Research Article - Current Pediatric Research (2021) Volume 25, Issue 9
MRI findings in migraine headache in different age groups and relation to Aura.
Rasha Nadeem Ahmed1*, Waseem Hashim Alkhaffaf1, Mohamed Muyaser Naif1, Akram M Al Mahdawi 2
1 Department of Surgery, Ninevah University, Mosul, Iraq
2 Department of Medicine, Al-Mustansyria University, Baghdad, Iraq
- Corresponding Author:
- Rasha Nadeem Ahmed
Department of Surgery
Ninevah University
Mosul
Iraq
Tel: +964 771-291-3336
E-mail: drnihadkhalawe@gmail.com
Accepted date: 15th September, 2021
Abstract
Background: Migraine is a common disorder with well-known association to structural brain changes. In the current study, we aim to identify the relation between structural brain Magnetic Resonance Imaging (MRI) changes and the migraine type as well their relation to the patient’s age. Methods: This cross sectional study was conducted in Iraq from July 2020 to January 2021, on 200 patients with migraine headache, of two age groups (100 patients of less than 18 years and 100 patients of 18-55 years). All the participants had MRI of the brain; the patient’s information and the MRI records were then studied. Results: Patients under age of 18 years (Group 1),were 43 male (43%), 57 female (57%), age range is 3-18 years old, the mean age is 10.36 ± 3.62 years,19 patients (19%) were having positive MRI findings and 17 patients (17%) were having Migraine with Aura (MA). On the other hand, the patients of 18 years old or more (Group 2) were 37 male (37%) and 63 (63%) female, their age range is 19-55 years, mean age is 35 ± 9.97 years, of them 32 patients (32%) have positive MRI findings and 21 patients(21%) had MA. The positive MRI findings, in form of White Matter Hyper Intensities (WMHI), were noted in 51 patients of the total 200 patients (25.5%) include 32% in group 2 and 19% in group 1, total number of WMHI were 120, of them 91 (75.8%) supratentorial and 29 (24.2%) infratentorial. The average No. of WMHI found to be significantly higher in group 2, compared to group 1 (1.9 vs. 2.5 per positive case respectively). The presence of positive MRI changes among MA is shown to be significantly high (p value=0.00), compared to the patients who don’t have an aura (65.8% vs. 16%), with an odds ratio of 10.0 (at 95% confidence interval), likewise this difference is clearly noted if each age were analyzed separately. Conclusion: Structural brain changes in form of WMHI are common MRI findings in the migraineurs patients, and it seems to be strongly associated with aura.
Keywords
White matter hyper intensities, Migraine, Headache, Aura, Brain MRI.
Introduction
Migraine is a common disorder [1]. It affects about 10%-15% of the general population and results in personal as well as social burden [2]. It is also one of the commonest headaches in children, with prevalence of about 3%-10% [3-5]. Migraine is characterized by paroxysmal attacks of headache, and has a close relation to the nervous and cardiovascular system [6-8]. Transient neurological symptoms are noted in some patients with migraine and called as aura which is defined by the international headache society as a recurrent disorder manifesting in attacks of reversible focal neurologic symptoms that usually develop gradually over 5–20 minutes and last for less than 60 minutes migraine with aura described in previous literature as classic migraine, complicated migraine or Migraine with Aura (MA) [9,10].
The aura classically precedes the headache, however is some patients it may starts after the onset of the headache or even happens without headache. Aura has different types, the most common is the visual aura, followed by sensory aura, and then language and motor aura and motor aura is however recently represented as a special type of migraine, known as hemiplegic migraine [10,11]. Although previously migraine was regarded as a benign disorder with no long term Squeal, the main purpose of migraine imaging was to exclude other more serious causes of the headache [12]. It is then founded that migraine and especially MA have been associated with increased risk of cardiovascular diseases and comorbidities and many studies demonstrated that the structural brain changes in form of silent brain infarcts, as well as White Matter Hyper-Intensities (WMHIs) have been associated with migraine [13-17].
The WMHIs are lesions of high signal intensities in the white matter of brain in Magnetic Resonance Imaging (MRI) on long TR (Repetition Time) images they are seen usually in patients with cognitive and motor dysfunctions [18]. Ten percent of normal population as well, may show WMHI, and the incidence of WMHI seems to be increasing with age [19]. The mechanism of WMHI is not fully understood, however, merging data suggest that WMHIs are foci of demyelination and gliosis of ischemic origin although evidences assume a relationship between structural brain changes and migraine however it is not clear how factors like type of migraine, age and sex affect this relationship so we aim in the current study to find out the relation between the brain MRI findings and the type of the migraine as well the relation of these findings to the age or sex [20-22].
Materials and Methods
This cross-sectional study was conducted at the outpatient neurology clinic in Al-Salam teaching hospital in Mosul city between July 2020 and January 2021, patients who complained of headache were evaluated by neurologists, then patients who met the International Classification of Headache Disorders-Third Edition (ICHD-3) diagnostic criteria for migraine (10) were asked to fill out a questionnaire which includes past medical history, drug history, some basic clinical information. Those who fulfill the study inclusion criteria have been invited to participate after full clarification of the study's objectives.
In the same hospital, all the participants then had a brain MRI done to them, The MRI examination was performed by using Philips 1.5 T MRI scanner with a standard 16 channel phase array head coil, the sequences used are T1-weighted, T2-weighted as well FLAIR images. A radiologist and neurologists then examined the MRI records as 100 patients under the age of 18 and 100 patients aged 18-55 years were included in the sample. The Inclusion criteria included: Patients with newly diagnosed migraine, 55 years old or younger, a six month or more duration of symptoms, with no known history of cardiovascular, neurological, metabolic, inflammatory or infectious disease.
The exclusion criteria included: patients aged more than 55 years, short duration of symptoms (less than 6 months), presence of atherosclerotic disease or coexistence of cerebrovascular risk factors, patients who was discovered by MRI to have other pathology like brain tumor, ischemic infarction in the brain or vascular malformation, those with history of chronic drug use like contraceptive pills, or drug or alcohol abuse. All patients who refused the MRI examination were also excluded. 16 patients (6 over 55 years of age), 2 (short duration of symptoms), 5 (coexistence of cardiovascular or neurological diseases), 2 (refusal of MRI), 1 female (contraceptive pill use) were excluded from the study. The study was approved by medical faculty's ethics committee of the Nineveh university. In the case of children, informed consent has been obtained from the participants or their guardians.
Statistics
The data analysis was carried out using SPSS (Statistical Software Package for Social Sciences), version 22.0. For Windows descriptive statistics including frequency, percentage, range, mean, and standard deviation (Std. Dev), were used to summarize the demographic data. Pearson's chi-square test for independence is used to measures the dependency between the categorical variables, including MRI findings and aura, MRI findings and age group, as well MRI findings and sex. The strength of the association between two variables is then expressed in odds ratio. A non-parametric test is used to compare the mean in scale variables with no normal distribution P value below 0.05 was considered to be significant.
Results
The studied sample includes two hundred patients (80 males and 120 females). The age range was (3-55 years) with a mean age of 22.7 years and standard deviation of 14.5 years. The positive MRI findings were observed in 51 patients (25.5%). Thirty-eight (19%) patients were presented with an aura (MA). Patients under 18 years of age (Group 1) (No=100), were 43 males (43%) and 57 females (57%), ranged from 3 to 18 years old, mean age was 10.36 years and 3.62 years of standard deviation 19 patients (19%) had positive MRI findings and 17 patients (17%) had MA. On the other hand, there were 100 patients aged 18 years or more (Group 2), 37 males (37%) and 63 (63%) females, their age ranged between 19 years and 55 years, the mean age was 35 years and 9.97 years of Standard deviation of whom 32 patients (32%) had positive MRI findings and 21 patients (21%) had MA, Table 1.
| Characteristics | Group 1 | Group 2 | ||||||
|---|---|---|---|---|---|---|---|---|
| No. | Range | Mean | Std. Dev. | No. | Range | Mean | Std. Dev. | |
| Age | 100 | 3-18 y | 10.36 | 3.62 | 100 | 19-55 | 35 | 9.97 |
| Frequency | Percent | P-value | Frequency | Percent | P-value | |||
| Gender | ||||||||
| Male | 43 | 43 | 0.16 | 37 | 37 | 0.009 | ||
| Female | 57 | 57 | 63 | 63 | ||||
| MRI findings | ||||||||
| Positive MR | 19 | 19 | 0 | 32 | 32 | 0 | ||
| Negative MR | 81 | 81 | 68 | 68 | ||||
| Type of Headache | ||||||||
| Aura | 17 | 17 | 0 | 21 | 21 | 0 | ||
| Without aura | 83 | 83 | 79 | 79 | ||||
Table 1. Patient’s demographics MRI findings and type of migraine headache in both groups. Group 1: Patients less than 18 years. Group 2: Patients of 18 years and older. y: Years.
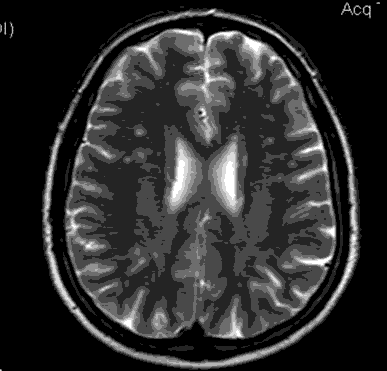
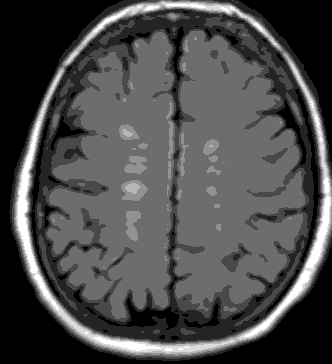
Positive MRI findings were observed in 51 of the 200 patients in total (25.5%). The positive MRI findings were found to be in the form of WMHI (infra and supratentorial) and the total number was 120, of them 91 (75.8%) supratentorial and 29 (24.2%) infratentorial. All WMHI were smaller than 5 mm, in any of the examined patients no brain infarct could be detected, multiple non-related incidental findings were also noted during the exam (Virchow-Robin spaces, arachnoid cyst, with extra cranial findings like sinusitis). The WMHI was found to be present in 32% in group 2 and 19% in group 1, with a statistically significant difference (p=0.035), the average no. of WMHI found to be significantly higher in group 2 (18 years or above) compared to group 1 (below 18 years group) (1.9 per positive case for group 1 and 2.5 per positive case for group 2), with (P=0.04). The presence of WMHI in MRI examinations of adult migraine patients is shown in Figure 1 and 2.
Compared to patients who do not have an aura, the presence of positive MRI changes in MA is shown to be significantly high (P value=0.00), as shown in Table 2, which demonstrates that 65.8% of patients with an aura had positive findings in MRI, compared with 16% only of patients without aura, with an odds ratio of 10.0 at 95% confidence interval. Likewise if each age group (below 18 and above 18 years) has been separately analyzed, this difference is clearly noted, the odds ratio for having positive MRI findings in MA in the first group was 17, while in the second group was 6.7 at 95% confidence interval respectively Table 3.
| Characteristics | Aura | Total | ||||
|---|---|---|---|---|---|---|
| MA | MW | |||||
| Positive | Count | 25 | 26 | 51 | ||
| % | 65.80% | 16.00% | 25.50% | |||
| Negative | Count | 13 | 136 | 149 | ||
| % | 34.20% | 84.00% | 74.50% | |||
| Total | Count | 38 | 162 | 200 | ||
| % | 100.00% | 100.00% | 100.00% | |||
Table 2. Relation of MRI findings to the presence of aura. MA: Migraine with Aura; MW: Migraine without Aura.
| Characteristics | Child | Adult | ||||||
|---|---|---|---|---|---|---|---|---|
| Type | Total | Type | Total | |||||
| MA | MW | MA | MW | |||||
| MRI | Positive | Count | 11 | 8 | 19 | 14 | 18 | 32 |
| % | 64.70% | 9.60% | 19.00% | 66.70% | 22.80% | 32.00% | ||
| Negative | Count | 6 | 75 | 81 | 7 | 61 | 68 | |
| % | 35.30% | 90.40% | 81.00% | 33.30% | 77.20% | 68.00% | ||
| Total | Count | 17 | 83 | 100 | 21 | 79 | 100 | |
| % | 100.00% | 100.00% | 100.00% | 100.00% | 100.00% | 100.00% | ||
Table 3. Relation of MRI findings to the presence of aura according to the age group. MA: Migraine with Aura; MW: Migraine without Aura.
Discussion
Studying brain structural changes in migraine continues to be a subject of many recent studies, linking the presence of white matter changes and silent brain infarcts to migraine as there is still no clear relationship between migraine, presence of WMHIs and brain infarcts, vascular ischemic risks, and the clinical relevance of these WMHIs to migraine [23-25]. A meta-analysis study showed an increased risk of having WMHIs in migraines patients with an odds ratio of 3.9 [26]. In our study, the prevalence of positive MRI findings in form of WMHI was found to be 25.5% (32% in adult group and 19% in child and adolescent group) with a statistically significant difference between the two groups, this finding seems to be logical and consistent with the results of the previous studies which showed that the incidence of MRI WMHI generally increases with advances of age, and that its incidence in migraine patients also increases with advancing age [27-30].
Wide population based studies done in France, USA and Canada showed that females are more commonly affected by migraine than males [31-34] especially at reproductive ages the global burden of disease study in 2015 demonstrated that females are two or three times more commonly affected by migraine, this difference in prevalence may suggest that female hormonal factors may be a determinant factor for migraine risk and characteristics [33,35]. Furthermore some studies observed that decreasing estrogen in hormonal replacement therapy decreases the aura in postmenopausal migraines ladies [35]. In the current study the migraine is shown to be higher in females than in males in both adult and pediatric groups (57% in child and adolescents) (63% in adult group) respectively, however the statistical significance is noted in the adult group only, this finding is consistent with the result of a large study held in the US in 2003 on 40,892 participants and aimed to assess the age and gender prevalence of migraine, the study demonstrated that the one-year prevalence of migraine in boys and girls (until the age of puberty) is similar, then it became more common in females after this age [36].
There is still no clear association between sex and structural brain changes in migraine some studies suggest that the prevalence but not the progression of the structural brain changes in migraine including WMHI and brain infarcts is more common in others denied this association [22,25,37]. In our study WMHIs were identified in 51 of 200 cases (25.5%) of them 29 were female (56.8%), with no significant sex difference in MRI findings among migraine patients (p=0.5). The previous literature showed that the percentage of aura among migraine patients ranged from 14%-54% [38-40]. In our study, 19% of the total 200 patients suffered from MA (17% in child, and 21% in adult), some studies including one large epidemiological study held by Rasmussen et al. demonstrated that migraines females are more commonly affected by aura than males [41]. In our study the percent of MA is higher in females than in males (60.5% vs. 39.5% respectively). However, no association between female sex and aura can be statistically proved.
The association of structural brain changes in migraine with aura is a subject of debates, several studies including meta-analysis study held by Bashir et al. demonstrated a strong association between MA and structural brain changes, including WMHs, silent brain infarct and volumetric changes in grey and white matter, others fail to prove such an association [9,17,42,43]. In our study, the presence of positive MRI changes among MA is shown to be significantly of high prevalence (p-value=0.00), compared to the patients who don’t have an aura, with an odds ratio of 10.0. In the present study, the MRI findings are in the form of WMHI, supratentorial WMHIs are the commonest findings, no brain infarct or grey matter changes are noted in any of the studied cases, and these findings are comparable to Pavese et al. and Uggetti et al. The limitations of our study include the cross-sectional type of the study which may show relations between events but not the sequential effect. In addition, the strict exclusion criteria used in the selection of the patients may carry some bias, however their no. is considered small compared to the sample, so that it has only a minor impact on the results.
Conclusion
Structural brain changes in form of WMHI are common MRI findings in the migraine patients. The presence of aura has been shown to be strongly associated with an increased risk of getting WMHI in migraine. This theory can lead to the conclusion that the aura may be a strong sign of ischemic brain changes, thus further longitudinal studies are required to emphasize this relationship and to study the underlying pathophysiology. Also, this study makes us suggest that the presence of aura may be one of the indicators of neuroimaging for migraine patients. Advancing age and female sex in adult, but not in children, are other factors that have been shown to be associated with brain WMHI in migraine but however, to a lesser extent.
Abbreviation
ICHD-3: The International Classification of Headache Disorders-Third Edition; MA: Migraine with Aura; MRI: Magnetic Resonance Imaging; MW: Migraine without Aura; No: Number; Std. Dev: Standard Deviation; TR: Time of Repetition; WMHI: White Matter Hyperintensities.
References
- Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: (2nd edn). Cephalalgia. 2004; 24 (Suppl 1): 9-160.
- Rasmussen BK, Jensen R, Schroll M, et al. Epidemiology of headache in a general population-a prevalence study. J Clin Epidemiol 1991; 44(11): 1147-57.
- Abu-Arefeh I, Russell G. Prevalence of headache and migraine in school children. BMJ 1994.
- Raieli V, Raimondo D, Cammalleri R, et al. Migraine headaches in adolescents: a student population-based study in Monreale. Cephalalgia 1995; 15(1): 5-12.
- Sillanpaa M. Prevalence of headache in prepuberty. Headache 1983; 23(1): 10-4.
- Jensen R, Stovner LJ. Epidemiology and comorbidity of headache. Lancet Neurol 2008; 7(4): 354-61.
- StovnerLj, Hagen K, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia 2007; 27(3): 193-210.
- Kurth T, Gaziano JM, Cook NR, et al. Migraine and risk of cardiovascular disease in women. JAMA 2006; 296(3): 283–291.
- Kurth T, Mohamed S, Maillard P, et al. Headache, migraine, and structural brain lesions and function: population based Epidemiology of vascular ageing-MRI study. BMJ 2011.
- https://ichd-3.org/1-migraine/
- Cutrer FM, Huerter K. Migraine aura. Neurologist 2007; 13(3): 118-25.
- Moschiano F, Amico DD, Rocca N, et al. The role of the clinician in interpreting conventional neuroimaging findings in migraine patients. Neurol Sci 2007; 28: 114-117.
- Hadjikhani N, Wu O, Schwartz D, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci USA 2001; 98(8): 4687-4692.
- Gaist D, Garde E, Blaabjerg M, et al. Migraine with aura and risk of silent brain infarcts and white matter hyperintensities: an MRI study. Brain 2016; 139(7): 2015-2023.
- Debette S, Markus HS. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ 2010.
- Palm Meinders IH, Koppen H, Terwindt GM, et al. Structural brain changes in migraine. JAMA 2012; 308(18): 1889-1897.
- Hamedani AG, Rose KM, Peterlin BL, et al. Migraine and white matter hyperintensities: the ARIC MRI study. Neurology 2013; 81(15): 1308-13.
- Pavese N, Canapicchi R, Nuti A, et al. White matter MRI hyperintensities in a hundred and twenty-nine consecutive migraine patients. Cephalalgia 1994; 14(5): 342-345.
- De Groot JC, de Leeuw FE, Oudkerk M, et al. Cerebral white matter lesions and cognitive function: the Rotterdam Scan Study. Ann Neurol 2000; 47(2): 145-51.
- Reisman M, Fuller CJ. Is patent foramen ovale closure indicated for migraine?: patent foramen ovale closure for migraine. Circ Cardiovasc Interv 2009; 2(5): 468-474.
- Fazekas F, Kleinert R, Offenbacher H, et al. Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology 1993; 43(9): 1683-9.
- Hougaard A, Amin FM, Ashina M. Migraine and structural abnormalities in the brain. Curr Opin Neurol 2014; 27(3): 309-14.
- Jin C, Yuan K, Zhao L, et al. Structural and functional abnormalities in migraine patients without aura. NMR Biomed 2013; 26(1): 58-64.
- Alkhaffaf WH, Naif MM, Ahmed RN. The association of MRI findings in migraine with the headache characteristics and response to treatment. Revista Latinoamericana de Hipertension 2020; 15: 345-351.
- Trauninger A, Leel-Ossy E, Kamson DO, et al. Risk factors of migraine-related brain white matter hyper intensities: an investigation of 186 patients. J Headache Pain 2011; 12(1): 97-103.
- Swartz RH, Kern RZ. Migraine is associated with magnetic resonance imaging white matter abnormalities: a meta-analysis. Arch Neurol 2004; 61(9): 1366-1368.
- Lee MJ, Moon S, Chung CS. White matter hyperintensities in migraine: A review. Precision and Future Medicine 2019; 3(4): 146-57.
- Soderlund H, Nyberg L, Adolfsson R, et al. High prevalence of white matter hyperintensities in normal aging: relation to blood pressure and cognition. Cortex 2003; 39: 1093-105.
- Hopkins RO, Beck CJ, Burnett DL, et al. Prevalence of white matter hyperintensities in a young healthy population. J Neuroimaging 2006; 16(3): 243-51.
- Cooney BS, Grossman RI, Farber RE, et al. Frequency of magnetic resonance imaging abnormalities in patients with migraine. Headache 1996; 36(10): 616-21.
- Henry P, Auray JP, Gaudin AF, et al. Prevalence and clinical characteristics of migraine in France. Neurology 2002; 59(2): 232-7.
- Lipton RB, Stewart WF, Diamond S, et al. Prevalence and burden of migraine in the United States: data from the American Migraine Study II. Headache 2001; 41(7): 646-57.
- Vosoughi K, Stovner LJ, Steiner TJ, et al. The burden of headache disorders in the Eastern Mediterranean Region, 1990-2016: findings from the Global Burden of Disease study 2016. J Headache Pain 2019; 20(1): 40.
- Brien OB, Goeree R, Streiner D. Prevalence of migraine headache in Canada: a population-based survey. Int J Epidemiol 1994; 23(5): 1020-1026.
- MacGregor A. Estrogen replacement and migraine aura. Headache 1999; 39(9): 674-678.
- Victor TW, Hu X, Campbell JC, et al. Migraine prevalence by age and sex in the United States: a life-span study. Cephalalgia 2010; 30(9): 1065-1072.
- Kruit MC, Van Buchem MA, Hofman PA, et al. Migraine as a risk factor for subclinical brain lesions. JAMA 2004; 291(4): 427-434.
- Kelman L. The aura: a tertiary care study of 952 migraine patients. Cephalalgia 2004; 24(9): 728-734.
- Avci AY, Lakadamyali H, Arikan S, et al. High sensitivity C-reactive protein and cerebral white matter hyperintensities on magnetic resonance imaging in migraine patients. J Headache Pain 2015; 16: 9.
- Rastogi RG, VanderPluym J, Lewis KS. Migrainous aura, visual snow, and alice in wonderland syndrome in childhood. Semin Pediatr Neurol 2016; 23(1): 14-17.
- Rasmussen BK, Olesen J. Migraine with aura and migraine without aura: an epidemiological study. Cephalalgia 1992; 12(4): 221-228.
- Bashir A, Lipton RB, Ashina S, et al. Migraine and structural changes in the brain: a systematic review and meta-analysis. Neurology 2013; 81(14): 1260-1268.
- Uggetti C, Squarza S, Longaretti F, et al. Migraine with aura and white matter lesions: an MRI study. Neurol Sci 2017; 38: 11-13.