- Biomedical Research (2012) Volume 23, Issue 2
Protein oxidation: A potential cause of hypoalbuminemia in oral cancer
Vinod Chandran*, Anitha M, Avinash SS, Gayathri M Rao, Beena V Shetty, Sudha K
Kasturba Medical College, Manipal University, Mangalore, India
- *Corresponding Author:
- Vinod Chandran
Department of Biochemistry
Kasturba Medical College
Manipal University
Mangalore, India
Accepted date: February 22 2011
Abstract
This study examined the role of the free radical induced protein oxidation and decreased antioxidant defense mechanism as a causative agent of hypoalbuminemia in patients with oral cancer. Plasma advanced oxidation protein products (AOPP) and % hemolysis were estimated as protein oxidation indicators. Antioxidant status was evaluated by determining GSH , total thiols, albumin and total antioxidant activity in plasma. The parameters were estimated by spectrophotometric methods. Data was compared with the samples collected from age and sex matched controls. It was observed that there was a significant increase in protein oxidation products and a non significant decrease in plasma protein thiols in oral cancer patients. However, the decrease in plasma total thiols was significant compared to normal subjects. The decrease in blood GSH, plasma GSH, plasma total protein and albumin were significant in these patients compared to controls. Total antioxidant activity of oral cancer patients decreased significantly compared to controls. Oxidative stress, weak antioxidant defense or both can be considered to be important players in causing hypoalbuminemia in multimechanistic pathogenesis of oral cancer
Key words
Oral cancer , Glutathione, thiols, antioxidants.
Introduction
Cancer, a disease characterized by uncontrolled growth and spread of abnormal cells is one of the major causes of death in humans. Recent studies indicate that increased production of reactive oxygen species (ROS) can promote development of malignancy .Over production of ROS within the tissue can damage DNA, proteins, lipids and carbohydrates [1,2], producing oxidized products like advanced oxidation protein products (AOPP), malondialdehyde (MDA),advanced glycation end products etc [3]. It is widely recognized that oxidation of proteins play an essential role in pathogenesis of cancer [4] Indeed knockout of various antioxidant defense enzymes promotes cancer development in animals. Oxidative events have remarkable importance in development of oral cancer. Since hypoalbuminemia is one of the most common features of cancer, this study aims at relating it with oxidative stress [1]. Hence in this study total protein, albumin, thiols , glutathione (GSH) and total antioxidant activity were determined in oral cancer patients to evaluate antioxidant status. To determine protein oxidation AOPP and % hemolysis were estimated. Data was compared with the samples of age and sex matched controls.
Materials and Methods
This study was approved by institutional ethical review committee. Informed consent was obtained from each subject before sample collection. The study population consisted of 25 normal subjects and 25oral cancer patients aged between 30-60 years of both the sex. Patients included for the study were all freshly diagnosed to have oral cancer on clinical and histopathological basis and who had not undergone any surgery. The patients who received radiotherapy or chemotherapy were excluded from the study. Those patients who were diabetic, hypertensive or those with liver and renal diseases which could influence the parameters under study were also excluded.
Four ml venous blood was collected in heparinised vaccutainers under aseptic precautions after taking informed consent. All the parameters were estimated on the same day. 0.2 ml of whole blood was taken for reduced glutathione estimation by Ernst method [5] where intensity of yellow chromogen with DTNB was measured. The remaining volume of blood was centrifuged at 3000 rpm and the plasma was separated into separate sterile vials. AOPP was estimated by Witkos method [6], where an aliquot of suitably diluted plasma was made to react with KI and acetic acid. Total thiols of plasma were measured by the reaction of sulfhydryl groups with dithionitro benzoic acid by Ellmans method [7] .Total protein and albumin was estimated by Biuret method [8], plasma GSH by Ernst method and total antioxidant activity by Koracevic method [9] based on measurement of thiobarbituric acid reactive substances.
Statistical analysis of the data was done using Mann- Whitney U test. Software statistical package for social sciences was used.
The separated RBC cells were washed thrice with 0.9% cold normal saline. After which they were suspended in an equal volume of the same saline solution. This 50% RBC cell suspension was used for estimation of % hemolysis by method of Kartha and Krishna Murthy [10].
Results
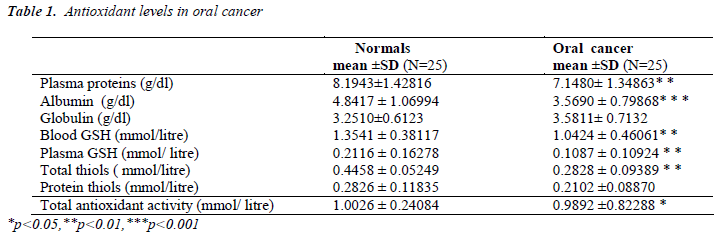
In the current study, blood glutathione, plasma glutathione, plasma total thiols, albumin and total antioxidant capacity were estimated in oral cancer patients and age and sex matched controls.
As a marker of oxidant damage % hemolysis and plasma AOPP were determined in oral cancer patients and compared with normals.
When compared to the controls, blood GSH and plasma GSH decreased significantly in oral cancer patients (p<0.01) (Table.1). Plasma proteins and albumin also decreased significantly in oral cancer patients compared to normal subjects. (Table.1). Plasma total thiols in oral cancer patients showed a significant decrease (p<0.001) (Table. 1). When compared to healthy individuals, the decrease in total antioxidant capacity of oral cancer patients was also statistically significant (p<0.01) (Table.1).
Significant increase in AOPP levels (p<0.001) was observed in oral cancer patients compared to controls (Table. 2). Increase in % hemolysis was also statistically significant (p<0.05) in these patients compared to healthy individuals.(Table.2).
Discussion
Free radical production is ubiquitous in all respiring organisms and is enhanced in many disease states and in carcinogen exposure[11]. Widely recognized fact is that oxidation of proteins play an essential role in pathogenesis of cancer [4]. Hypoproteinemia in cancer may be an expression of cachexia, representing homeostatic derangement in which the utilization and destruction of albumin by the tumor cannot be compensated by liver. A study indicated that on glycoprotein electrophoresis the albumin region glycoproteins were significantly low and gamma region glycoproteins were significantly elevated in healthy individuals with habit of tobacco consumption, in patients with oral precancerous conditions and untreated cancer patients compared to healthy individuals without the habit of tobacco consumption [12]. Significantly lower plasma protein and albumin levels in patients with oral cancer compared to normal subjects in the present study, is in agreement with earlier studies on cancer of GIT [13, 14] and bladder cancer [15]. In one of the earlier studies, it was shown that plasma albumin was lower in pre cancer condition of oral cancer [12]. Serum protein levels were significantly lower in patients with oral sub mucous fibrosis [16].It may be proposed that proteins and albumin in plasma of patients with oral cancer have been subjected to oxidative damage resulting in their decreased concentration. Advanced oxidation protein products (AOPP) are predominantly albumin and its aggregates damaged by oxidative stress [3]. These AOPP molecules can induce production of cytokines which may be responsible for advancement of cancer. AOPP, the terminal products of protein oxidation are reliable markers of oxidative stress [3]. Increased ROS production might have further aggravated the AOPP levels in plasma of oral cancer patients in the present study.
In cancer, a high flux of oxidants not only depletes the cellular thiols and GSH but also damages the entire cell. Rats with 4-Nitro quinoline- 1-oxide induced oral cancer, showed reduced levels of blood GSH and protein thiols [17].Results of our study are in agreement with this animal study. Plasma protein thiols and GSH were significantly decreased in bladder cancer [15], colon cancer [18] and hepato cellular cancer patients [19]. Damage to proteins of human plasma by carcinogens causes oxidative damage to them, which can be measured by loss of –SH groups. It can be stated that due to oxidant surge, plasma thiols in oral cancer patients may be excessively oxidized to lower free radical toxicity and are depleted resulting in their low levels in plasma. Studies in plasma and cells exposed to hydroxyl and peroxyl radicals indicated that protein thiol groups can inhibit albumin oxidation in vitro [20]. This clearly indicates that low levels of thiols & GSH in oral cancer patients might have led to albumin oxidation resulting in low plasma albumin levels. GSH detoxifies cigarette smoke carcinogens, a key etiological factor of oral cancer. Thiols such as glutathione interferes with the complex carcinogenic process by conjugating with electrophilic carcinogens there by preventing tumour initiation and defending against oxidative stress [21].
In the present study total antioxidant capacity of oral cancer patients were significantly decreased compared to normals. Decreased total antioxidant activity was seen in both tumor tissue and serum of colorectal cancer and liver metastasis cases [22]. This was justified by earlier studies which showed decreased antioxidant activity in gastric cancer patients [23].
Oral cancer patients have shown significant increase in percentage hemolysis compared to healthy controls in the present study. Blood GSH level is an indicator of potential oxidative damage to erythrocytes [3]. Increased percentage hemolysis in oral cancer patients may be due to decrease in glutathione level or due to increased oxidant surge. Erythrocyte lipid peroxidation was found to be increased in gastric cancer patients [23] which might have led to increase in hemolysis.
The present study highlights the occurrence of protein oxidation and possible break down of antioxidant status in patients with oral cancer. We conclude that protein oxidation may have lead to hypoalbuminemia in oral cancer.
While awaiting results on further human studies research evidence suggests that increased antioxidants in blood can provide protection against damage in cancer patients.
References
- Haliwell B. Oxidative stress and cancer, have we moved forward? Biochem Journal 2007; 401: 1-10.
- Manonaran S, Kolanjiappan K, Suresh ?, Panchmurthy K .Lipid peroxidation and antioxidant status in patients with oral squamous cell carcinoma. Indian journal of medical research 2005; 122: 529-534.
- Vijayamalini M, Manoharan S. Lipid peroxidation, vitamin C, E and reduced glutathione levels in patients with pulmonary tuberculosis. Wiley Inter Science Journal 2003; 22 (1): 19-22.
- Funda K, Bahad C, Melin AK, Sabanattin A, Zeki AR, Aylin S et al. Advanced oxidation protein products, Ferrous oxidation in xylenol orange and malondialdehyde levels in thyroid cancer. Annals of surgical oncology 2007; 14: 2616-2620.
- Sathish T, Shetty BV, Rao GM, Sudha K. Effect of grandmal seizures on oxidative stress and antioxidants in epileptics. Biomedical Research 2007; 18 (3): 167- 169.
- Avinash SS, Anitha M, Chandran V, Rao GM, Sudha K, Shetty BV. Advanced oxidation protein products and total antioxidant activity in colorectal carcinoma. Indian Jr. Physiol Pharmacol 2009; 53 (4): 370-374.
- Malgorzata I, Grazyna C, Elzbieta LK, Edward B, Lidia W. Plasma levels of total, free and protein bound thiols as well as sulfane sulfur in different age groups of rats. Acta Biochemica Polonica 2004; 51 (3): 815- 824.
- Abraham Z, Reznick ?, Carroll EC, Miao LH, Yuichiro JS et al. Modified plasma proteins by cigarette smoke as measured by protein carbonyl formation. Biochem J 1992; 286: 607-611.
- Koracevic D, Koracevic G, Djordevic V, Androjevic S, Cosic V. Method of measurement of antioxidant activity in human fluids. Journal of clinical Pathology 2001; 54: 356-361.
- Sudha K, Rao AV, Rao A. Free radical toxicity and antioxidants in Parkinsons disease. Neurology India 2003; 51(1): 60-62.
- Lu W, Ogasawara MA, Huang P. Models of reactive oxygen species in cancer. Drug Discov Today Dis Models 2007; 4: 67-73.
- Patel PS, Rawal RM, Rawal GN, Pater BP, Patel YV, Shah SG et al Glycoprotein electrophoretic pattern in sera of patients with oral pre cancer conditions and upper aero digestive tract cancer. Journal of Clin cancer research1997; 161: 65-70.
- Daac LN, England A, Freng A. Serum protein in patients with recently discovered cancer in the oral cavity/ throat. What experiences were gained by measurements of concentration and electrophoresis? Tidsslor Nor Laege Foren 1997; 117(25): 3671-3673.
- Liuja ?, Tsai WI, Wong YK , Poon CK, Chao SY, Hsiao YC, Chan MY, Cheng CS, Wang CC, Wang CP. Nutritional factors and survival of patients with oral
- Iker A, Imaz Y, Tulay A, Ufuk C, Suleyman A, Veliyale ?. Relation between bladder cancer and protein oxidation. Urology and nephrology 2003; 35(3): 345- 350.
- Rajendra R, Vasudevan DM, Vijayakumar T. Serum levels of iron and proteins in oral submucous fibrosis. Bull World Health Organ 1994; 72 (6): 985-996.
- Srinivasan P, Sabitha KS, Shyamadevi CS. Therapeutic efficacy of green tea poly phenols on cellular 4- Nitroquinoline 1- oxide induced oral carcinogenesis. Chem biol interact 2004; 149 (2-3): 81-87.
- Claudia DG, Rasaria A, Raffaele L, Francesca L, Antonio L, Angello V. Non Protein Antioxidant status in plasma of subjects with colon cancer. Experimental biology and medicine 2003; 228: 525-528.
- Lee kT, Tsai SM, Wang SN, Lin SK, Wu SH, Chuang SC, Wu SH, Tsai LY. Glutathione status in the blood and tissues of patients with virus originated hepatocellularl carcinoma. Clin Biochem. 2007; 40 (15): 1157- 1162.
- Thomas JA, Mallis RJ. Aging and oxidation of reactive protein sulfhydryls. Exp Geront 2000; 36: 1519-1526.
- Huber WW, Parzefall W. Thiols in chemoprevention of cancer. Curr opin pharamacol 2007; 7: 404-409.
- Czeczot H, Skrzycki M, Dodsiad M, Gawry SE, Nycowski P, Porembska Z. Antioxidant status of patients with primary colorectal cancer and liver metastasis of colorectal cancer. Pol merkur Lekarski 2005; 18 (103): 58-61.
- Arivazhagan S, Kavitha K, Nagini S. Erythrocyte lipid peroxidation and antioxidants in gastric cancer patients. Cell Biochemistry and function 1998; 15 (1): 15-18.