Research Article - Biomedical Research (2017) Volume 28, Issue 9
Nonmetrical sexual dimorphism in mandibles of Brazilian individuals
Naira F. Deana1 and Nilton Alves2,3*1Máster Program in Dentistry, Faculty of Dentistry, Universidad de La Frontera, Temuco, Chile
2Department of Integral Dentistry, Faculty of Dentistry, Universidad de La Frontera, Temuco, Chile
3Applied Morphology Research Centre (CIMA), Faculty of Dentistry, Universidad de La Frontera, Temuco, Chile
- *Corresponding Author:
- Nilton Alves
Department of Integral Dentistry
Faculty of Dentistry
Universidad de La Frontera
Chile
Accepted date: March 6, 2017
Abstract
The sexual differences can be determined through morphological characteristics found in the ramus and body of mandible. The accuracy in determining sex is variable and the degree of sexual dimorphism in mandibles is different for each population. The aim of this study was to analyse nonmetric morphological characteristics of mandibles in adult Brazilian individuals, determining the anatomical variations found. In this study, 128 macerated mandibles of black and white Brazilian adults of both sexes were used. Of the 7 indicators analysed, the morphological characteristic that best distinguished the sexes was the shape of the chin, with an accuracy of 82.7% in black males, 61.9% in white males, 73.0% in black females and 75.0% in white females. The divergence of the gonial angle presented statistically significant differences between black males and black females, with an accuracy of 93.1% in black males and 34.3% in black females. An analysis of the shape of the chin is recommended as a reliable morphological indicator for the mandible in the diagnosis of sex from the skeletal remains of unknown individuals.
Keywords
Forensic science, Forensic odontology, Sex determination, Nonmetric characteristics, Mandibles
Introduction
The mandible is a symmetrical bone that forms the lower third of the face [1]. It is one of the most durable and dimorphic skull bones [2]. It is horseshoe-shaped with two rami that come away from the posterior extremities of the body of mandible in the direction of the cranium. It is the insertion site of several muscles, such as the masseter, temporal, lateral pterygoid and medial pterygoid [1].
Many studies have shown that the mandible is very important for identifying sex and that sexual differences can be determined through morphological characteristics found in the ramus and body of mandible. Loth and Henneberg [3] established sexual differences by analysing mandibular ramus flexure, finding 99.1% accuracy for males and 98.8% for females. Other authors analysed gonial eversion, finding 75.4% accuracy in determining male mandibles and 45.2% for female mandibles [4]. For Hu et al. [2], the simultaneous use of two characteristics of the mandible-the shape of the base of mandible and the shape of the chin-is the best method for predicting sex, to accuracy over 90%.
It is important to note that accuracy in determining sex is variable and the degree of sexual dimorphism in mandibles is different for each population [5]. Genetic, external and environmental factors such as climate, nutrition and social class can influence the mandibular morphology [6-9].
The aim of this study was to analyse nonmetric morphological characteristics of mandibles in black and white adult Brazilians of both sexes, determining the anatomical variations found.
Methods
Sample
One hundred twenty-eight macerated mandibles of Brazilian black and white adult individuals of both sexes belonging to the Department of Morphology and Genetics, UNIFESP (Brazil) were used in this study. All the mandibles had information regarding the country of origin, sex and skin color. The sample was divided into 56 females, 18 white (mean age: 39.22 years), 38 black (mean age: 37.52 years), and 72 males, 28 white (mean age: 40.03 years) and 44 black (mean age: 36.54 years).
Edentulous mandibles and/or those that did not contain information on sex and race were excluded from this study. We included only wholly or partially dentate hemimandibles that had at least one molar in the hemiarcade being studied.
Procedure
When analysing the mandibles, the examiner did not know the sex of the mandible reported in the corresponding identification, making this a blind study. For the sexual determination of the mandibles, seven nonmetric characteristics were selected that were examined by a previously calibrated investigator. Morphological characteristics 1-6 were analysed according to the proposal by Hu et al. [2] (modified), and characteristic 7 was analysed according to the classification of Loth and Henneberg [3]:
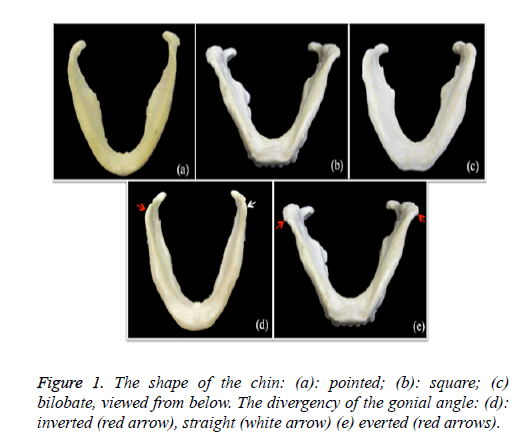
1. The shape of the chin (Figures 1a-1c): pointed, square or bilobate. This was analysed from inferior and anterior views.
2. The divergence of the gonial angle (Figures 1d and 1e): inverted, everted or straight. The angle of mandible was examined as to whether it was bent inward or outward.
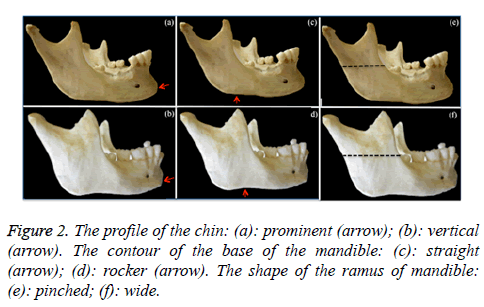
3. The profile of the chin (Figures 2a and 2b): vertical or prominent. This was analysed according to the protrusion, observed from a lateral view.
4. The contour of the base of mandible (Figures 2c and 2d): straight, undulating or rocker. The base of mandible was analysed according to the depth of the antegonial notch.
5. The shape of the ramus of mandible (Figures 2e and 2f): pinched or wide. This was analysed at its midportion.
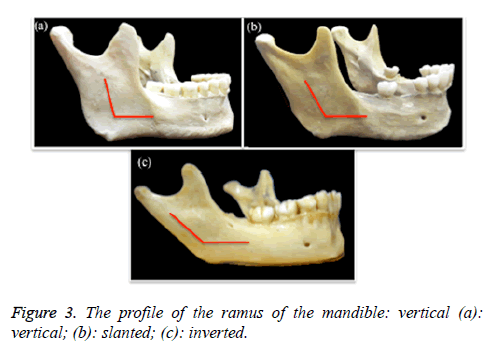
6. The profile of the ramus of mandible (Figure 3): vertical, inverted, or slanted. This was analysed according to the curvature of the ramus against the body of mandible.
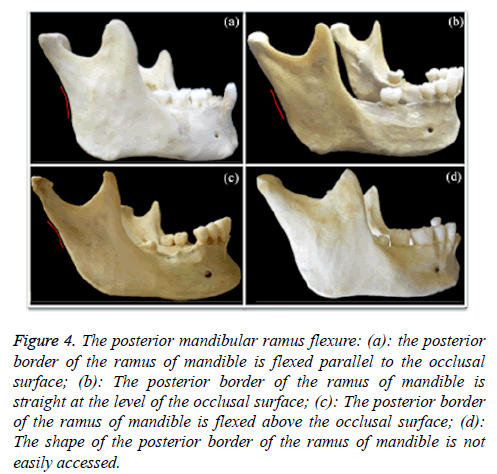
7. The posterior mandibular ramus flexure (Figure 4): A score of +1 was assigned if flexure was present, -1 for a straight border, and 0 if the shape was neither clearly flexed nor straight. The scores for both rami totalled +2, +1, 0, -1 and -2. Those with a score -1 and -2 were considered female mandibles, and male mandibles obtained a score of 0, +1 and +2. The score was compared to the sex recorded in the identification of the mandibles and prediction accuracy was calculated. The right and left sides of each ramus of mandible were analysed separately.
Figure 4: The posterior mandibular ramus flexure: (a): the posterior border of the ramus of mandible is flexed parallel to the occlusal surface; (b): The posterior border of the ramus of mandible is straight at the level of the occlusal surface; (c): The posterior border of the ramus of mandible is flexed above the occlusal surface; (d): The shape of the posterior border of the ramus of mandible is not easily accessed.
Statistical analysis
For the statistical analysis, the significance tests were performed by either calculating random error to build confidence intervals of percentage frequencies or by the Chi-squared test, with P ≤ 0.05 being considered significant.
Results
The shape of the chin
The shape of the chin presented significant statistical differences between the sexes for black individuals (P=0.0001) and for white individuals (P=0.05). The square shape was more frequent in males and the pointed form more frequent in females. The bilobate shape presented similar percentages for black males, black females and white females; the white males had the lowest percentage (Table 1). The accuracy in determining sex using this morphological indicator was 82.7% in black males, 61.9% in white males, 73.0% in black females and 75.0% in white females.
| Classification of nonmetric items | Types | Black Males | White Males | Black Females | White Females |
|---|---|---|---|---|---|
| 1. The shape of the chin | Bilobate | 34.00% | 22.30% | 31.60% | 33.30% |
| Square | 54.60% | 48.10% | 18.40% | 16.70% | |
| Pointed | 11.40% | 29.60% | 50% | 50.00% | |
| 2. The divergence of the gonial angle | Inverted | 4.60% | 3.70% | 28.90% | 22.10% |
| Straight | 34.00% | 22.30% | 15.90% | 16.70% | |
| Everted | 61.40% | 74.00% | 55.20% | 61.20% | |
| 3. The profile of the chin | Vertical | 52.30% | 37.00% | 52.60% | 33.30% |
| Prominent | 47.70% | 63.00% | 47.40% | 66.70% | |
| 4. The contour of the base of mandible | Straight | 25.00% | 11.10% | 21.10% | 16.60% |
| Rocker | 75.00% | 88.90% | 78.90% | 83.30% | |
| Undulating | 0.00% | 0.00% | 0.00% | 0.00% | |
| 5. The shape of the ramus of mandible | Pinched | 34.10% | 44.50% | 26.30% | 44.50% |
| Wide | 65.90% | 55.50% | 73.70% | 55.50% | |
| 6. The profile of the ramus of mandible | Vertical | 22.70% | 37.00% | 31.60% | 5.60% |
| Slanted | 77.30% | 63.00% | 68.40% | 94.40% | |
| Inverted | 0.00% | 0.00% | 0.00% | 0.00% |
Table 1: Comparison of the 6 nonmetric characteristics between males and females, black and white Brazilian individuals.
The divergence of the gonial angle
The divergence of the gonial angle presented a statistically significant difference between black males and females (P=0.01). In white individuals the males presented the everted shape more frequently than the females, and the females presented the inverted shape more frequently than the males; however, no statistically significant difference was observed (P=0.13) (Table 1). The accuracy in determining sex using this morphological indicator was 93.1% in black males, 95.2% in white males, 34.3% in black females and 26.6% in white females.
The profile of the chin
In black individuals of both sexes the vertical shape of the chin profile was slightly more frequent than the prominent shape. In white individuals of both sexes the prominent shape of the chin profile was considerably more frequent than the vertical shape. In the statistical analysis no differences were observed between sexes or races (P=0.19 and P=0.13, respectively) (Table 1).
The contour of the base of mandible
The undulating shape of the contour of the base of mandible was not found in any case. White and black individuals of both sexes presented the rocker shape significantly more frequently than the straight shape (Table 1). No statistical differences were observed for this morphological indicator between the sexes.
The shape of the ramus of mandible
The pinched shape of the ramus was less frequent than the wide type in white and black individuals of both sexes, particularly in black females (Table 1). No statistical differences were observed between the sexes for this morphological indicator.
The profile of the ramus of mandible
The profile of the slanted type of ramus was more frequent in both black and white individuals of both sexes, especially in white females where it was observed that only 5.6% of the mandibles were vertical. A statistically significant different was found between white males and white females (P=0.03) and between white and black females (P=0.04). No case of a profile of an inverted ramus of mandible was found (Table 1).
The posterior mandibular ramus flexure
The predictive accuracy for the posterior mandibular ramus flexure in males was greater than in females for both black and white individuals, with black females presenting the lowest accuracy (Table 2). The accuracy found for white individuals was slightly lower than that found for blacks. 65.9% of the black males and 64.0% of the white males exhibited bilateral flexure (+2), and 3.2% of the black females and 11.7% of the white females presented a straight ramus bilaterally. The distribution of ramus shape scores by sex and skin color is presented in Table 3. The bilateral mandibular ramus flexure was the most frequent shape in our study with 57.3%. No statistical difference was found between sexes for white or black individuals.
| Sex | N | Scores | Accuracy | |||
|---|---|---|---|---|---|---|
| -1, and -2 | 0, +1 and +2 | |||||
| N | % | N | % | |||
| Black individuals | ||||||
| Males | 44 | 7 | 50 | 37 | 55.2 | 84.00% |
| Females | 31 | 7 | 50 | 24 | 44.8 | 22.50% |
| Total | 82 | 14 | 100 | 61 | 100 | 58.60% |
| p-value 0.67 | ||||||
| White individuals | ||||||
| Males | 24 | 8 | 53.4 | 17 | 63.4 | 68.00% |
| Females | 17 | 7 | 46.6 | 10 | 36.6 | 41.20% |
| Total | 45 | 15 | 100 | 30 | 100 | 57.10% |
| p-value 0.77 | ||||||
| Note: -1 and -2 are female, and 0, +1, and +2 are male | ||||||
Table 2: Overall accuracy of ramus shape as an indicator of sex, according to skin color.
| Sample | 2 | 1 | 0 | -1 | -2 |
|---|---|---|---|---|---|
| White males | 64.00% | 4.00% | 0.00% | 32.00% | 0.00% |
| Black males | 65.90% | 15.90% | 2.30% | 15.90% | 0.00% |
| White females | 23.50% | 17.70% | 17.70% | 29.40% | 11.70% |
| Black females | 58.00% | 12.90% | 6.50% | 19.40% | 3.20% |
| Total | 57.30% | 12.80% | 5.20% | 22.20% | 2.50% |
Table 3: Distribution of ramus shape scores by sex and skin color of Brazilian sample.
Discussion
The sexual dimorphism of the mandible is characterized mainly by the size, which is specific to each population [7]. Males and females can be distinguished based on the shapes of various parts of the mandible [2]. Generally females produce less muscle force, so the muscle attachments have smoother surfaces. Males produce greater muscle force, resulting in rough surfaces on muscle attachments, particularly in the coronoid process and the gonion [10,11].
The shape of the chin is a nonmetric characteristic used to distinguish the sexes, and in general it is bilobate or square in males and pointed in females [2]; in our study we found important differences between the sexes for this characteristic, corroborating previous findings reported by Hu et al. [2] and Bass [12]. In the present study, the shape of the chin was square in 54.6% of black males and 48.1% of white males, in black and white females it was pointed in 50% of the mandibles. The bilobate shape obtained similar percentages for white and black individuals of both sexes. Hu et al. [2] reported 54.4% were pointed in females and 58.3% were square in males. In our study the accuracy to predict sex using this morphological characteristic was greater in males than females and ranged between 61.9 and 82.7%, corroborating the findings reported by Williams and Rogers [13] who also found better accuracy in males (88%) than in females (72%).
Another frequently used nonmetric characteristic to distinguish sex is the contour of the base of mandible, being rocker-shaped in males and straight in females [2]. In our study we failed to validate this characteristic to differentiate the sexes since we found the majority were rocker-shaped in both sexes.
In the present study, eversion of the gonial angle was decisive for sexual differentiation only in black individuals. The accuracy in determining the male sex was quite high, 93.1%, whereas for females it was quite low, only 34.3%; Williams and Rogers [13] found 64% accuracy for males and 52% for females. Loth and Henneberg [14] described eversion of the gonial angle as being a characteristic of the facial architecture that can be traced back to the selective robustness of the masseter muscle rather than of sexual dimorphism. Hu et al. [2] indicated that the everted gonial angle is the most divergent characteristic in both sexes. Given the poor accuracy in determining the female sex and having diagnosed the sex only in black individuals, we considered this an unreliable indicator for sexual determination in the mandibles of Brazilians.
The slanted profile of the ramus of mandible was significantly more frequent than the vertical type for all the individuals in the sample, particularly for white females, where only 5.6% were observed to be vertical.
For the morphological characteristics shape of the ramus of mandible and profile of the chin, we found no statistically significant differences, a situation also reported by Hu et al. [2]; therefore, we considered these indicators to be unreliable in diagnosing the sex.
The presence or absence of mandibular ramus flexure is considered a valuable tool for identifying the sex in both modern forensic cases and in mandibles found in historic archaeological and rare fossil hominids [2]. Saini et al. [15] also showed that this method can be used to diagnose sex with an average accuracy of up to 82%, a value close to that reported by Haun [16] with 78.2%. The accuracy of this method has been reported by several authors, reaching 94.2% in blacks, whites and Amerindians [3], 58.8% in the German population [4], 62.5% in American blacks, American whites and Amerindians [17], 63.25% in Chilean females and 48.25% in Chilean males [18], 83.9% in the modern Egyptian population [19], 79.1% in European-Americans and African- Americans [20], 85.8% in the Turkish population [21], 95.2% in Jordanian females and 77.8% in Jordanian males [22]; in our study we obtained an overall accuracy of 58.6% for black individuals 57.1% for white individuals, average accuracy values close to those reported for the German and Chilean populations, black and white Americans and Amerindians [4,17,18] and considerably lower than those reported by Loth and Henneberg [3], Saini et al. [15], Hill [20], Balci et al. [21] and Badran et al. [22]. In our study we excluded the mandibles that presented a loss of more than 2 molars because excessive tooth loss makes it difficult to analyse the position of the flexure and consequently undermines determination of the sex. We agree with Donnelly et al. [17] and Hill [20] when they point out that a complication of this indicator for sexual determination is that many mandibles are asymmetrical, i.e., one side is classified as male and the other as female, which raises ambiguity when establishing the sex. According to Loth and Henneberg [3] these mandibles are classified as male; however, their correct classification would be “unable to sex” [20]. Additionally, flexure is more likely to be identified than its absence, leading to a consistent misidentification of females [17,20]. In a study on Koreans, Hu et al. [2] found no difference in flexure between the sexes; nevertheless, it is worth noting that these authors used a different method than we did to analyse the mandibular ramus flexure. Some authors conclude that the use of the ramus flexure provides a moderately acceptable predictive accuracy and can be considered supplementary for diagnosing the sex [22]. We agree with Donnelly et al. [17] and Hill [20] who showed that due to low overall accuracy, high intraobserver error and an invalid scoring system, the mandibular ramus flexure is an unreliable indicator for estimating sex.
Kemkes-Grottenthaler et al. [4] described the estimation of sex as being seriously undermined by observer error, given the difficulty in identifying the location and degree of both mandibular ramus flexure and gonial eversion; this could explain the existence of conflicting scientific evidence.
Some researchers consider the mandible an excellent indicator for sexual determination [2]. However, there are reports that relate low accuracy using the mandible to diagnose sex. In a study to determine the sex using the skull (cranium + mandible) compared to the pelvis through non-metrical morphological sex-discriminant features, Matt et al. [23] concluded that sex diagnosis using the mandible corresponded to using the pelvis in 69.5%. In the female pelvis, 51.6% of the related mandibles were diagnosed as ‘male’.
In our study the mandibles from some females presented morphological indicators considered male according to the literature due to the ramus flexure and contour of the base of mandible, which made it impossible to validate these indicators to diagnose the sex. In African populations, mandibular sexual dimorphism is rarely pronounced because this population suffers from malnutrition, which can affect pubertal development in males, where a greater degree of hormonal control is found than in females; however, if the conditions are favorable, the mandible develops normally, demonstrating well-pronounced traits [24]. It is important to note that the magnitude and pattern of sexual dimorphism in human mandibles depend on the circumstances of a specific population, being associated with genetic background and environmental conditions [5] including diet, the effects associated with masticatory stress, medical attention and oral hygiene [5,25,26].
Conclusions
In the present study the morphological characteristic that best distinguished the sexes was the shape of the chin. We consider this morphological characteristic a reliable indicator to determine sex in the mandibles of white and black Brazilian individuals. Nevertheless, it is important to use other resources to aid in forensic identification when diagnosing the sex from skeletal remains of unknown individuals.
Acknowledgements
Department of Morphology and Genetics, UNIFESP, Sao Paulo, Brazil.
Conflicts of Interest
The authors declare they have no conflicts of interest.
References
- Alves N, Candido PL. Anatomía para o curso de odontología geral e específica. Santos (4th edn.) 2016.
- Hu KS, Koh KS, Han SH, Shin KJ, Kim HJ. Sex determination using nonmetric characteristics of the mandible in Koreans. J Forensic Sci 2006; 51: 1376-1382.
- Loth SR, Henneberg M. Mandibular ramus flexure: a new morphologic indicator of sexual dimorphism in the human skeleton. Am J Phys Anthropol 1996; 99: 473-485.
- Kemkes-Grottenthaler A, Lobig F, Stock F. Mandibular ramus flexure and gonial eversion as morphologic indicators of sex. Homo 2002; 53: 97-111.
- Bejdova S, Krajicek V, Veleminska J, Horak M, Veleminsky P. Changes in the sexual dimorphism of the human mandible during the last 1200 years in Central Europe. HOMO 2013; 64: 437-453.
- Nicholson E, Harvati K. Quantitative analysis of human mandibular shape using three-dimensional geometric morphometrics. Am J Phys Anthropol 2006; 131: 368-383.
- Smith HF. Which cranial regions reflect molecular distances reliably in humans? Evidence from three-dimensional morphology. Am J Hum Biol 2009; 21: 36-47.
- Thayer ZM, Dobson SD. Sexual dimorphism in chin shape: implications for adaptive hypotheses. Am J Phys Anthropol 2010; 143: 417-425.
- Ulijaszek SJ, Mann N, Elton S. Evolving human nutrition: implication for public health. Cambridge University Press 2012.
- Rosas A, Bastir M, Martinez-Maza C, Bermudez de Castro JM. Sexual dimorphism in the Atapuerca-SH hominids: the evidence from the mandibles. J Hum Evol 2002; 42: 451-474.
- Vodanovic M, Dumancic J, Demo Z, Mihelic D. Determination of sex by discriminant function analysis of mandibles from two Croatian archaeological sites. Acta Stomatol Croatica 2006; 40: 263-277.
- Bass WM. Human osteology. Missouri Archaeological Society Press (3rd Edn.) 1987.
- Williams BA, Rogers T. Evaluating the accuracy and precision of cranial morphological traits for sex determination. J Forensic Sci 2006; 51: 729-735.
- Loth SR, Henneberg M. Gonial eversion: facial architecture, not sex. HOMO 2000; 51: 81-89.
- Saini V, Srivastava R, Shamal SN, Singh TB, Pandey AK, Tripathi SK. Sex determination using mandibular ramus flexure: a preliminary study on Indian population. J Forensic Leg Med 2011; 18: 208-212.
- Haun SJ. Brief Communication: A study of the predictive accuracy of mandibular ramus flexure as a singular morphologic indicator of sex in an archaeological sample. Am J Phys Anthropol 2000; 111: 429-432.
- Donnelly SM, Hens SM, Rogers NL, Schneider KL. Technical note: a blind test of mandibular ramus flexure as a morphological indicator of sexual dimorphism in the human skeleton. Am J Phys Anthropol 1998; 107: 363-366.
- Suazo GIC, San Pedro VJ, Schilling QNA, Celis CCE, Hidalgo RJA, Cantín LM. Ortopantomographic blind test of mandibular ramus flexure as a morphological indicator of sex in Chilean young adults. Int J Morphol 2008; 26: 89-92.
- Kharoshah MAA, Almadani O, Ghaleb SS, Zaki MK, Fattah YAA. Sexual dimorphism of the mandible in a modern Egyptian population. J Forensic Leg Med 2010; 17: 213-215.
- Hill CA. Technical note: evaluating mandibular ramus flexure as a morphological indicator of sex. Am J Phys Anthropol 2000; 111: 573-577.
- Balci Y, Yavuz MF, Caazdir S. Predictive accuracy of sexing the mandible by ramus flexure. Homo 2005; 55: 229-237.
- Badran DH, Othman DA, Thnaibat HW, Amin WM. Predictive accuracy of mandibular ramus flexure as a morphologic indicator of sex dimorphism in Jordanians. Int J Morphol 2015; 33: 1248-1254.
- Maat GJR, Mastwijk RW, Van der Velde EA. On the reliability of non-metrical morphological sex determination of the skull compared with that of the pelvis in the low countries. Int J Osteoarchaeol 1997; 7: 575-580.
- Oettle AC, Pretorius E, Steyn M. Geometric morphometric analysis of the use of mandibular gonial eversion in sex determination. Homo 2009; 60: 29-43.
- Kaifu Y. Changes in mandibular morphology from the Jomon to modern periods in eastern Japan. Am J Phys Anthropol 1997; 104: 227-243.
- Martin DC, Danforth ME. An analysis of secular change in the human mandible over the last century. Am J Hum Biol 2009; 21: 704-706.