Case Report - Journal of Gastroenterology and Digestive Diseases (2018) Volume 3, Issue 3
Peutz-Jeghers syndrome revealed by recurrent vomiting.
Lamia Gargouri1, Bayen Maalej1, Hamdi Louati2*, Manel Weli1, Lassad Chtourou3, Yosr Hentati4, Zeineb Mnif4, Leila Mnif3, Mahdi Ben Dhaou2, Nabil Tahri3, Riadh Mhiri2 and Abdelmajid Mahfoudh1
1Department of Pediatrics, Pediatric Emergency and Intensive Care, Hedi Chaker Hospital, Sfax, Tunisia
2Department of Pediatric Surgery, Hedi Chaker Hospital, Sfax, Tunisia
3Department of Gastroenterology, Hedi Chaker Hospital, Sfax, Tunisia
4Department of Radiology, Hedi Chaker Hospital, Sfax, Tunisia
- *Corresponding Author:
- Hamdi Louati
Department of Pediatric Surgery
Hedi Chaker Hospital
Sfax, Tunisia
Tel: +216 74 244 422
E-mail: drhamdilouati85@yahoo.com
Accepted on June 29, 2018
Citation: Gargouri L, Maalej B, Mahfoudh A, et al. Peutz-Jeghers syndrome revealed by recurrent vomiting. J Gastroenterol Dig Dis. 2018;3(1):44-46.
Abstract
Background: Peutz-Jeghers syndrome is a rare, autosomal dominant inherited disease caused by mutation of STK11/ LKB1 (serine threonine kinase 11) characterized by gastrointestinal hamartomatous polyps, mucocutaneous pigmentation and increased risk of malignant disease. The case of a 12-year-old girl with perioral pigmentation who present with recurrent vomiting and weight loss is reported. Endoscopic and radiologic investigation demonstrated a mass in the second portion of the duodenum. She underwent surgery because of a sharp abdominal pain caused by bowel intussusception. Laparotomy revealed two polyps: one in the second portion of the duodenum: Intussusception could be easily reduced and polyps are resected. Histologically, it was a hamartoma.
The child was found to be heterozygote for the mutation c.863-2A>C in intron 6 of the STK11 gene. The patient is followed and is well.
Conclusion: Peutz-Jeghers syndrome should be suspected in any child suffering from recurrent abdominal pain and /or other gastrointestinal symptoms in the presence of characteristic pigmentation.
Keywords
Peutz-Jeghers syndrome, Hamartomatous polypes, Lentiginosis, Bowel intussusception.
Introduction
Peutz-Jeghers syndrome is characterized by the association of two symptoms: gastrointestinal hamartomas mainly located in the small intestine and lentiginosis essentially on the lips and oral mucosa [1]. It is inherited as an autosomal dominant disorder. It is due to heterozygous mutations in the serine-threonine kinase STK11 tumor suppressor gene located on chromosome 19p13.3 [2]. The main risk that we should emphasize on is the amplified risk of cancer in many organs especially in the small bowel [1-3].
We described a 12-year-old girl presented with a two-month history of recurrent vomiting complicated by bowel intussusception. Two polyps were found to be the cause of an intussesception and be resected. Histology confirmed that it was a hamartomatous polyp.
Case Report
A 12-year-old girl presented with two months history of an epigastric pain, recurrent vomiting and weight loss of 6 kg. Her parents were consanguineous and there was no family history.
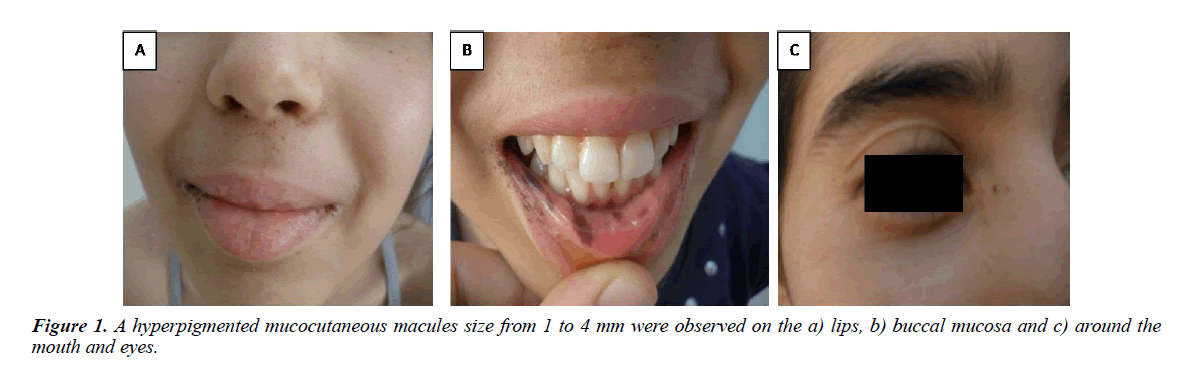
On physical examination, she was dehydrated and pale. She weighed 30 kilograms (-1.5 DS) and she has a normal height (145 cm). Her BMI was lower than 3rd percentile. Abdominal examination revealed diffuse tenderness essentially in the right side of the abdomen. A hyperpigmented mucocutaneous macules size from 1 to 4 mm were observed on the lips, buccal mucosa and around the mouth and eyes (Figure 1).
Laboratory studies revealed: serum sodium concentration, 118 mEq/l; potassium, 3.9 mEq/l; chloride 102 mEq/l, blood urea nitrogen 19 mmol/l, serum creatinine, 130 μmol/l. The complete blood cell count showed white blood cells 9100/mm3, hemoglobin 6 g/dl, hematocrit 18% and platelet count 150000/ mm3. Serum iron level was 4 μmol/l. Intravenous rehydratation with restoring electrolyte balance and a blood transfusion were done.
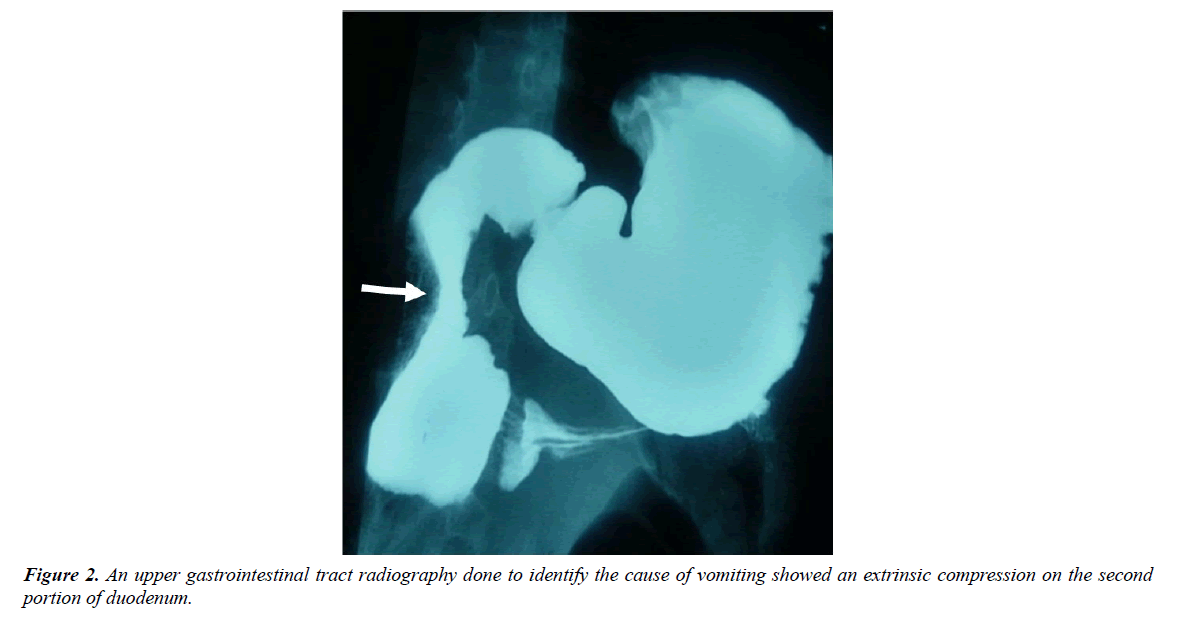
Abdominal ultrasonophy was normal. An upper gastrointestinal tract radiography done to identify the cause of vomiting showed an extrinsic compression on the second portion of duodenum (Figure 2). The patient underwent esophagogastroduodenoscopy that showed a mass of 2 × 2 cm in size at the second duodenum.
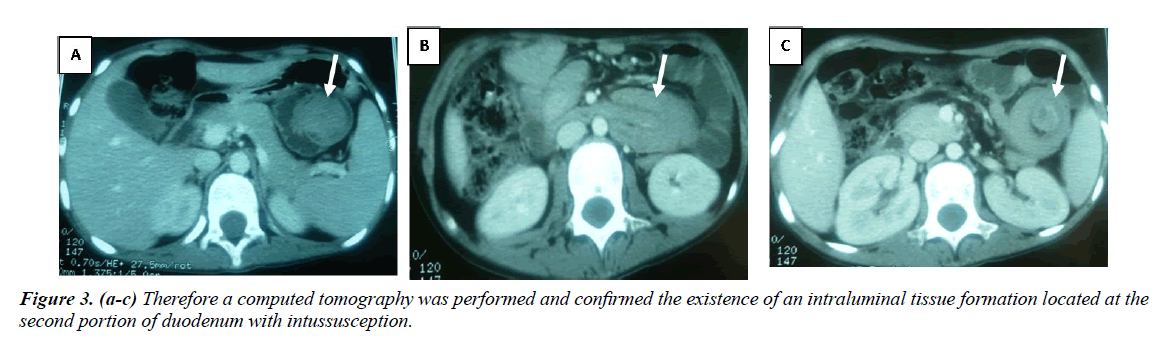
The patient had during her hospitalization a sharp abdominal pain. Therefore a computed tomography was performed and confirmed the existence of an intraluminal tissue formation located at the second portion of duodenum with intussusception (Figure 3).
She underwent surgery. Intussusception could be easily reduced. Intraoperative findings revealed 2 polyps, in the second portion of the duodenum, that were removed. The largest polyp measured 3.5 × 3 × 2.7 cm. The second polyp was smaller and measured 0.4 cm in diameter and was localized at 1.5 cm from the first. The exploration by the introduction of the optic up to 70 cm from the second portion of the duodenum did not find other polyps.
Histological examination of the polyps revealed branching bundles of smooth muscle fibers covered by hyperplasic duodenal mucosa defined as a hamartomatous polyps and confirmed the diagnosis of Peutz Jehgers syndrome. Genetic study identified a heterozygous mutation c.863-2A>C in intron 6 of the STK11 gene.
Abdominal complaint and vomiting were fading after surgery and the patient gained weight. After tow year of following up, she was currently asymptomatic. Her weight is 45 kg (-0.5 DS). The abdominal computed tomography scanning with oral contrast did not showed other polyps.
Discussion
Peutz-Jeghers syndrome is a rare disorder. The estimated prevalence is approximately 1/200 000 births [2,3,4]. It is clinically characterized by gastrointestinal hamartomatous polyps, mucocutaneous pigmentation and the increased risk of developing cancer [1].
It is an autosomal dominant inherited disease caused by the mutation of the serine/threonine kinase gene (STK11/LKB1). This gene is located in the chromosome 19p13.3 and it has been suggested that it is a tumor suppressor gene [2]. The germline mutation was identified in 30% to 80% of PJS cases [2]. Our patient was found to be heterozygote for the mutation c.863-2A>C in intron 6 of the STK11 gene. To the best of our knowledge, it was the first description of this mutation.
Mucocutaneous hyperpigmentation is observed in more than 95% of PJS patients and it appears at an early age and tends to fade during puberty but buccal mucosa lesions persist. It’s a brown-black macula measuring 2-4 mm in size. It’s located in the perioral region and in the buccal mucosa, in various facial, body and oral surfaces [2,3].
Intestinal polyps are mostly found in the small bowel [1,5]. The number of polyps does not exceed 10 in the majority of cases. Most patients develop gastrointestinal symptoms between the ages of 10 and 30 years. Neonatal detection is unusual [6]. Intestinal polyps can cause abdominal pain due to intermittent intussusception or to obstruction which presents as recurrent vomiting, as seen in our patient [2,3,5]. Intestinal polyps also can produce occult bleeding into the gastrointestinal lumen causing iron deficiency anemia [3,4].
Surgical treatment is frequently required for gastrointestinal involvement and. Laparotomy may be indicated for small intestinal intussusception, obstruction or polyp’s removal [2].
Diagnosis of PJS can be established by the following criteria: [1] three or more benign hamartomatous polyps confirmed by histology, or [2] any number of hamartomatous polyps with a family history of PJS, or [3] characteristic mucocutaneous pigmentation with a family history of PJS, or [4] any number of hamartomatous polyps and mucocutaneous pigmentation [7,8].
Patients with PJS have 15 times greater risk for cancer than general population. Malignancies include gastrointestinal tract carcinoma or cancers in other organs such as: pancreas, lung, breast, ovaries, uterus and testis [9,10].
That’s why, regular cancer screening is recommended. Upper and lower gastrointestinal endoscopy should be done every two years from 10 years of age or at the time of the first symptom. Screening for extra-intestinal cancers should began around the age of 20 years with pelvic and breasts examination once yearly, an annual abdominal and pelvic ultrasound and a mammogram every 2 to 3 years [9-11].
Conclusion
Physicians must suspect PJS in any child suffering from recurrent abdominal pain and /or other gastrointestinal symptoms in the presence of characteristic pigmentation. These patients should be regularly and closely monitored because of the risk of malignant disease in the gastrointestinal tract and extra intestinal organs. It is also important to screen family members.
References
- Susanne KE, Pieter D, Ernst KJ, et al. Small bowel endoscopy and Peutz-Jeghers syndrome. Best Pract Res Clin Gastroenterol. 2012; 26(3):263-278.
- Paola H, Faizan A, Eric ST. Medical management update: Peutz Jeghers syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010; 109(1): 5-11.
- Xingwei W, Chaoqiang F, Dan Q, et al. Peutz-Jeghers syndrome: a report of 5 cases. Journal of Medical Colleges of PLA. 2011; 26(4): 238-241.
- Meng-Tzu W, Yen-Hsuan NI, Yi-Nien SU, et al. Clinical and genetic analysis of Peutz-Jeghers syndrome patients in Taiwan. Journal of the Formosan Medical Association. 2010; 109(5): 354-361.
- Francis G, Stephen G, Malcolm D, Peutz–Jeghers syndrome: A case report and discussion of surveillance recommendations. Eur J Radiol Open. 2007; 62(3): 81-84.
- Christine B, Felix S, Gundula S. Gastric outlet obstruction in a neonate because of Peutz-Jeghers syndrome. J Pediatr Surg. 2012; 47(8): e1-e3.
- Francis GM, Jill TD. Peutz-Jeghers syndrome and management recommendations. Clin Gastroenterol Hepatol. 2006;4(4): 408-415.
- Ian Roy S, Maria B, Christopher A, et al. The hamartomatous polyposis syndromes: a clinical and molecular review. Am J Gastroenterol. 2005; 100(2): 476.
- Hinds R, Philp C, Hyer W, et al. Complications of childhood Peutz-Jeghers syndrome: implications for pediatric screening. J Pediatr Gastroenterol Nutr. 2004; 39(2): 219-220.
- Lier V, Wagner MGF, Mathus-Vliegen A, et al. High cancer risk in Peutz–Jeghers syndrome: A systematic review and surveillance recommendations. Am J Gastroenterol. 2010; 105(6): 1258.
- Nicholas H, Valerie S, Fred MH, et al. Frequency and spectrum of cancers in the Peutz-Jeghers syndrome. Clin Cancer Res. 2006; 12(10): 3209-3215.