate that both resveratrol treatment and miR-126 knock-out can improve the vascular hypertrophy and lumen stenosis of the mesenteric artery in hypertensive rats.
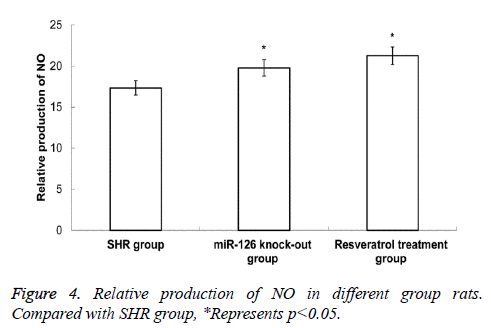
Production of NO in rat mesenteric arteries
Chemical assay was applied to determine the production of NO in rat mesenteric arteries. As shown in Figure 4, the NO contents in mesenteric arteries in resveratrol treatment group and miR-126 knock-out group were higher than in SHR group, and the difference was statistically significant (P<0.05). This data suggest that the decrease of rat blood pressure by resveratrol treatment or miR-126 knock-out may be related to the increased expression of NO.
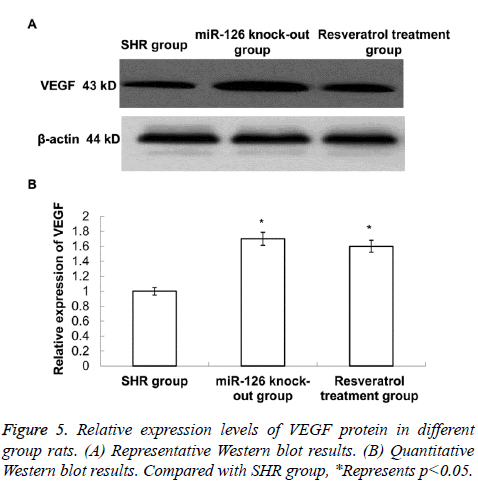
Expressions of VEGF in rat mesenteric arteries
The Western blot was applied to detect the expression of VEGF in different rat mesenteric arteries. As shown in Figure 5, the expression levels of VEGF in mesenteric arteries in resveratrol treatment group and miR-126 knock-out group were higher than in SHR group, and the difference was statistically significant (P<0.05). This data suggest that both resveratrol treatment and miR-126 knock-out can promote the expression of VEGF.
Discussion
miR-126 plays an important role in the regulation of vascular integrity, endothelial cell proliferation and migration capacity [9,10]. miR-126 is highly expressed in hypertensive patients, which may affect the expression of VEGF and inhibit endothelial proliferation and injury cell repair, promote blood vessel thickening, and reduce NO generation [11]. In this study, the lentiviral vector was applied as a vector for miR-126 knocking out, and the changes of rat blood pressure and vascular structure were also observed. Results showed that rat blood pressure was decreased after miR-126 knock out, suggesting that miR-126 plays a role in the regulation of blood pressure. Meanwhile, the vascular hypertrophy of mesenteric arteries, lumen stenosis, W/L and WT/LR were significantly decreased in miR-126 knock-out group, suggesting that reducing the expression of miR-126 is helpful to improve the blood vessel structure.
Resveratrol intervention was also applied in this study, and the relationship between resveratrol and miR-126 was analyzed. Results showed blood pressure, the vascular hypertrophy of mesenteric arteries, lumen stenosis, and the expression levels of miR-126 were also significantly decreased in resveratrol treatment group rats, suggesting that the role of resveratrol in reducing blood pressure may be related to the regulation of miR-126 expression. The expression levels of NO and VEGF were further studied. It is reported that in hypertensive patients, the endothelial function was injured and the inflammatory response was activated [12]. NO is an important humoral factor in regulating the function of endothelial cells. In hypertensive patients, the activity of endothelial nitric oxide synthase (eNOS) decreased [13]. Excessive proliferation of vascular smooth muscle cells resulted in vascular hypertrophy and lumen stenosis, which further promoted the increase of blood pressure and formed a vicious circle of hypertension [14,15]. There was a positive feedback relationship between VEGF and NO [16,17]. Activated VEGF can promote production of NO to reverse the disorders of renin-angiotensin-aldosterone system, thus promoting cell injury repair, inhibiting blood vessel thickening and improving vascular remodeling [18-20].
In the present study, results showed that resveratrol treatment or miR-126 knock-out resulted in up-regulated expression of VEGF and production of NO in mesenteric arteries, which may be related to the improvement of vascular structure in hypertensive rats. In short, blood pressure was decreased and vascular structure was improved after miR-126 knock out in rats, suggesting miR-126 is closely related to hypertension. And, NO production and VEGF expression were both increased after miR-126 knock out. Resveratrol treatment showed similar effect as miR-126 knock out. Therefore, resveratrol may improve vascular structure and decrease blood pressure by interfering the expression of miR-126, VEGF and NO.
Acknowledgement
The authors want to thank Dr Zhen Liu from Affiliated Hospital of Qingdao University for his valuable help for this manuscript.
References
- Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 2005; 120: 15-20.
- Nicoli S, Standley C, Walker P, Hurlstone A, Fogarty KE, Lawson ND. MicroRNA-mediated integration of haemodynamics and Vegf signalling during angiogenesis. Nature 2010; 464: 1196-1200.
- Wang S, Aurora AB, Johnson BA, Qi X, McAnally J, Hill JA, Richardson JA, Bassel-Duby R, Olson EN. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev Cell 2008; 15: 261-271.
- Harris TA, Yamakuchi M, Ferlito M, Mendell JT, Lowenstein CJ. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule 1. Proc Natl Acad Sci USA 2008; 105: 1516-1521.
- Fang ZF, Xie LD. Effect of rosuvastatin on blood pressure, mesenteric arteries structure and vasodilatation function in spontaneously hypertensive rats. Chin J Hypertens 2015; 23: 433-435.
- Jin HF, Tian Y, Yan H, Tang CS, Du JB. Effect of hydrogen sulfide on the expression of PCNA and p-ERK in the aortic smooth muscle cells of normal rats and spontaneous hypertensive rats. Chin Pharmacol Bull 2008; 24: 160-165.
- Lee CS, Choi EY, Lee SC, Koh HJ, Lee JH, Chung JH. Resveratrol Inhibits Hypoxia-Induced Vascular Endothelial Growth Factor Expression and Pathological Neovascularization. Yonsei Med J 2015; 56: 1678-1685.
- Liu Z, Li Y, Yang R. Effects of resveratrol on vascular endothelial growth factor expression in osteosarcoma cells and cell proliferation. Oncol Lett 2012; 4: 837-839.
- Harris TA, Yamakuchi M, Ferlito M, Mendell JT, Lowenstein CJ. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule. Proc Natl Acad Sci USA 2008; 105: 1516-1521.
- Wei XJ, Han M, Yang FY, Wei GC, Liang ZG, Yao H, Ji CW, Xie RS, Gong CL, Tian Y. Biological significance of miR-126 expression in atrial fibrillation and heart failure. Braz J Med Biol Res 2015; 48: 983-989.
- Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 1987; 327: 524-526.
- Paul M, Mehr AP, Kreutz R. Physiology of local renin-angiotensin systems. Physiol Rev 2006; 86: 747-803.
- Qin X, Hou X, Liang T, Chen L, Lu T, Li Q. Farrerol can attenuate the aortic lesion in spontaneously hypertensive rats via the upregulation ofeNOS and reduction of NAD(P)H oxidase activity. Eur J Pharmacol 2015; 769: 211-218.
- Hong F, Li Y, Xu Y. Decreased placental miR-126 expression and vascular endothelial growth factor levels in patients with pre-eclampsia. J Int Med Res 2014; 42: 1243-1251.
- Shesely EG, Maeda N, Kim HS, Desai KM, Krege JH, Laubach VE, Sherman PA, Sessa WC, Smithies O. Elevated blood pressures in mice lacking endothelial nitric oxide synthase. Proc Natl Acad Sci U S A 1996; 93: 13176-13181.
- Bhavina K, Radhika J, Pandian SS. VEGF and eNOS expression in umbilical cord from pregnancy complicated by hypertensivedisorder with different severity. Biomed Res Int 2014; 2014: 982159.
- Luizon MR, Sandrim VC, Palei AC, Lacchini R, Cavalli RC, Duarte G, Tanus-Santos JE. Epistasis among eNOS, MMP-9 and VEGF maternal genotypes in hypertensive disorders of pregnancy. Hypertens Res 2012; 35: 917-921.
- Gélinas DS, Bernatchez PN, Rollin S, Bazan NG, Sirois MG. Immediate and delayed VEGF-mediated NO synthesis in endothelial cells: role of PI3K, PKC and PLC pathways. Br J Pharmacol 2002; 137: 1021-1030.
- Jesmin S, Maeda S, Mowa CN, Zaedi S, Togashi H, Prodhan SH, Yamaguchi T, Yoshioka M, Sakuma I, Miyauchi T, Kato N. Antagonism of endothelin action normalizes altered levels of VEGF and its signaling in the brain of stroke-prone spontaneously hypertensive rat. Eur J Pharmacol 2007; 574: 158-171.
- Liu Y, Wang G, Yang M, Chen H, Zhao Y, Yang S, Sun C. Arctigenin reduces blood pressure by modulation of nitric oxide synthase and NADPH oxidase expression in spontaneously hypertensive rats. Biochem Biophys Res Commun 2015; 468: 837-842.