Case Report - Otolaryngology Online Journal (2022) Volume 12, Issue 9
PRIMARY COMBINED SMALL CELL CARCINOMA OF THE HYPOPHARYNX
Ana Isabel Gonçalves*, André Carção, Delfim Duarte, Ditza Vilhena
Department of Otorhinolaryngology, Pedro Hispano Hospital, Matosinhos, Oporto, Portugal
- Corresponding Author:
- Gonçalves IA
Department of Otorhinolaryngology
Pedro Hispano Hospital, Matosinhos
Oporto, Portugal
Tel. No.: 351932792516
E-mail: gc.anaisabel@gmail.com
Received: 03-Sep-2022, Manuscript No. JORL-22-74274; Editor assigned: 05-Sep-2022, PreQC No. JORL-22-74274 (PQ); Reviewed: 19-Sep-2022, QC No. JORL-22-74274; Published: 27-Sep-2022, DOI: 10.35841/2250-0359.12.9.292
Abstract
A male in his sixties with a 40-pack-year smoking history presented with a 1 month history of left-sided neck swelling. Upon neck palpation, the existence of a left, hardened, painful and fixed adenopathy was evident occupying lymph node levels II and III. Laryngoscopy identified a large ulcerovegetative lesion centered on the left piriform sinus, extending to the left pharyngoepiglottic and aryepiglottic folds, with normal vocal cord movement. An ultrasoundguided microbiopsy of the adenopathy revealed a metastatic squamous cell carcinoma (SqCC). A biopsy of the hypopharynx lesion was also done, revealing a combined carcinoma, comprising both SqCC and small cell carcinoma (SmCC) components. Computed tomography (CT) showed a heterogeneously enhanced tumor extending through the left piriform sinus. The neck adenopathy sized 65 × 52 mm. There was no evidence of distant metastasis. The final diagnosis was a primary combined SmCC of the hypopharynx cT3N3aM0. The patient was proposed for chemoradiotherapy.
Keywords
Combined small cell carcinoma, Hypopharynx cancer, Head and neck cancer, Chemoradiotherapy
Introduction
Hypopharyngeal carcinomas represent <10% of all carcinomas of the aerodigestive tract. These carcinomas generally have low survival rates (overall survival at 5 years rarely exceeds 35% regardless of the therapeutic approach) associated with a late diagnosis when the tumor is already locally advanced and with cervical lymph node metastasis frequently present. The average age of diagnosis of hypopharyngeal carcinomas is around 60 years and there is a well-known significant association between the development of these lesions and the consumption of tobacco and alcohol, with a synergistic carcinogenic effect of the two substances. Of all hypopharyngeal tumors, the histological type most frequently involved is squamous cell carcinoma, corresponding to 95% of cases, followed by adenocarcinoma.
Depending on the location of the tumor, the initial symptoms may also vary, although in hypopharyngeal carcinomas the initial symptoms are vaguer (unlike, for example, laryngeal tumors, in which dysphonia appears earlier), which ends up delaying the diagnosis. Piriform Hypopharyngeal carcinomas represent <10% of all carcinomas of the aerodigestive tract. These carcinomas generally have low survival rates (overall survival at 5 years rarely exceeds 35% regardless of the therapeutic approach) associated with a late diagnosis when the tumor is already locally advanced and with cervical lymph node metastasis frequently present. The average age of diagnosis of hypopharyngeal carcinomas is around 60 years and there is a well-known significant association between the development of these lesions and the consumption of tobacco and alcohol, with a synergistic carcinogenic effect of the two substances. Of all hypopharyngeal tumors, the histological type most frequently involved is squamous cell carcinoma, corresponding to 95% of cases, followed by adenocarcinoma [1, 2].
Depending on the location of the tumor, the initial symptoms may also vary, although in hypopharyngeal carcinomas the initial symptoms are vaguer (unlike, for example, laryngeal tumors, in which dysphonia appears earlier), which ends up delaying the diagnosis. Piriform sinus carcinomas are most often manifested by unilateral odynophagia and dysphagia, usually late and indicative of intraluminal growth of the neoformation. As these symptoms worsen, they also begin to be associated with weight loss due to reduced oral food intake. Otalgia may arise associated with invasion through the lateral wall and dysphonia may occur due to involvement of the laryngeal musculature. Profuse lymphatic drainage from the piriform sinus results in early cervical lymph node metastasis and generally up to 75% of patients have lymph node involvement at diagnosis, with 10% bilateral [1].
The concept of combined carcinoma means that there are two carcinomas that are a constituent part of the same neoformation, but do not invade each other, and there is a clear histological separation [1-6]. It is this feature that differentiates combined carcinoma from mixed carcinoma, in which there is a combination of histopathological features of two different types of carcinoma, sometimes with no obvious histological transition/separation in the same tumor mass [1,7].
Combined carcinoma is rare in the head and neck region but, when it happens, it is more frequent reported in the thyroid gland [7,8]. It is believed that the treatment of these tumors should be based on the treatment of the histological part of the most aggressive tumor, although there are no established therapeutic guidelines to date [7,9].
Small cell carcinoma (SmCC) of the hypopharynx is a rare entity and SmCC with a squamous component is designed to as combined SmCC. This combined carcinoma is even more rare in the hypopharynx, with only 6 cases previously reported in the literature, to the best of our knowledge [2, 4, 5, 10,11]. In the larynx, it represents <10% of all SmCC, being less rare than in the hypopharynx [3,11,12].
In this paper we aim to present a case report and review of the literature concerning primary combined SmCC of the hypopharynx.
Besides being extremely rare, combined SmCC is often difficult to diagnose because most of the SmCC cells are found in the tumor at a greater depth [5]. It has a very poor prognosis due to early metastasis, without an established treatment regimen. As such, the treatment of combined SmCC ends ups following the same treatment as SmCC alone [7,11]. Combined chemoradiotherapy aiming organ preservation is the option preferred, regarding the high propensity to metastatic dissemination, even in patients with clinically localized tumor [4, 7,11].
Case Presentation:
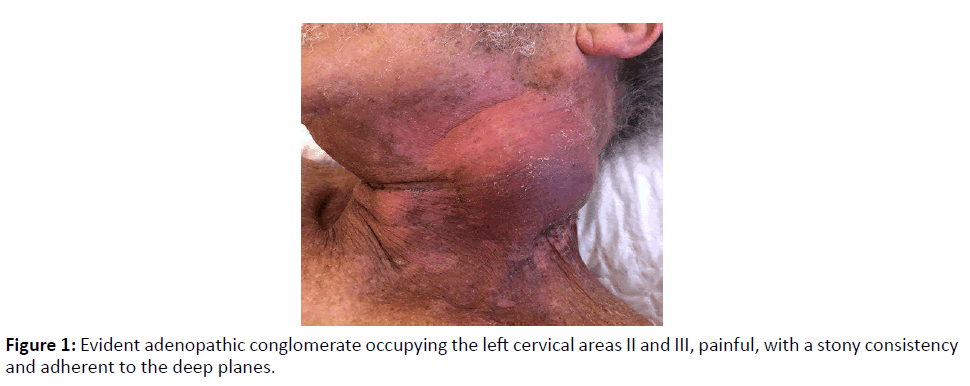
A male patient in his sixties was referred for observation at the Emergency Department, for presenting a left cervical swelling with about 1 month of evolution, as he mentioned, and without other associated signs/symptoms. When questioned, the patient denied dysphonia, dysphagia, or dyspnea. Regarding personal medical history, the patient had atrial fibrillation, type 2 diabetes, dyslipidemia and arterial hypertension, medicated, respectively, with edoxaban 60 mg id, metformin 1000 mg id, simvastatin 20 mg id and ramipril 5 mg id. The patient also reported being a former smoker for 6 years, with a previous smoking history of 40 UMA (he smoked from 18 to 58 years old 1 pack of cigarettes/ day). Possessed alcoholic habits, consuming about 2 glasses of red wine a day. On physical examination, the patient looked emaciated; although he reported that he had maintained his usual weight for several years and denied recent weight loss. Flexible nasopharyngolaryngoscopy showed a vegetating lesion centered on the left piriform sinus and extending to the left pharyngoepiglottic and arytenoepiglottic folds, with maintained laryngeal mobility and a permeable glottic lumen. Neck palpation revealed the existence of an adenopathic conglomerate occupying the left areas II and III, painful, with a stony consistency and adherent to the deep planes (Figure 1).
Investigations:
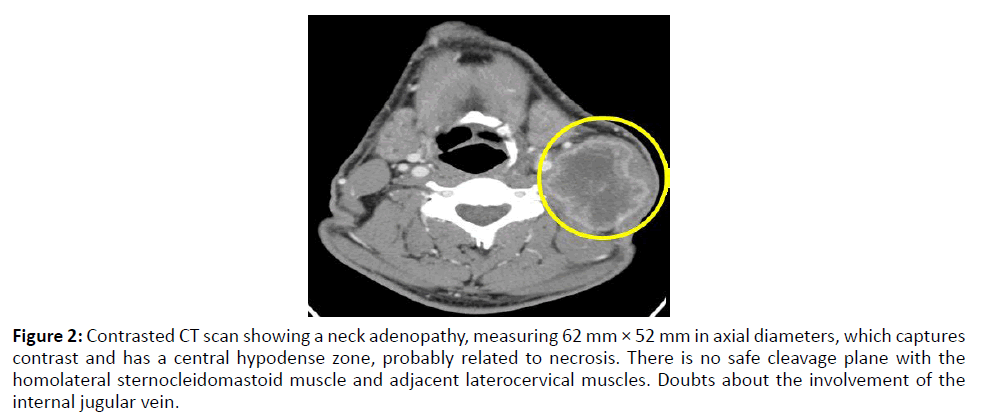
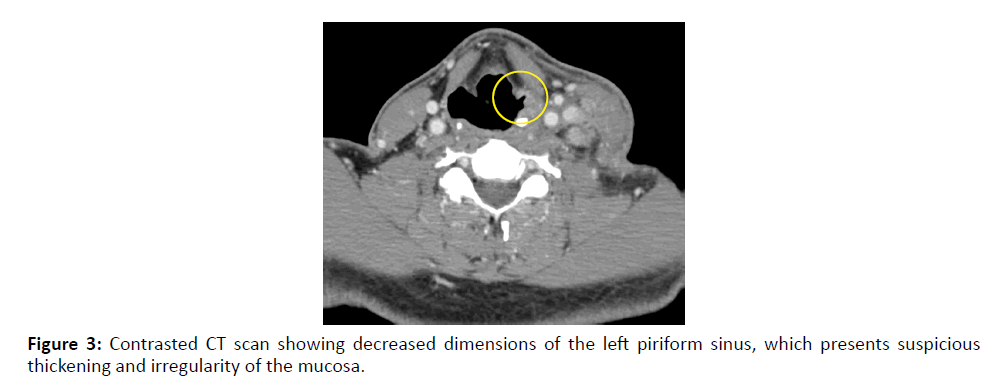
The patient underwent a cervicothoracic CT scan, which showed a decrease in the dimensions of the left piriform sinus, with thickening and irregularity of its entire mucosa, with a suspicious aspect of malignancy (Figure 2). The imaging exam also showed a left cervical adenopathy occupying left cervical areas II and III and measuring 62 × 52 mm in axial diameter. This adenopathy captured contrast and presented a central hypodense zone, probably related to necrosis. There was no safe cleavage plane with the homolateral sternocleidomastoid muscle and adjacent laterocervical muscles. Also, there were doubts about the involvement of the internal jugular vein by the adenopathy (Figure 3). On CT, no distant metastases were evident.
Figure 2: Contrasted CT scan showing a neck adenopathy, measuring 62 mm × 52 mm in axial diameters, which captures contrast and has a central hypodense zone, probably related to necrosis. There is no safe cleavage plane with the homolateral sternocleidomastoid muscle and adjacent laterocervical muscles. Doubts about the involvement of the internal jugular vein.
Figure 3: Contrasted CT scan showing decreased dimensions of the left piriform sinus, which presents suspicious thickening and irregularity of the mucosa.
Given the presence of these two suspicion neoformations, the piriform sinus lesion and the neck adenopathy, it was important to clarify their anatomopathological nature so that a timely histological diagnosis could be established, followed by the staging of the disease and the planning of the therapeutic approach.
Thus, a microbiopsy, guided by ultrasound, of the cervical adenopathy was initially performed, which revealed that it was a metastasis of squamous cell carcinoma.
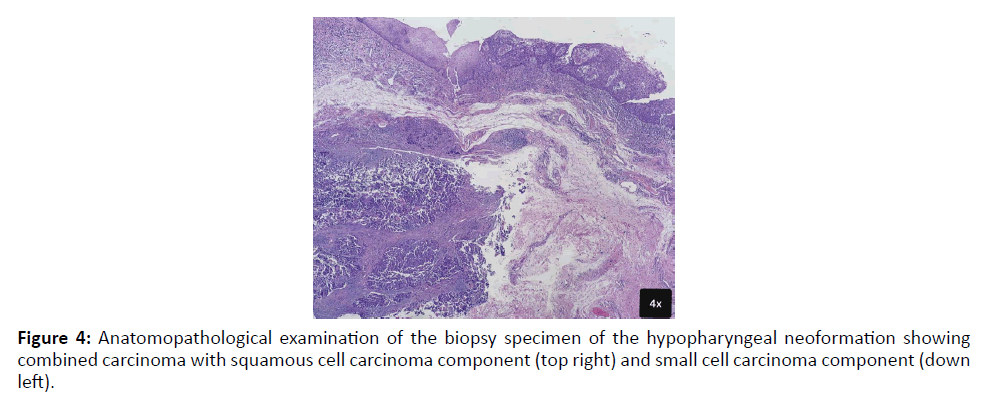
Subsequently, a suspension laryngoscopy was performed, in the operating room and under general anesthesia with orotracheal intubation, to reassess the left pyriform sinus neoformation extent more accurately and for biopsies of this lesion. It was possible to infer that the lesion occupied the anterior and lateral walls of the piriform sinus, with its medial wall and posterior pharyngeal wall being free of lesion. As clinically evident on initial physical examination, the lesion extended to the homolateral pharyngoepiglottic and arytenoepiglottic folds. The laryngeal aditus was free, with vocal cord, ventricular band and homolateral ventricle without alterations. The anatomopathological result of the biopsies was indeed surprising, revealing the presence of a combined carcinoma of the hypopharynx with components of squamous cell carcinoma and small cell neuroendocrine carcinoma (Figure 4). The cell proliferation index revealed positivity of 90% of neoplastic cells for Ki67, indicator of a high degree of tumor proliferation. Taking into account the fact that we are facing a locally advanced neoplastic disease with two different histological components, positron emission tomography (PET) was performed for a more thorough assessment of the possibility of second primary tumors in other locations or distant metastasis foci. PET showed no disease in locations other than those already known.
Figure 4:Anatomopathological examination of the biopsy specimen of the hypopharyngeal neoformation showing combined carcinoma with squamous cell carcinoma component (top right) and small cell carcinoma component (down left).
Differential Diagnosis:
By this time, we had established our most probable diagnosis taking into account the histological result of biopsies and CT scan: primary combined small cell carcinoma of the hypopharynx clinically staged as a cT3N3aM0 (according to the classification by the American Joint Committee on Cancer 8th edition, 2017), corresponding to a IVb disease stages [13,14]. The primary tumor was classified as T3 considering its size, clinically perceived through the laryngoscopy performed in the operating room as being greater than 4 centimeters.
In these cases, faced with the appearance of a rapidly growing, painful, hard neck adenopathy that adheres to the deep cervical planes and, taking into account the fact that the patient was a smoker, we should be alerted to the possibility of neoplastic disease. Thus, it is important to establish the diagnosis in time and, in the presence of a neck swelling, to think about the possibility of metastatic disease of a primary tumor of the head and neck region, which is necessary to diagnose.
In this particular case, we have a histological diagnosis of combined hypopharyngeal carcinoma. The diagnosis of these tumors is difficult because the two histological components that constitute the tumor must be present in the biopsied region. In addition, only regional metastasis of the squamous cell carcinoma component was found. Therefore, in addition to the biopsy, it is important to confirm that the components of the combined carcinoma are not metastases of primary tumors in other locations, namely the small cell component (not present in the metastatic neck adenopathy) being imperative to exclude other neoformations, specifically in the lungs, hence the fact that PET was performed.
Treatment:
The proposed treatment, after a multidisciplinary consultation in a head and neck cancer group, was concomitant chemo radiotherapy. It was planned to perform three cycles of high dose cisplatin chemotherapy but the patient only tolerated the performance of two cycles, having been later hospitalized for complaints of neck pain and intolerance of the nasogastric feeding tube placed in the context of post-radiotherapy oral mucositis. During the hospitalization, pain was controlled through the administration of analgesia, namely tramadol 100 mg 3id, paracetamol 1000 mg 3 id and 10 mg of subcutaneous morphine in SOS (up to a maximum of 4 hours interval between administrations), whenever, despite the fixed analgesia prescribed, the patient had pain complaints. Also, a percutaneous gastrostomy was performed. The pain ended up being controlled, maintaining fixed analgesia at home and feeding by percutaneous gastrostomy until the mucositis condition improved, after which he was able to eat orally again. Regarding radiotherapy regimen, the patient performed 35 radiotherapy fractions, VMAT technique (intensity modulated arc therapy).
Outcome and Follow-Up:
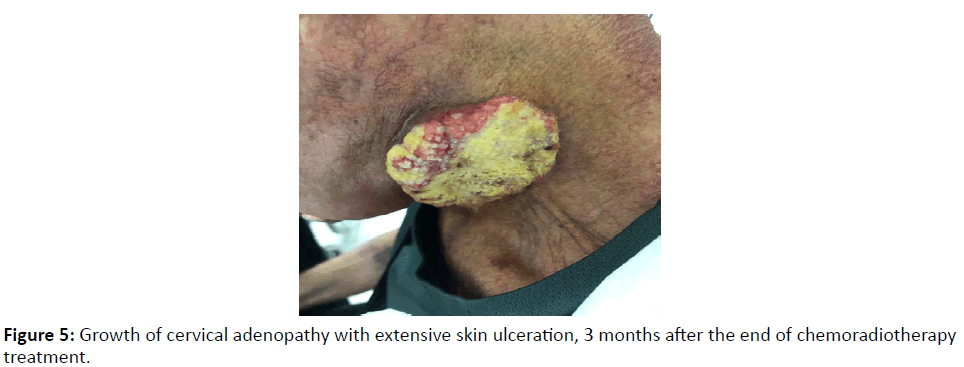
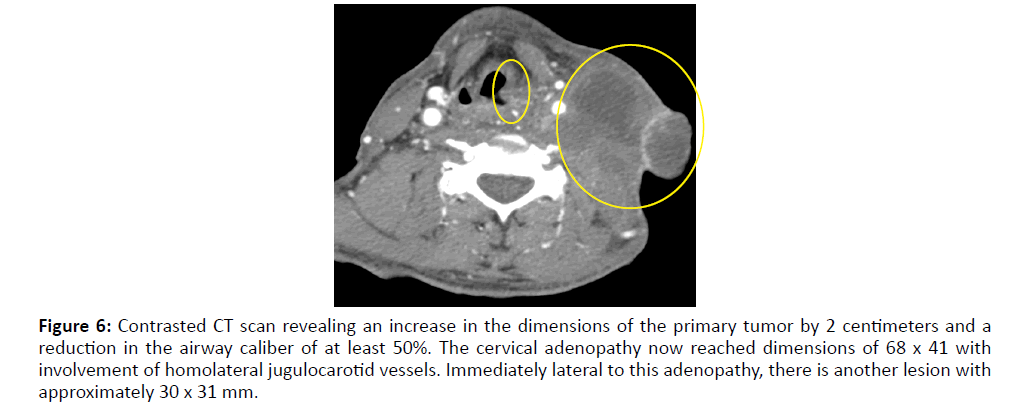
The patient was evaluated in an Otorhinolaryngology consultation 3 months after the end of the initial treatment to evaluate the therapeutic response. He presented with severe complaints of neck pain despite being on oral tramadol 100 mg 3 id, oral paracetamol 1000 mg 3 id and oral morphine 10 mg 3 id solution at home. He also complained of dysphonia again, but without dysphagia (he was on an oral diet and did not use percutaneous gastrostomy) and without dyspnea. On physical examination, nasopharyngolaryngoscopy has now revealed an extension of the hypopharynx neoformation to the ventricular band, ventricle and vocal cord ipsilateral to the lesion, conditioning left hemilarynx fixation, with maintained mobility of the right vocal cord, allowing a reasonable glottic lumen. Neck examination showed increased dimensions of the adenopathic conglomerate, with extensive skin ulceration (Figure 5). The patient underwent a new CT scan for restaging of the disease, which revealed an increase in the dimensions of the primary tumor by 2 centimeters, leading to a reduction in the airway caliber of at least 50%. The cervical adenopathy now reached dimensions of 68 x 41 centimeters and clearly involved the homolateral jugulocarotid vessels, without signs of thrombosis. Immediately lateral to this adenopathy, in the cervical superficial planes, another lesion with approximately 30 x 31 mm of axial axes was again identified, compatible with the progression of the underlying oncological disease (Figure 6). There was no evidence of distant metastases. Combined hypopharyngeal carcinoma was restaged as ycT3N3bM0. The patient was referred to multidisciplinary head and neck consultation, where he was proposed to undergo palliative chemotherapy with paclitaxel and cetuximab. The patient was not able to even complete the first cycle, due to worsening of his general condition. He was followed up by a home palliative care team, so that comfort measures could be privileged, and died 9 months after diagnosis and 6 months after the end of the initial treatment with chemoradiotherapy.
Figure 5:Growth of cervical adenopathy with extensive skin ulceration, 3 months after the end of chemoradiotherapy treatment.
Figure 6:Contrasted CT scan revealing an increase in the dimensions of the primary tumor by 2 centimeters and a reduction in the airway caliber of at least 50%. The cervical adenopathy now reached dimensions of 68 x 41 with involvement of homolateral jugulocarotid vessels. Immediately lateral to this adenopathy, there is another lesion with approximately 30 x 31 mm.
Discussion:
Combined small cell carcinoma is a very rare entity, especially in the hypopharynx, as we can infer from the fact that there have been only six cases published in the medical literature worldwide so far, with the first case described by Ferlito et al., in 1980 [4]. These carcinomas are thought to originate from a pluripotent stem cell that develops features of squamous cell carcinoma as well as neuroendocrine features or from a neoplastic cell with divergent differentiation potential, but the pathophysiological mechanism is still unclear [3,7,11].
Combined small cell carcinoma is more frequent in males, between the sixth and seventh decades of life, and in the cases described so far in the hypopharynx, ages ranged between 49-75 years, all of which were men, as in our clinical case. The most frequent presenting signs/symptoms are the appearance of cervical swelling and dysphagia. In the 6 reported cases, all patients had cervical swelling and/or dysphagia as the initial sign/symptom, except for one case in which the first symptoms were odynophagia and dysphonia [2,4,5,10,11]. Also in our case, the initial sign was cervical swelling with rapid growth, still without other associated symptoms. In our case dysphonia appeared later, associated with tumor growth and disease progression.
The diagnosis is established through biopsy and turns out to be a difficult diagnosis, because both neoplasm components must be present in the biopsied material. Generally, in these combined small cell carcinomas, the small cell histologic component is located deeper in the tumoral mass [5]. This leads us to think that probably not all cases of combined small cell carcinoma are diagnosed, because only the most superficial component (squamous cell carcinoma or adenocarcinoma component) ends up being included in the biopsy and a diagnosis of squamous cell carcinoma or adenocarcinoma is more frequently established. In the six cases described, cervical lymph node metastasis revealed all to be from combined small cell carcinoma, having both components of the tumour [2,4,5,10,11]. In our case, metastasis was only from the squamous cell carcinoma component, which seems to be rarer. We can also question whether the ganglion microbiopsy was deep enough and could not have included the small cell component. Only one of the reported cases had no regional or distant metastases at presentation, while four had cervical lymph node metastases and one had distant, liver and bone metastases. Distant metastasis of these tumours occurs more often on the liver, bones or lungs. Of the six reported cases, four died within one year of diagnosis [2,4,5,10,11].
Mainly because of its rarity, but also because of the very poor prognosis associated with the described cases of combined small cell carcinoma of the head and neck region and specifically of the hypopharynx, there are no guiding therapeutic guidelines and what ends up being recommended is a treatment similar to the one applied for small cell neuroendocrine tumours, the most aggressive component of combined small cell carcinoma [2,5]. This component is, therefore, the one with the greatest propensity for regional and distant metastases [2]. According to the literature, patients with combined small cell carcinoma of the hypopharynx have an overall 2-year survival rate of 8.5% [1,11]. In therapeutic terms, the most consensual treatment involves systemic medical treatment with concomitant chemoradiotherapy (such as in the treatment of small cell lung and extrapulmonary carcinoma), thus opting for an organ-sparing strategy [7,11]. Platinum-based agents are known to be effective in the treatment of squamous cell carcinoma and in the treatment of small cell carcinoma, although in combined small cell carcinoma there is some resistance to existing chemoradiotherapy protocols, hence the resulting poor prognosis [1,11]. However, there are authors who advocate multimodal treatment, with radical surgery followed by adjuvant treatment, which was the strategy adopted in most of the cases described in the literature [4,5,10].
Learning Points/Take Home Messages:
1. Combined carcinoma means that there are two carcinomas in the same tumoral mass, with a clear histological separation between them.
2. Combined small cell carcinoma of the hypopharynx is a very rare clinical entity, with only 6 cases described in literature so far, to the best of our knowledge.
3. Diagnosis of combined carcinomas is difficult and the biopsy must be profound enough to include both tumor components.
4. There are still no established therapeutic guidelines for the treatment of combined small cell carcinoma, but chemo radiotherapy is the most consensual treatment.
5. Combined small cell carcinoma of the hypopharynx has a very poor prognosis, with patients having an overall 2-year survival rate of 8.5%.
Conclusion
In our opinion, and considering the cases described, the evolution of the disease and the prognosis, with a high propensity for early metastasis, even in patients with a clinically localized tumour, systemic treatment is the preferred option.
References
- Flint P, Haughey B, Lund V, Robbins K, J. Regan Thomas, et al. (2021). Franciset al. Cummings Otolaryngology. 7 ed. Philadelphia: Elsevier.
- Misawa K, Kawasaki H, Endo S, Mochizuki D, Morita K, et al. (2016) Primary combined small and squamous cell carcinoma of the hypopharynx: A case report. 4:709-14.
- Aggarwal G, Jackson L, Sharma S (2011) Primary combined small cell carcinoma of larynx with lateralized histologic components and corresponding side-specific neck nodal metastasis: report of a unique case and review of literature. Int J Clin Exp Pathol 4:111-117.
- Ferlito A, Recher G, Caruso G (1985) Primary combined small cell carcinoma of the larynx. Am J Otolaryngol 6:302-308.
- Misawa K, Kawasaki H, Endo S, Mochizuki D, Morita K, et al. (2016) Primary combined small and squamous cell carcinoma of the hypopharynx: A case report. Mol Clin Oncol 4:709-14.
- Zhang G, Chen Y, Li C, Liu M (2018) Primary combined squamous and small cell carcinoma of the larynx: a case report and review of the literature. In J Clin Exp Med 11:10136-0139.
- Yu Q, Chen YL, Zhou SH, Chen Z, Bao YY, et al. (2019) Collision carcinoma of squamous cell carcinoma and small cell neuroendocrine carcinoma of the larynx: a case report and review of the literature. World J Clin Cases 7:242-252.
- Coca-Pelaz A, Triantafyllou A, Devaney KO, Rinaldo A, Takes RP, et al. (2016) Collision tumors of the larynx: A critical review. Am J Otolaryngol 37:365-368.
- Ryan N, Walkden G, Lazic D, Tierney P (2015) Collision tumors of the thyroid: A case report and review of the literature. Head Neck 37: E125-E129.
- Mills SE, Cooper PH, Garland TA, Johns ME (1983) Small cell undifferentiated carcinoma of the larynx: report of two patients and review of 13 additional cases. Cancer. 51:116-120.
- Nakahira M, Kuba K, Matsumura S, Sugasawa M (2017) Primary small cell carcinoma of the hypopharynx: a report of two cases and review of nine additional cases. Case Rep Otolaryngol 2017.
- Bares L, Eveson JW, Reichart P, Sidransky S (2005) Neuroendocrine tumors. World Health organization classification of tumors, pathology and genetics of head an neck tumors. IARC 135-139.
- Amin MB, Edge SB, Greene FL, Compton CC, Gershenwald JE, et al. (2017) AJCC Cancer Staging Manual. 8th Ed. New York: Springer.
- Ferlito A, Silver CE, Bradborf CR, Rinaldo A (2009) Neuroendocrine neoplasms of the larynx: an overview. Head Neck 31:1634-46.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref