Research Article - Journal of Infectious Diseases and Medical Microbiology (2018) Volume 2, Issue 3
Pattern of antibiotic usage and predictors of hospital outcome among patients with systemic bacterial infection in Nekemte referral Hospital, Western Ethiopia.
Getachew Alemkere1*, Getu Bayisa2, Abrham Belachew3
1Department of Pharmacology and Clinical Pharmacy, School of Pharmacy, College of Health Science, Addis Ababa University, Ethiopia
2Department of Pharmacy, College of Health Science, Wollega University, Wollega, Ethiopia
3Department of Pharmacy, College of Health Science, Bahir Dar University, Bahir Dar, Ethiopia
- *Corresponding Author:
- Getachew Alemkere
Department of Pharmacology and Clinical Pharmacy
School of Pharmacy
Addis Ababa University
Ethiopia
Tel: +251-118-959055
E-mail: getachewalemkere@gmail.com
Accepted date: August 02, 2018
Citation: Alemkere G, Bayisa G, Belachew A. Pattern of antibiotic usage and predictors of hospital outcome among patients with systemic bacterial infection in Nekemte referral Hospital, Western Ethiopia. J Infectious Disease Med Microbiol. 2018;2(3):12-20.
Abstract
Objective: Pattern of antibiotic usage and predictors of hospital outcome among patients with presumed systemic bacterial infection in Nekemte Referral Hospital Western Ethiopia. Methods: An institution based prospective observational study was performed from December 1 to March 30, 2017 in the internal medicine wards of Nekemte Referral Hospital. Patients who had presumed systemic bacterial infections were strictly followed. Data was collected on demographic, disease and drug related factors using a data abstraction format. Antibiotic use practice was described and predictors for mortality and length of stay were identified. Descriptive statistics and binary logistic regression were used for statistical analysis. Results: Females accounted for about 55% of the total 193 study participants whose mean (± SD) age was 39.97 ± 17.12. More than half (58.6%) of the participants had presumed systemic bacterial infections on admission. Whilst pneumonia was the first most prevalent infection presumed (47.7%), cephalosporins were the most widely prescribed (66.7%) class of drugs. Only one culture and 8 gram stain reports were documented and all the drugs were empirically used. About 8% of the wards patients were died during the in-hospital stay. The mean (± SD) inhospital length of stay was 6.98 ± 3.22 days (range: 3-18). While presence of a medical device was a positive predictor (AOR=4.50, 95% CI: 1.09, 18.60, p=0.038) and prolonged length of stay was the negative predictor (AOR=0.22, 95% CI: 0.05, 0.90, p=0.035) of mortality. On the other hand only presence of multidrug resistance (MDR) risk (AOR=6.14, 95% CI: 1.68, 22.41, p=0.006) was positively associated with prolonged in-hospital length of stay. Conclusion: Generally, these observations showed that all patients with systemic bacterial infection received antibiotics on an empiric basis. Broad spectrum third generation cepalosporins were the most commonly used drugs. These warrant an appropriate antimicrobial use policy in the context of resource-limited settings.
Keywords
Antibiotic use, Length of Stay, In-hospital Mortality, Nekmte referral Hospital, Western Ethiopia.
Introduction
Based on alarming accumulated facts in the previous few years, antimicrobial resistance is an increasingly important patient safety and public health issue [1]. About half of antimicrobial agents prescribed to hospital in-patients are considered inappropriate [1]. As per the different studies undertaken in Tikur Anbessa Specilaized Hospital (TASH) in the previous year, antimicrobials were the most common drug categories associated with both prescribing [2,3] and administration errors [3]. These malpractices have been associated with increased mortality, adverse drug reactions, financial cost and the development of resistant bacteria, which hold the threat to the generation [4-6]. As a result, it is more difficult than ever to challenge infections caused by antibiotic-resistant microbes [5].
Different approaches have been promoted to save these precious drugs from the threat of resistant bacterial selection [7-9].
Although resistance is a global concern, it is primarily a local problem: selection for and amplification of resistant members of a species are occurring in individual hospitals (and communities), which can then spread worldwide [10,11]. Single and multiple drug resistance to the commonly used antibiotics were high among bacterial isolates in different areas of Ethiopia, warranting rational use of drugs in the local environment [12,13]. Thus, it will need a widespread effort at the individual institutional level to impact antimicrobial usage and, by extension (hopefully), antimicrobial resistance.
This study was done to describe the pattern of antibiotic use, to identify priority areas of future intervention and to set appropriate recommendations that can help to improve future antibiotic utilization in the hospital. The aim of the current study is therefore, to describe different types of infections and drugs used to treat such infections; to quantify the changes/adjustments made to the initial therapy; to determine appropriateness of antibiotic use using standardized quality indicators and to identify independent predictors of prolonged hospital stay and in-hospital mortality.
Method
Study setting and period
The study was conducted in Nekemte referral Hospital (NRH), Nekemte town, Eastern Wollega zone, Oromia regional state which is located at 331 km from west of Addis Ababa. NRH was established in 1932 (1924 E.C) by Swedish missionary. Hospital catchment population is more than 2,000,000. It has a different departments and wards like outpatient department (OPD), medical wards, gynecology and obstetrics ward, pediatrics ward and surgical ward. The internal medicine ward had 60 beds. The study was conducted in all these internal medicine beds, since most adult cases were supposed to be seen in this unit. It was conducted from December 1 to March 30, 2017 for 4 consecutive months. Almost all the recording systems of the hospital during the study period were carried manually.
Study design
Institution based cross-sectional study was conducted.
Study population and sampling
All patients attending the adult internal medicine wards of NRH during the study period and who had presumed systemic bacterial (non-mycobacterial) infection were considered.
Inclusion criteria: All in-patients admitted to the adult internal medicine wards, prescribed and dispensed with systemic antibacterial agents during the study period were included in the study. Clients not dispensed with antibacterial agents; clients taking antibacterial for <72 hours; clients aged <18 years; clients lost to be followed or discharged against the medical advice; clients taking anti-mycobacterial, non-systemic antibacterial, prophylactic antibacterial were excluded.
Study variables
Dependent variables: Pattern of antibiotic usage, in hospital mortality and prolonged LoS (taking the Mean value).
Independent variables: Socio-demographic factors; Diseases and related factors; Drugs and related factors; guideline concordant use and intravenous to oral conversion for appropriateness of therapy.
Data collection process
Data abstraction format was adopted and validated from TASH study [14]. The data collectors were three nurse staffs at the hospital. To maximize quality of the data, training, supervision and monitoring of the data collectors was conducted. In addition, pre-test was conducted using 5% of the initially assumed antibiotic user patients, outside of the study period. In each ward, the principal investigator was the supervisor leading the team of data collectors and fully responsible for the whole quality of data. He assured that all the facilities were measured in the same way in all the months considered.
Data interpretation and statistical analysis
All adult patients admitted to the internal medicine wards during the study period were eligible for enrollment. Those who had presumed systemic bacterial (except mycobacterial) infection either on admission or after admission (in the hospital stay) and for whom antibiotics prescribed, were strictly followed. Demographic characteristics, admission diagnosis, presumed infection diagnosis, laboratory procedures for the infection diagnosed (culture and gram stain), antibiotics administered, and adjustments made on the antibiotic regimen were recorded properly on daily basis. The dates’ of each antibiotic initiation were also included. Criteria used for diagnosis, microbiologic techniques, the decision to prescribe and modify antibiotics were left to the physician’s discretion. Then after, those who did not satisfy the inclusion criteria and the intent of the data interpretation were excluded from the analysis.
The collected data was checked, cleaned and double entered. Epi info 7 for entry and SPSS for windows version 21.0 for data analysis, were used. Descriptive statistics such as means were used to summarize descriptive data. Binary logistic regression was carried out to see the association after all continuous variables were converted to categorical. Variables having p<0.25 in the univariate models were included in the final model of multivariate logistic regression. Crude and Adjusted Odds Ratio (COR/AOR) at 95% confidence level were calculated and the association was declared significant at p<0.05.
Ethical consideration
Ethical approval for the protocol was obtained from the Department of Pharmacy, College of Health Sciences, Wollega University. The letter was then communicated to the Hospital administrators and the staffs in the internal medicine ward. Written informed consent was not obtained from patients and health personnel’s, since the study was strictly observational that did not modify any clinical practice. Instead, information was given to the patients, their physicians and other health workers, as required. To ensure confidentiality, name and other identifiers of patients and prescribers were not recorded on the data abstraction formats. The collected data was kept in a locked cabinet and only the researchers had access to the data.
Operational definitions
Length of stay (LoS): LoS was defined as the number of days (Midnights) between admission and discharge, regardless of the number of hours, because the precise time was often not available. Prolonged LoS is to mean the LoS above the mean (>7 days).
Signs and symptoms of disease: Based on ICD 10, it refers to the signs and symptoms of the underlying disease (e.g., hemiparesis, secondary to hypertension) that were not classified elsewhere under the primary admission diagnosis but which were the primary reasons for admission.
Antibiotics: In this study refers to any drugs used for systemic bacterial infection.
Adjustment: Any changes made on the antibiotic/regimen after 48-72 hours of the initial therapy that refers to either of following.
• Discontinued: To mean any discontinuation of all antibiotics found to be unnecessary (e.g., no presumed infection); Modified: to mean either de-escalation (narrowing by either discontinuation of either agent or using the narrower spectrum option) or broadening (addition or using a much broader spectrum instead or starting a new regimen of treatment after a day and before 7 days of completion of the first course of treatment period) of therapy; Antibiotic metrics: refers to the following antibiotic use measures [15].
• Antibiotic courses: Any period during which the same agent (regardless of dose or route) was administered to the same patient on consecutive days; Treatment periods: a period of consecutive days on which any antibiotic or combination of antibiotics was administered to a patient; Agent days: the number of days that a patient received a particular agent during the ward admission period; Antibiotic days: the number of days on which a patient received any antibiotics during the ward admission period.
Results
Socio-demographic and disease characteristics of patient
Patients had a mean age of 39.97+17.12 (range: 18-80). Females accounted for about 55% of the study participants. Of all, 58.6% of the patients had infection during ward admission. Patients with circulatory diagnosis (26.9%) accounted for the second most category of primary admission diagnosis. Only one patient has documented culture report. Twenty six (13.5%) patients had multidrug resistant infection risk. Thirty five (18.1%) patients had underling immunosuppression (Table 1).
Table 1. Socio-demographic and disease characteristics of hospitalized patients with bacterial infection in the internal medicine ward of Nekmte Referral Hospital in 2017, Nekemte, Ethiopia.
| Variables | Total, N=193 (Freq, %) | |
|---|---|---|
| Average age (mean + SD (range)) | 39.97+ 17.12 (18-80 | |
| Sex | Female | 107 (55.4) |
| Male | 86 (44.6) | |
| Admission Diagnosis (ICD 10)† | ||
| Infectious 113 (58.6) | Infectious 113 (58.6) | |
| Circulatory 52 (26.9) | Circulatory 52 (26.9) | |
| Digestive 37(19.2) | Digestive 37(19.2) | |
| Signs and symptoms of disease 21 (10.9) | Signs and symptoms of disease 21 (10.9) | |
| Endocrine & metabolic 21 (10.9) | Endocrine & metabolic 21 (10.9) | |
| Respiratory 20 (10.4) | Respiratory 20 (10.4) | |
| Genitourinary 19 (9.8) | Genitourinary 19 (9.8) | |
| Blood related 4 (2.1) | Blood related 4 (2.1) | |
| Other Diagnosis†† 10 (5.18) | Other Diagnosis†† 10 (5.18) | |
| Microbiologic reports | Gram stain | 8 (41.45) |
| Culture | 1 (0.52) | |
| MDR risk ♦ | Absent | 123 (63.73) |
| Present | 26 (13.47) | |
| No enough evidence | 44 (22.80) | |
| Imunocompromized ♦ ♦ | Yes | 35(18.1) |
| No | 158(81.9) | |
†a given patient may have >1 diagnosis, based on International classification of disease (ICD); †† (Others: Psychosis(2,)Erythromathos (2), Breast Abscess (1), Dementia (1), Fracture (1), Genital Ulcer (1), Scar (1) and Status Epileptics (1)); SD-standard deviation; HIV-human immunodeficiency virus; ♦MDR risk criteria: prior antibiotic receipt in the past 3 months, previous hospital admission during the last 3 months, late-onset hospital-acquired infections (HAIs) (as defined by the physicians and/or the date of antibiotic administration relative to the admission date) (i.e., >5 days after admission), and presence of preexisting immunosuppressive disease (IDSA/ATS, 2005); ♦ ♦ patients with febrile neutropenia, cirrhosis, disseminated TB and HIV infection.
Of all the patients, 47.67% had pneumonia. Community acquired pneumonia (72.83%) and aspiration pneumonia (15.22%) were the commonest types of pneumonia presumed, respectively. Patients with Acute febrile infections (13.47%) and meningitis (13.47%) respectively were the second and the third most common infections following pneumonia (Table 2).
Table 2. Types of systemic bacterial infection presumed in hospitalized patients in the internal medicine ward of Nekmte Referral Hospital in 2017, Nekemte, Ethiopia.
| Bacterial Diagnosis* | Total, N=193 (Freq, %) |
|---|---|
| Pneumonia | 92 (47.67) |
| Community acquired | 67 (72.83) |
| Aspiration | 14 (15.22) |
| Hospital acquired | 3 (3.26) |
| Other Pneumonia | 8 (8.70) |
| Acute febrile infections | 26 (13.47) |
| Meningitis | 20 (10.36) |
| Urinary Tract Infection | 12(6.22) |
| Sepsis | 7 (3.63) |
| Spontaneous Bacterial Peritonitis | 7 (3.63) |
| Post infectious Acute glomerular nephritis (AGN) | 6 (3.11) |
| Skin infections | 5 (2.59) |
| Surgical site infections | 3 (1.55) |
| Diabetic foot ulcer | 3 (1.55) |
| Other Bacterial infections** | 29 (13.47) |
*As per the labeling of the prescribing physician a given patient may have > 1 infectious diagnosis;
**Other bacterial infections:
Anthrax (GI) (1), Asthma Exacerbation (3), Breast Abscess (1), Group B Streptococcal myelitis (1), Genital ulcer Disease(1), infective endocarditis (1), Neuro-Syphilis (2), Opportunistic Infection (Bacterial) (3), Pancreatitis (2), Pleural effusion (3), Pulmonary Edema (1), Right Hydro Pneumothorax (1), Tetanus (2), Unknown (3), Upper respiratory infection (1)
Infection diagnosis
Antibiotics and antibiotic related factors
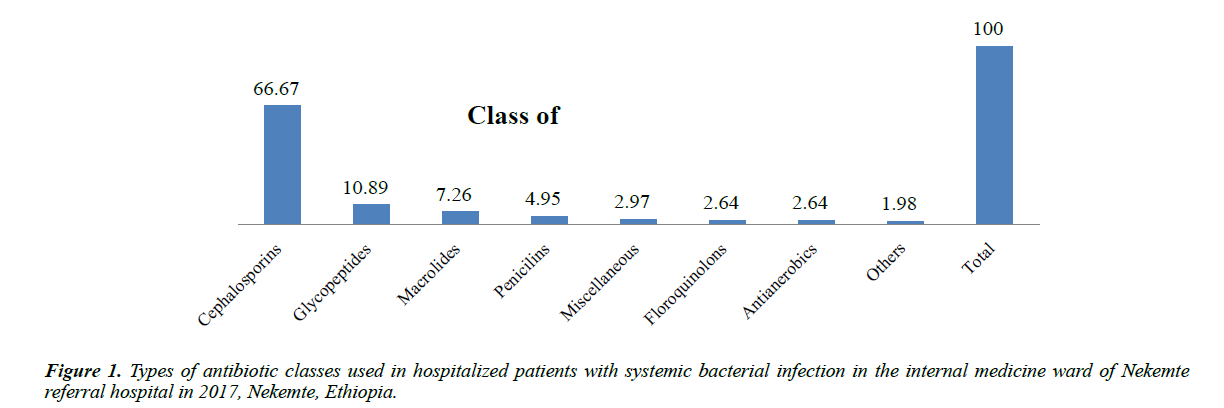
Class of and specific antibiotics used: Cephalosporin (202 (66.67%)), glycopeptides (33 (10.89%)) and macrolides (22 (7.26%)) were most prevalently used class of drugs (Figure 1).
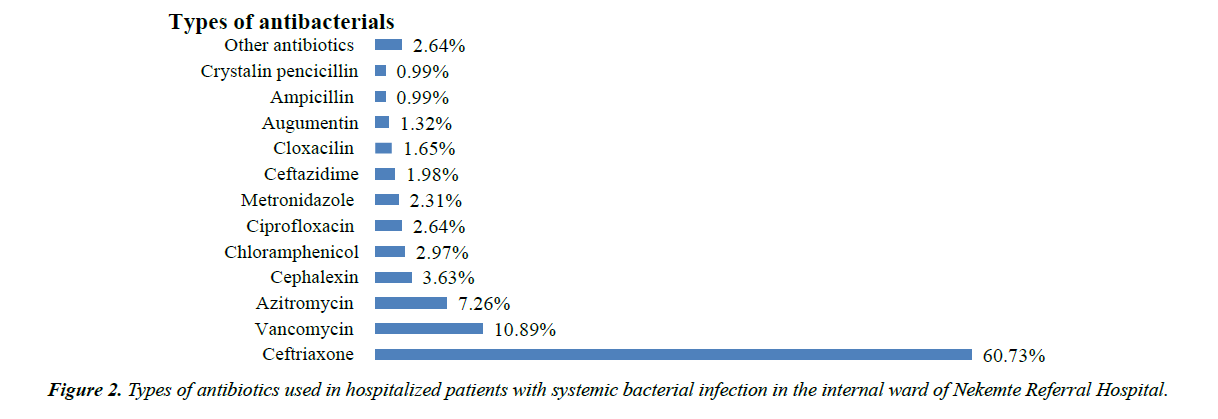
The most frequently prescribed antibiotics were ceftriaxone (60.73%), vancomycin (10.89%), Azithromycin (7.26%) and Cephalexin (3.63%) respectively (Figure 2).
Other Antibiotics: Cotrimoxazole (2), Doxycycline (2), Gentamicin (2), Clindamycin (1), Cefoxitine (1).
Antibiotic metrics: A given patient with a bacterial infection was exposed to 1-5 antibiotic courses with a mean of about 1.5 (Table 3). The mean number of days that elapsed while the patient was on any systemic antibiotic was 7.47+2.51 standard deviations, likewise, the average agent day was 10.20+6.37 standard deviations (Table 3).
Table 3. Antibiotic use based on different metrics for hospitalized patients with bacterial infection in the internal medicine ward of Nekmte referral hospital in 2017, Nekemte, Ethiopia.
| Variable | Frequency (N=193) (%) | Mean + Standard Deviation (Range) | |
|---|---|---|---|
| Agent days, Mean + SD (R) | 10.20+6.37 (3-35) | ||
| Antibiotic days, Mean + SD (R) | 7.47+2.51 (3-14) | ||
| Maximum no. of antibiotics at a time | 1.38+0.34 (1-3) | ||
| 1 | 130 (67.36) | ||
| 2 | 53 (27.46) | ||
| 3 | 10 (5.18) | ||
| Treatment periods | One | 189(97.93%) | 1.02+0.14 (1-2) |
| Two | 4 (2.07%) | ||
| Antibiotic course | 1 | 112 (58.03) | 1.54+0.73 (1-5) |
| 2 | 59 (30.57) | ||
| >3 | 22 (11.40) | ||
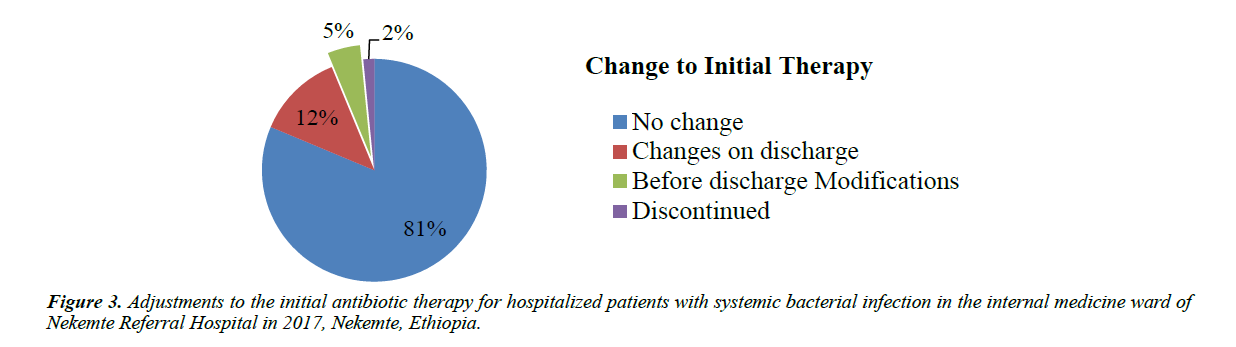
Changes to initial therapy: This portion specifically deals with the initial therapy and its adjusted component. The initial therapy was not adjusted in 157 (81.35%) patients. However, it was discontinued in 3 (1.55%) of the patients, and modified in 9 (4.66%) of the patients. The remaining patients, 24 (12.44%), had changes on discharge (Figure 3).
Appropriateness of antibiotic use: About 81% of patients had initial antibiotics prescribed according to the national guidelines. Of all 193 patients studied, 71 were not candidates for intravenous to oral switch therapy. Among the remaining 122 candidates for intravenous to oral switch, only 7 (5.74%) of them had oral switches (Table 4).
Table 4. Appropriateness of antibiotic use for hospitalized patients with systemic bacterial infection in the internal medicine ward of Nekmte Referral Hospital in 2017, Nekemte, Ethiopia.
| Quality indicators for appropriate antibiotic usage | Appropriate (Frequency(Percentage)) |
|---|---|
| Initial therapy was according to the guidelines | 157 of 183 = 81.35% |
| IV to PO changes made within 5 antibiotic days | 7 of 122 patients who had changes within <5 antibiotic days=5.74 % |
IV: Intravenous, PO: Per-oral route
Hospital outcome indicators: Of all, 16 (8.29%) patients were died during in the hospital stay. The average Length of in-hospital stay (mean + SD (range)) was 8.9+4.9 (3-23) days (Table 5).
Table 5. Hospital outcome indicators in hospitalized patients with bacterial infection in the internal medicine ward of Nekmte referral hospital in 2017, Nekemte, Ethiopia.
| Variables | Frequency (N=193) | Percent |
|---|---|---|
| Final status of patient | ||
| Improved | 159 | 82.38% |
| Dead | 16 | 8.29% |
| Referred | 11 | 5.70% |
| Medical Against | 7 | 3.63% |
| Length of Stay, mean + SD(R) | 6.979 + 3.22 (3-18) | |
SD: Standard deviation; R: Range
Predictors of mortality: Presence of signs and symptoms of disease (COR=4.57, 95% CI (1.41, 14.82), p=0.011), vancomycin use (COR=3.33, 95% CI (1.12, 9.93), p=0.031) and presence of MDR risk (COR=4.31, 95% CI: (1.35, 13.76), p=0.014) positively and guideline concordant antibacterial use (COR=0.25 95% CI (0.09, 0.73), p=0.011) negatively were associated with mortality in the univariate analysis but not in the multivariate regression model.
Presence of a medical device is a strong positive predictor of mortality both in the univariate (COR=8.61, 95% CI (2.92, 25.36), p=0.000) and multivariate models (AOR=4.50 (1.09, 18.60) p=0.038). Similarly prolonged length of stay is the negative predictor of mortality both in the univariate (COR=0.24, 95% CI (0.07, 0.87), p=0.029) and multivariate models (AOR=0.22 95% CI (0.05, 0.90) p=0.035) (Table 6).
Table 6. Binary logistic regression analysis for predictors of mortality for hospitalized patients with systemic bacterial infection in the internal medicine ward of Nekmte referral Hospital in 2017, Nekemte, Ethiopia.
| Variables | Mortality (%) | COR (95% CI) | AOR (95% C.I.) |
|---|---|---|---|
| Sex (male) | 10(11.6) | 2.22 (0.77, 6.36) | 1.30 (0.38, 4.46) |
| Immunosuppressed (yes) | 5 (14.3) | 2.23 (0.72, 6.88) | 3.67 (0.88, 15.32) |
| Presence of Medical Device (yes) | 9 (28.1) | 8.61 (2.92, 25.36)*** | 4.50 (1.09, 18.60)* |
| Signs and Symptoms of disease (yes) | 5 (23.8) | 4.57 (1.41, 14.82)* | 2.63 (0.56, 12.46) |
| Guideline concordant antibiotic use (yes) | 9 (5.7) | 0.25 (0.09, 0.73)* | 0.27 (0.06, 1.29) |
| Vancomycin use (yes) | 6 (18.2) | 3.33 (1.12, 9.93)* | 2.55 (0.56, 11.71) |
| Prolonged Length of stay (yes) | 3 (3.3) | 0.24 (0.07, 0.87)* | 0.22 (0.05, 0.90)* |
| MDR risk (Absent) | 8 (6.5) | (1.00) | (1.00) |
| MDR risk (unknown) | 2 (4.5) | 0.69 (0.14, 3.35) | 0.60 (0.10, 3.64) |
| MDR risk (Present) | 6 (23.1) | 4.31 (1.35, 13.76)* | 1.08 (0.22, 5.26) |
*p < 0.05; ***p<0.001; MDR-multidrug resistant infection; COR: crud odds ratio; AOR: Adjusted odds ratio; CI: confidence interval
Predictors of prolonged length of stay: Presence of comorbidity (COR=1.78 95% CI (1.01, 3.15), p=0.049), prolonged antibiotic days (COR=2.18 95% CI (1.18, 4.03), p=0.013) and prolonged agent days (COR=3.01 95% CI (1.55, 5.83), p=0.001) were positively associated with prolonged antibiotic days in the univariate model, however, this association was not seen in the multivariate analysis. Only presence of MDR risk (AOR=6.14, 95% CI: 1.68, 22.41, p=0.006) was positively associated with prolonged LoS in the multivariate model (Table 7).
Table 7. Binary logistic regression analysis for hospitalized patients with bacterial infection in the internal medicine ward of Nekmte referral hospital in 2017, Nekemte, Ethiopia.
| Variables | LoS (>7 days) | COR (95% CI) | AOR (95% CI) |
|---|---|---|---|
| Human Immunodeficiency Virus(Yes) | 13 (68.4) | 2.73 (0.99, 7.51) | 0.55 (0.06,5.30) |
| Comorbidity (Yes) | 53 (53.5) | 1.78 (1.01, 3.15)* | 0.85 (0.27, 2.68) |
| Ceftriaxone use (yes) | 84 (45.7) | 0.42 (0.10, 1.73 | 0.52 (0.03, 10.99) |
| Chloramphenicol use (yes) | 6 (66.7) | 2.38 (0.58, 9.81) | 0.59 (0.03, 11.81) |
| Prolonged antibiotic days (>7 days) | 37 (59.7) | 2.18 (1.18, 4.03)* | 0.96 (0.18, 5.14) |
| Prolonged agent Days (>10 days) | 35 (66.0) | 3.01 (1.55, 5.83)** | 0.26 (0.03, 2.01) |
| MDR risk (Absent) | 54 (43.9) | (1.00) | (1.00) |
| MDR risk (unknown) | 25 (56.8) | 0.94 (0.40, 2.21) | 0.64 (0.12, 3.45) |
| MDR risk (present) | 11 (42.3) | 1.68 (0.84, 3.37) | 6.14 (1.68, 22.41) |
| One antibiotic Courses | 50 (44.6) | (1.00) | (1.00) |
| Two antibiotic courses | 23 (39.0) | 0.79 (0.42, 1.51) | 0.98 (0.23, 4.10) |
| Three and above antibiotic courses | 17 (77.3) | 4.22 (1.45, 12.22)** | 2.60 (0.22, 30.99) |
*p < 0.05; **p< 0.01; ***p<0.001; MDR risk: multidrug resistant infection risk; COR: crud odds ratio; AOR: Adjusted odds ratio; CI: confidence interval
Discussion
Different from Storey et al. [16] and similar to Alemkere et al. [14], in the present study, infection was the primary diagnosis for admission followed by circulatory disorders. Consistent with different reports [14,16,17] pneumonia was the most common infection in hospitalized patients. In line to Alemkere et al. [14] and Hall et al. [18], majority (72.8%) of the pneumonia cases were community acquired. Similar to the study conducted in TASH [14] and most studies conducted in regional hospitals of Ethiopia [19,20], cephalosporins were the most commonly used drugs. Comparable to previous studies conducted in TASH [21,22], empiric therapy was initiated almost in all patients. Including the current, all these studies conducted in Ethiopia were in disagreement with a study performed in one teaching hospital [23], where empiric therapy was initiated only in 19.4% of the patients. Antibiotics ordered empirically were found to be less appropriate than those ordered with evidence of culture and susceptibility report [24]. In the current study, however, none of the antibiotic uses were justified by microbiologic cultures.
One of the important issues addressed in this study was the need assessment performed in line with hospital outcome indicators. Being one of the outcome indicators, prolonged LoS was enormously associated with higher hospital costs [25]. The mean LoS reported for the medical wards in this study was more than two times lower than the TASH hospital study [14] and about two times higher than reported by Usman et al. (2011) and Ghods et al. [26,27] for the general patients. On the other hand it is relatively similar with Battleman et al. and Suter-Widmer et al. [28,29] report for pneumonia cases.
A study conducted among pneumococcal bacteremia patients in 21 hospitals in 10 countries (including developed and developing countries) reported a mortality rate of 16.9% [30]. This was higher than the ward mortality found in the current study (8.29%). In addition the mortality in our hospital is also about three times lower than the mortality reported in TASH (27.7%) [14]. Not as such different from ours, another study conducted in a Gambian hospital [31] reported an overall mortality of 6% and bacteremia attributed mortality of 8.3%. All these collectively indicate that the mortality in the current study was comparably not bad. In the current study presence of signs and symptoms of disease (COR=4.57, 95% CI (1.41, 14.82), p=0.011), was associated with mortality in the univariate analysis but not in the multivariate. However, studies by Alemkere et al. (2014), Gordon (2003) and Diekema et al. [14,32,33] found an independent association with mortality.
Similar to a study conducted in Tikur Anbesa Specialized Hospital [14] vancomycin use positively associated with mortality in the univariate models (COR=3.33, 95% CI (1.12, 9.93), p=0.031) but not in the mutlivariate models. Although no studies were found with similar methodological approach in support of these evidences, from other counters, this could possibly be explained by the inappropriate use of the drug in the Ethiopian hospitals. In support of this, vancomycin use evaluation conducted in the internal medicine ward of TASH revealed that vancomycin dose was not adjusted or adjusted inadequately in 96.5% of the cases [34]. The absence of association in the multivariable models of both studies, however, weakness our argument. Therefore, this deserves a future study with advanced methodological designs.
Similar to van der Kooi et al. [35] presence of a medical device is a strong independent predictor of mortality (AOR=4.50 (1.09, 18.60) p=0.038). On the other hand prolonged length of stay is the negative predictor of mortality both in the univariate (COR=0.24, 95% CI (0.07, 0.87), p=0.029) and multivariate models (AOR=0.22 95% CI (0.05, 0.90) p=0.035). However, the TASH study [14] reveal an association only in the univariable model (AOR=0.53, 95% CI (0.31, 0.91)).
In contrast with an observational study by De Buyser et al. [36] primary admission diagnoses and consecutive infection diagnosis in the multivariate analysis have no association with prolonged LoS. However, only the presence of MDR risk is associated with prolonged length of stay (AOR=6.14, 95% CI: 1.68, 22.41, p=0.006). Unlike the current study, Diekema et al. and Zilberberg et al. [33,37] reported association of nosocomial origin of infection with mortality. Nevertheless, the current study is in agreement with [37] the statistically significant association found between patients having MDR risk infections and prolonged LoS (AOR=6.14, 95% CI: 1.68, 22.41, p=0.006). In agreement with our study, the study performed by Lye et al. [38] also reported an independent association of MDR risk with prolonged LoS in survivors. A similar study Lye et al. [38] reported the association of MDR with 30-day mortality, despite the absence of such evidence in the multivariate analysis.
Hence, given the cost of combination therapy, guidelines restrict such treatment approaches for certain group of patients [39] and recommend prompt de-escalation based on the patient’s clinical course, and culture & susceptibility test results [39,40]. Despite this concept and Mettler et al. report [23], though all our patients started with empiric therapies, modification was done only for 5% of the cases. Even these modifications didn’t necessarily indicate streamlining (lowering the estimate), since majority of the modifications involved addition of therapy.
The two quality indicators, concordance to the guideline and intravenous to oral switch, were tested for statistical association. Guideline concordant antibacterial use was negatively associated (COR=0.25 95% CI (0.09, 0.73), p=0.011) with mortality in the univariate analysis but not in the multivariate regression model, however, intravenous to oral switch was found to be associated neither in the univariate nor in the multivariate model with both outcome indicators.
Despite attempts were made to fill the information gaps in the charts through seeking assistance from physicians on the spot, being an observational study, however, it won’t be free of limitations. The study was conducted in only one hospital, and practice patterns, patients’ characteristics and microbiology resistance patterns may vary among hospitals, which may limit its generalizability. Although the use of multivariate analysis helped to control a substantial proportion of any confounding variable, some unaddressed factors might have affected the outcomes.
In conclusion, this observation showed that all patients with systemic bacterial infection received antibiotics on an empiric basis in which none were justified based on microbiologic cultures. Pulmonary infections were the most frequent type of infections and cephalosporins were the most commonly used drugs. Local guidelines or any other stewardship activities should give priority to these issues. Therefore, in view of the limited number of publications on this topic, the results should be of interest to clinicians and other concerned stakeholders in the field. Furthermore, future researchers in the hospital should better focus on modifiable risk factors and use advanced methodological options to prove real association.
Competing Interests
The authors declare that they have no competing interests.
Authors’ Contributions
GA conducted the data collection and analysis, and drafted the manuscript. GA, GB and AB participated in the design of the study, coordination and revised the manuscript. All authors read and approved the final manuscript.
Acknowledgments
We thank the data collectors of the research, the health care works and the patients during the data collection and Wollega University, College of Health Science, Department of Pharmacy for their respective cooperation.
Funding
This research received incentive from Wollega University.
References
- Gottlieb T, Nimmo GR. Antibiotic resistance is an emerging threat to public health: an urgent call to action at the Antimicrobial Resistance Summit 2011. Med J Aust. 2011;194(6):281-3.
- Feyissa M, Gedif T, Azajh A (2013) Medication prescribing errors in emergency department medical wing of TASH. Addis Ababa University.
- Gashaw S, Nedi T, Tadesse Y (2013) Assessment of Medication prescribing and administration errors involving patients in the internal medicine wards of TASH. Addis Ababa University.
- Roberts R, Hota B, Ahmad I, et al. Hospital and societal costs of antimicrobial-resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship. Clin Infect Dis. 2009;49(8):1175-84.
- Gupta N, Limbago BM, Patel JB, et al. Carbapenem-resistant enterobacteriaceae: epidemiology and prevention. Clin Infect Dis. 2011;53(1):60-7.
- De Kracker ME, Davey PG, Grundmann H. Mortality and hospital stay associated with resistant Staphylococcus aureus and Escherichia coli bacteremia: estimating the burden of antibiotic resistance in Europe. PLoS Med. 2011;8(10):e1001104.
- Powers JH. Increasing the efficiency of clinical trials of antimicrobials: the scientific basis of substantial evidence of effectiveness of drugs. Clin Infect Dis. 2007;45(Suppl 2):S135-62.
- Fonseca MJ, Santos CL, Costa P, et al. Increasing Awareness about Antibiotic Use and Resistance: A Hands-On Project for High School Students. PLoS ONE. 2012;7(9):e44699.
- Lee CR, Cho IH, Jeong BC, et al. Strategies to Minimize Antibiotic Resistance. Int J Environ Res Public Health. 2013;10(9):4274-305.
- O’Brien TF. The global epidemic nature of antimicrobial resistance and the need to monitor and manage it locally. Clin Infect Dis. 1997;24(Suppl 1):S2-8.
- O’Brien TF. Emergence, spread, and environmental effect of antimicrobial resistance: how use of an antimicrobial anywhere can increase resistance to any antimicrobial anywhere else. Clin Infect Dis. 2002;34(Suppl 3):S78-84.
- Yismaw G, Negeri N, Kassu A. A five-year antimicrobial resistance pattern observed in Shigella species isolated from stool samples in Gondar University Hospital, northwest Ethiopia. Ethiop J Health Dev. 2006;20(3):194-8.
- Biadglegne F, Abera B. Antimicrobial resistance of bacterial isolates from urinary tract infections at Felge Hiwot Referral Hospital, Ethiopia. Ethiop J Health Dev. 2009;23(3):236-8.
- Alemkere G, Tenna A, Engidawork E. Antibiotic Use Practice and Predictors of Hospital Outcome among Patients with Systemic Bacterial Infection at Tikur Anbessa Specialized Hospital: Identifying Targets for Antibiotic and Health Care Resource Stewardship. PLOS ONE, 2017.
- Berrington A. Antimicrobial prescribing in hospitals: be careful what you measure. J Antimicrob Chemother. 2010;65(1):163-8.
- Storey DF, Pate PG, Nguyen AT, et al. Implementation of an antimicrobial stewardship program on the medical-surgical service of a 100bed community hospital. Antimicrob Resist Infect Control. 2012;1(1):32.
- Camins BC, King MD, Wells JB, et al. Impact of an antimicrobial utilization program on antimicrobial use at a large teaching hospital: A randomized controlled trial. Infect Control Hosp Epidemiol. 2009;30(10):931-8.
- Hall MJ, DeFrances CJ, Williams SN, et al. National Hospital Discharge Survey: (2007) summary. Natl Health Stat Report. 2010;(29):1-20.
- Desta Z, Abula T, Yohannes AG, et al. Drug prescribing patterns for outpatients in three hospitals in north-west Ethiopia. Ethiop J Health Dev. 2002;16(2):183-9.
- Getachew E, Aragaw S, Adissie W, et al. Antibiotic prescribing pattern in a referral hospital in Ethiopia’. Afr J Pharm Pharmacol. 2013;7(38):2657-61.
- Ayele F, Engidawork E, Seboxa T (2013) Drug use evaluation of third generation cephalosporins in the internal medicine wards of Tkur Anbesa Specializd Hospital. Addis Ababa University.
- Sileshi A, Shibeshi W, Tenna A (2015) Evaluation of ceftriaxone utilization in the medical and emergency wards of TASH. Addis Ababa University.
- Mettler J, Simcock M, Sendi P, et al. Empirical use of antibiotics and adjustment of empirical antibiotic therapies in a university hospital: a prospective observational study. BMC Infect Dis. 2007;7:21.
- Erbay A, Bodur H, Akinci E, et al. Evaluation of antibiotic use in intensive care units of a tertiary care hospital in Turkey. J Hosp Infect. 2005;59(1):53-61.
- Mauldin PD, Salgado CD, Hansen IS, et al. Attributable hospital cost and length of stay associated with health care-associated infections caused by antibiotic-resistant gram-negative bacteria. Antimicrob Agents Chemother. 2010;54(1):109-15.
- Usman M, Jamal S, Tabassum S, et al. Length of stay of patients in tertiary care hospital. Gomal Journal of Medical Sciences. 2011;9(1):55-8.
- Ghods AA, Khabiri R, Raeisdana N, et al. Predictors of Inappropriate Hospital Stay: Experience from Iran. Glob J Health Sci. 2015;7(3):82-9.
- Battleman DS, Callahan M, Thaler HT. Rapid antibiotic delivery and appropriate selection reduces length of hospital stay of patents with community acquired pneumonia. Arch Intern Med. 2002;162(6): 682-8.
- Suter-Widmer I, Christ-Crain M, Zimmerli W, et al. Predictors for length of hospital stay in patients with community-acquired Pneumonia: Results from a Swiss Multicenter study. BMC Pulm Med. 2012;12:21.
- Yu VL, Chiou CC, Feldman C, et al. An International Prospective Study of Pneumococcal Bacteremia: Correlation with In Vitro Resistance, Antibiotics Administered, and Clinical Outcome. Clin Infect Dis. 2003;37(2):230-7.
- Hill PC, Onyeama CO, Ikumapayi UNA, et al. Bacteraemia in patients admitted to an urban hospital in West Africa. BMC Infect Dis. 2007;7:2.
- Gordon SB. Patient outcome in adults with pneumococcal meningitis or bacteremia admitted to Queen Elisabeth Central Hospital. Malawi Med. 2003;15(2):38-42.
- Diekema DJ, Beekmann SE, Chapin KC, et al. Epidemiology and outcome of nosocomial and community-onset bloodstream infection. J Clin Microbiol. 2003;41(8):3655-60.
- Zeleke B, Ephrem E, Nedi T (2013) Drug utilization review of vancomycin among hospitalized patients in the internal medicine wards TASH. Addis Ababa University.
- van der Kooi TI, de Boer AS, Manniën J, et al. Incidence and risk factors of device-associated infections and associated mortality at the intensive care in the Dutch surveillance system. Intensive Care Med. 2007;33(2):271-8.
- De Buyser SL, Petrovic M, Taes YE, et al. A Multicomponent Approach to Identify Predictors of Hospital Outcomes in Older In-Patients: A Multicentre, Observational Study. PLoS ONE. 2014;9(12):e115413.
- Zilberberg Shorr AF, Micek ST, Hoban AP. Epidemiology and Outcomes of Hospitalizations with Complicated Skin and Skin-Structure Infections: Infection Risk Factors. Infect Control Hosp Epidemiol. 2009;30(12):1203-10.
- Lye DC, Earnest A, Ling M-L, et al. The impact of multidrug resistance in healthcare-associated and nosocomial Gram-negative bacteraemia on mortality and length of stay: cohort study. Clin Microbiol Infect. 2012;18(5):502-8.
- Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580-637.
- Marcus R, Paul M, Elphick H, et al. Clinical implications of beta-lactam-aminoglycoside synergism: systematic review of randomised trials. Int J Antimicrob Agents. 2011;37(6):491-503.