Research Article - Biomedical Research (2017) Volume 28, Issue 20
MR spectroscopy features of normal appearing white matter in patients with lung cancer
Adil Dogan1*, Ahmet Sigirci2, Hilal Ermis3, Zeynep Ayfer Aytemur3, Tamer Baysal4, Alpay Alkan5, Saim Yologlu6
1Department of Radiology, K?r?kkale Universitesi T?p Fakültesi, Turkey
2Department of Radiology, ?nönü Üniversitesi Turgut Özal T?p Merkezi, Turkey
3Department of Chest Medicine, ?nönü Üniversitesi Turgut Özal T?p Merkezi, Malatya, Turkey
4Department of Radiology, ?stanbul Kartal Dr. Lütfi K?rdar E?itim ve Ara?t?rma Hastanesi, Turkey
5Department of Radiology, Bezmialem Vak?f Üniversitesi T?p Fakültesi, Turkey
6Department of Biostatistics, ?nönü Üniversitesi Turgut Özal T?p Merkezi, Turkey
- *Corresponding Author:
- Adil Do?an
Department of Radiology K?r?kkale Üniversitesi T?p Fakültesi Turkey
Accepted date: September 23, 2017
Abstract
Context: Spectroscopic imaging can be helpful to identify metabolite changes in a normal appearing brain parenchyma after MR imaging in patients with lung cancer. Aims: To detect metabolic changes in the cerebellar and cerebral normal appearing white matter with single-voxel H-MR spectroscopy (MRS) in patients with lung cancer.
Methods and material: MRS was performed in the Cerebellar White Matter (CWM) and Parietal White Matter (PWM) of patients with lung cancer (n=38; age range: 44-82 y; mean age: 66.1 ± 8.7) and an agematched control group (n=32; age range: 42-80 y; mean age: 62.9 ± 8). Biopsies of lung masses showed 25 Non-Small Cell Lung Cancer (NSCLC) and 13 Small Cell Lung Cancer (SCLC). NAA/Cr, NAA/Cho and Cho/Cr ratios were calculated. Statistical analysis used: The unpaired-t test was used for the assessment of metabolite ratio differences between patients and control subjects. Mann-Whitney U test was used among SCLC, NSCLC, and control group for the evaluation of metabolite ratio differences.
Results: The Cho/Cr ratios in the CWM were significantly lower in the patients compared to controls (p=0.03). The Cho/Cr ratio of patients with SCLC was significantly lower in PWM than both NSCLC (p=0.02) and control group (p=0.03).
Conclusion: Decreased Cho/Cr ratio possibly represents increased uptake of Cho or reduced brain function. These results may be important in assessing patients with lung cancer in order to help with treatment planning and prognosis.
Keywords
Brain, Lung cancer, Magnetic resonance spectroscopy.
Introduction
Lung cancer is the most common cause of cancer-related death in men and women, at 31% and 26%, respectively [1]. Brain metastasis is an important issue in the overall management of lung cancer since brain metastases may develop in up to 65% of cases [2]. Approximately 80% of brain metastases develop during the first two years of Small-Cell Lung Cancer (SCLC) [3]. Brain metastases also develop in 30% of patients with non- SCLC (NSCLC) [4].
Evaluation of the metabolic status of Normal Appearing White Matter (NAWM) may be critical in patients with lung cancer in order to help with treatment planning and prognosis. Proton Magnetic Resonance Spectroscopy (MRS) has made it possible to analyse several brain metabolites [5], such as metabolite changes in NAWM that are below the threshold of detection by Magnetic Resonance Imaging (MRI) [6,7]. The goal of this study was to use MRS to demonstrate cerebellar and cerebral NAWM neurometabolic changes in patients with lung cancers compared to a control group.
Subjects and Methods
44 consecutive patients (41 males, 3 females, age range: 44-82 y, mean age: 65.9 ± 8.4) were initially included in this prospective study between September 2010 to August 2012.
These patients were referred to the Radiology Department from the Department of Chest Diseases with the diagnosis of lung masses seen on their chest X-rays and consequent thorax CT. Of these patients, 6 were excluded. Three of them were excluded from the study due to brain metastases and the other 3 who did not want to participate in our study. As a result, we had a total of 38 patients (36 males, 2 females, age range: 44-82 y; mean age: 66.1 ± 8.7). 13 of 38 lung masses found to be SCLC and the remaining 25 were NSCLC (20 squamous cell carcinoma, 4 adenocarcinoma, and 1 large cell carcinoma) by biopsy.
Our control group comprised 32 age-matched healthy volunteers (29 males, 3 females, age range: 42-80; mean age: 62.9 ± 8). They were examined clinically and by chest X- rays to exclude lung cancer at that time. Exclusion criteria for both groups included systemic disease, medications, psychiatric, and neurological problems. The patients and controls on average had been smoking 1 pack of cigarettes daily for 22 and 18 years, respectively. Informed, written consent was obtained from all participants, and the institutional ethics committee on human subject approved the study.
All participants underwent brain MRI examinations that consisted of conventional imaging and Single-Voxel H-MR Spectroscopy (SVS). MRI was performed on a 1.5 T scanner (Gyroscan Intera Master, Philips, Best, Netherlands). Axial T1- weighted (TR: 450, TE: 10 ms), axial and sagittal T2- weighted (TR: 4366, TE: 120 ms), and coronal fluid attenuated inversion recovery (TR: 6000, TE: 110, TI: 2000 ms) images were obtained from patients and healthy subjects. Axial, coronal, and sagittal gadolinium enhanced images were acquired after unenhanced images had been taken.
SVS was obtained from all participants by using a pointresolved spectroscopy sequence (TR: 2000/TE: 136 ms) acquired in the Cerebellar (CWM) and Parietal White Matters (PWM) with a voxel size of 17 × 17 × 17 mm3 and number of excitations equal 128. Prior to MRS, shimming was performed to optimize field homogeneity and water suppression was optimized using automated routines. The water signal was suppressed with a chemical shift selective saturation pulse. A spectral sweep width of 1000 Hz and a data size of 1024 points were used. All data post-processing was performed using software provided by the manufacturer (MMR 5461 1H spectroscopy 1.5 T package). Spectra were processed automatically using baseline correction and curve-fitting procedures to determine the resonance areas of N-Acetyl- Aspartate (NAA), Creatine (Cr), and Choline (Cho). Resonances were assigned as follows: NAA, 2.0 ppm; Cr, 3.02 ppm; and Cho, 3.2 ppm. Peak area metabolite ratios (NAA/Cr, Cho/Cr and NAA/Cho) were calculated.
All statistical analyses were performed using the Statistical Package for Social Sciences version 13.0 (SPSS Inc., Chicago, IL, USA). The results are reported as mean ± standard deviation. Normality for continuous variables in groups was determined by the Shapiro Wilks test. The variables manifested normal distribution (p ≥ 0.05). The unpaired-t test was used for the assessment of metabolite ratio differences between patients and control subjects. Mann-Whitney U test was used among SCLC, NSCLC, and control group for the evaluation of metabolite ratio differences. A value of p<0.05 was considered statistically significant.
Results
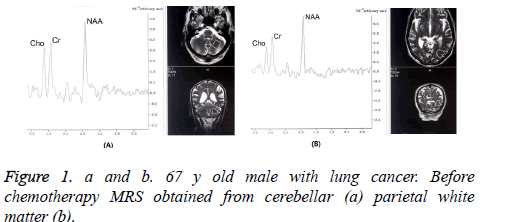
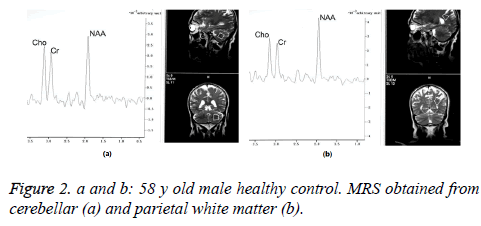
Between the patients and controls, there were no statistically significant age differences. All participants had normal conventional MRI findings. Tables 1 and 2 show their mean metabolite ratios for both CWM and PWM. Cho/Cr ratios in the CWM were significantly higher in the controls compared to those of the patients (1.05 ± 0.27 and 0.89 ± 0.13, respectively, p=0.03) (Figures 1 and 2).
| CWM | |||
|---|---|---|---|
| NAA/Cr | NAA/Cho | Cho/Cr | |
| Patients (n=38) | 1.38 ± 0.21 | 1.52 ± 0.33 | 0.89 ± 0.13a |
| Control (n=32) | 1.40 ± 0.40 | 1.39 ± 0.36 | 1.05 ± 0.27a |
ap=0.03; CWM: Cerebellar White Matter.
Table 1: Magnetic resonance spectroscopy findings of controls and patients from cerebellar white matter before treatment.
| PWM | |||
|---|---|---|---|
| NAA/Cr | NAA/Cho | Cho/Cr | |
| Patients (n=38) | 1.78 ± 0.27 | 1.90 ± 0.18 | 0.96 ± 0.21 |
| Control (n=32) | 1.83 ± 0.39 | 1.85 ± 0.36 | 1.03 ± 0.21 |
PWM: Parietal White Matter
Table 2: Magnetic resonance spectroscopy findings of controls and patients from parietal white matter before treatment.
Tables 3 and 4 show the mean metabolite ratios of the controls compared to the SCLC and NSCLC patients. The Cho/Cr ratios of both group of patients were significantly lower in CWM (p=0.03 and p=0.04, respectively) than in the control group (Table 3). The Cho/Cr ratio of patients with SCLC was significantly lower in the PWM of both the NSCLC (p=0.02) and the control group (p=0.03) (Table 4).
| CWM | ||
|---|---|---|
| NAA/Cr | Cho/Cr | |
| SCLC (n=13) | 1.38 ± 0.34 | 0.87 ± 0.18a |
| NSCLC (n=25) | 1.35 ± 0.43 | 0.90 ± 0.01b |
| Control (n=32) | 1.40 ± 0.40 | 1.05 ± 0.27a,b |
ap=0.04; bp=0.03; CWM: Cerebellar White Matter; NSCLC: Non-Small Cell Lung Cancer; SCLC: Small Cell Lung Cancer.
Table 3: Magnetic resonance spectroscopy findings of patients with small cell and non-small cell lung cancers and controls from cerebellar white matter before treatment.
| PWM | ||
|---|---|---|
| NAA/Cr | Cho/Cr | |
| SCLC (n=13) | 1.76 ± 0.20 | 0.90 ± 0.23a,b |
| NSCLC (n=25) | 1.71 ± 0.58 | 1.06 ± 0.14a |
| Control (n=32) | 1.83 ± 0.39 | 1.03 ± 0.21b |
ap=0.02; bp=0.03; PWM: Parietal White Matter; NSCLC: Non-Small Cell Lung Cancer; SCLC: Small cell lung cancer.
Table 4: Magnetic resonance spectroscopy findings of patients with small cell and non-small cell lung cancers and controls from parietal white matter before treatment.
Discussion
Evaluation of the metabolic status of NAWM may be necessary before making decisions about chemotherapy, whole brain radiotherapy, or prognosis in the patients with lung cancer. This study has two major findings. The first one is that normalappearing CWM of cancer patients has a significantly lower Cho/Cr ratio compared to controls. The second finding is that the normal-appearing PWM in patients with SCLC has a Cho/Cr ratio significantly lower than that of both the NSCLC and of the controls. Our results support the hypothesis that significantly altered metabolism in normal appearing CWM and PWM can be detected in lung cancer patients.
Cho peaks represent cellular membrane density and turnover [8,9]. Elevated Cho represents active inflammation and demyelination, as well as membrane proliferation in reactive astrocytes [6]. Cho/Cr ratios for all patients in CWM and for SCLC in both CWM and PWM were significantly lower than in the controls. Our expectation was that the Cho/Cr ratio would be higher in patients than in the controls. Therefore, these findings were remarkable. As a result, MRS could be helpful for classifying brain metastases into three stages: early (evidenced by increased Cho), intermediate (further increase of Cho plus presence of lipids), and late (decreased Cho and presence of lactate) [10,11]. Although MRI of our patients showed no metastases, lung masses were anticipated to have subtle effects on the brain.
Reduced Cho/Cr ratio would indicate myelin damage [12] or decelerated turnover of membrane [13]. Using proton MRS, Ciszkowska-Lyson et al. [12] evaluated the neurotoxicity of chemotherapy in 31 patients with lung cancer and found that the cerebellar Cho/Cr ratios had significantly decreased after chemotherapy but the cerebral ratios had not.
The other reason for decreased Cho/Cr ratio could be increased uptake of Cho from the free phase for the building of cell membranes [7,14]. Gustafsson et al. [7] showed decreased concentration of Cho in the NAWM of patients who had multiple sclerosis. They explained this finding as either an increased ability to protect against lesions or a decreased ability to form lesions. In our study, the decrease in the Cho/Cr ratio may be the result of increased uptake of Cho due to the protective reflex of cells to the lung cancer.
Benveniste et al. [15] used short-TE MRS on a 3 T machine and reported that absolute concentration changes of Glutamate (Glu) in the occipital cortex was, before treatment, lower in patients who had lung cancer than in the controls. They found no difference in the concentrations of other metabolites. Cerebellar metabolite concentrations were not evaluated in their study. Their explanation for the decrease in Glu was that Glu represents brain energy metabolism, and that prior to treatment, the presence of lung cancer itself reduces brain function. The brain may respond to reduced function by increasing Cr concentration thus leading to a decrease in the Cho/Cr ratio. The reduction in brain function before treatment of lung cancer could be an explanation of our findings.
Haga et al. [16] performed a systematic literature review on MRS metabolites in older subjects to determine whether metabolites change during the course of healthy aging. They found that NAA may decrease, and choline and creatine do increase with age. Gruber et al. [17] revealed reduced NAA/Cr and elevated Cho/Cr in an older group compared to a younger group. In our study, the mean age of patients and controls were approximately the same; however, Cho/Cr ratios were lower in the patients than in the controls. Age does not account for our results.
The major limitation of our study is the small sample size of the patients with SCLC thus tumor staging could not be correlated with metabolite ratios. The other limitation is SVS was used instead of multivoxel for MRS that a restriction of our MRI machine. The patients were evaluated after being diagnosed as having early stage lung cancer. We had planned to evaluate metastases, but had difficulty in enrolling patients who had metastatic lung cancer and in differentiating the effects of chemotherapy versus metastases on MRS.
In conclusion, our lung cancer patients had a lower Cho/Cr ratio in NAWM, both in CWM and in PWM. The reason could be increased uptake of Cho for the building of cell membranes, or reduced brain function. Such information may be important in assessing patients with lung cancer in order to help with treatment planning and prognosis.
References
- Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin 2010; 60: 277-300.
- Johnson JD, Young B. Demographics of brain metastases. Neurosurg Clin N Am 1996; 7: 337-344.
- Gavrilovic IT, Posner JB. Brain metastases: Epidemiology and pathophysiology. J Neurooncol 2005; 75: 5-14.
- Kelly K, Bunn PA. Is it time to reevaluate our approach to the treatment of brain metastases in patients with non-small cell lung cancer? Lung Cancer 1998; 20: 85-91.
- Ratai EM, Pilkenton S, Lentz MR, Greco JB, Fuller RA, Kim JP. Comparisons of brain metabolites observed by HRMAS 1H NMR of intact tissue and solution 1H NMR of tissue extracts in SIV-infected macaques. NMR Biomed 2005; 18: 242-251.
- Alkan A, Sarac K, Kutlu R, Yakinci C, Sigirci A, Aslan M. Proton MR spectroscopy features of normal appearing white matter in neurofibromatosis type 1. Magn Reson Imaging 2003; 21: 1049-1053.
- Gustafsson MC, Dahlqvist O, Jaworski J, Lundberq P, Landtblom AM. Low choline concentrations in normal appearing white matter of patients with multiple sclerosis and normal MR imaging brain scans. AJNR Am J Neuroradiol 2007; 28: 1306-1312.
- Saindane AM, Cha S, Law M, Xue X, Knopp EA, Zagzag D. Proton MR spectroscopy of tumefactive demyelinating lesions. AJNR Am J Neuroradiol 2002; 23: 1378-1386.
- Poptani H, Gupta RK, Roy R, Panday R, Jain VK, Chhabra DK. Characterization of intracranial mass lesions with in vivo proton MR spectroscopy. AJNR Am J Neuroradiol 1995; 16: 1593-1603.
- Sijens PE, Levendag PC, Vecht CJ, van Dijk P, Oudkerk M. 1H MR spectroscopy detection of lipids and lactate in metastatic brain tumors. NMR Biomed 1996; 9: 65-71.
- Sijens PE, van Dijk P, Oudkerk M. Correlation between choline level and Gd-DTPA enhancement in patients with brain metastases of mammary carcinoma. Magn Reson Med 1994; 32: 549-555.
- Ciszkowska-Lyson B, Krolicki L, Teska A, Janowicz-Zebrowska A, Krzakowski M, Tacikowska M. Brain metabolic disorders after chemotherapy in the study by magnetic resonance spectroscopy. Neurol Neurochir Pol 2003; 37: 783-798.
- Hong ST, Choi CB, Park C, Moon HY, Hong KS, Cheong C. Specific hippocampal choline decrease in an animal model of depression. Br J Radiol 2009; 82: 549-553.
- Richards TL, Alvard EC, He Y, Petersen K, Peterson J, Cosgrove S. Experimental allergic encephalomyelitis in non-human primates: diffusion imaging of acute and chronic brain lesions. Mult Scler 1995; 1: 109-117.
- Benveniste H, Zhang S, Reinsel RA, Li H, Lee H, Rebecchi M. Brain metabolomics profiles of lung cancer patients prior to treatment characterized by proton magnetic resonance spectroscopy. Int J Clin Exp Med 2012; 5: 154-164.
- Haga KK, Khor YP, Farral A, Wardlaw JM. A systematic review of brain metabolite changes, measured with H magnetic resonance spectroscopy, in healthy aging. Neurobiol Aging 2009; 30: 353-363.
- Gruber S, Pinker K, Riederer F, Chmelik M, Stadlbauer A, Bittsansky M. Metabolic changes in the normal aging brain: consistent findings from short and long echo time proton spectroscopy. Eur J Radiol 2008; 68: 320-327.